Chemistry:Benzocyclobutene
Benzocyclobutene (BCB) is a benzene ring fused to a cyclobutane ring. It has chemical formula C
8H
8.[1]
BCB is frequently used to create photosensitive polymers. BCB-based polymer dielectrics may be spun on or applied to various substrates for use in Micro Electro-Mechanical Systems (MEMS) and microelectronics processing. Applications include wafer bonding, optical interconnects, low-κ dielectrics, or even intracortical neural implants.
Reactions
Benzocyclobutene is a strained system which, upon heating to approximately 180 °C, causes the cyclobutene to undergo a conrotatory ring-opening reaction, forming o-xylylene. Since this process destroys the aromaticity of the benzene ring, the reverse reaction is highly favored.
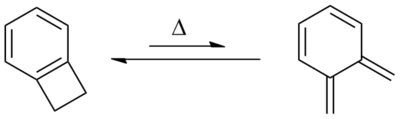
o-Xylylenes generated in this way have been used prolifically in cycloaddition reactions, which restore the aromaticity to the benzene ring, while forming a new annulated species.[2]
Derivatives
The benzocyclobutene moiety has also appeared in a number of chemical compounds with pharmacological properties such as ivabradine and S33005.
Additionally, cyclized phenethylamine psychedelic drugs containing the benzocyclobutene ring system, including TCB-2, tomscaline, bromotomscaline, and 2CBCB-NBOMe (NBOMe-TCB-2), have been described.[3] A benzocyclobutene-derived amphetamine has been patented as well,[4] and a benzocyclobutene containing designer drug, the substituted cathinone derivative 3,4-EtPV, has been sold in Europe.[5] The benzocyclobutene derivative 2C-G-2 has also been claimed in a patent as an antiinflammatory,[6] but no synthesis or activity data is provided so it is unclear if it has actually been made.
See also
References
- ↑ 164410 Benzocyclobutene 98%
- ↑ Mehta, G.; Kotha, S. (2001). "Recent chemistry of benzocyclobutenes". Tetrahedron Lett. 57 (4): 625–659. doi:10.1016/s0040-4020(00)00958-3. http://eprints.iisc.ac.in/2691/1/01-SRK-Tetra-625.pdf.
- ↑ Trachsel, D.; Lehmann, D.; Enzensperger, C. (2013) (in de). Phenethylamine: von der Struktur zur Funktion. Nachtschatten-Science (1 ed.). Solothurn: Nachtschatten-Verlag. ISBN 978-3-03788-700-4. OCLC 858805226. https://books.google.com/books?id=-Us1kgEACAAJ. Retrieved 30 November 2025.
- ↑ "Substituted 7-aminoalkylbicyclo-[4. 2. 0]octa-1,3,5-trienes" US patent 3149159
- ↑ "European Drug Report 2025 – Full Book". European Union Drugs Agency (euda.europa.eu). 6 June 2025. https://www.euda.europa.eu/system/files/documents/2025-06/edr-2025-full-book-6.06.2025-en.pdf.
- ↑ Halpert M. Substituted phenethylamine for treating inflammation and psychological disorders. WO 2022/271982
 |
