Chemistry:Substituted cathinone


Substituted cathinones, which include some stimulants and entactogens, are derivatives of cathinone. They feature a phenethylamine core with an alkyl group attached to the alpha carbon, and a ketone group attached to the beta carbon, along with additional substitutions.[1][2][3][4][5] Cathinone occurs naturally in the plant khat whose leaves are chewed as a recreational drug.[6]
List of substituted cathinones
The derivatives may be produced by substitutions at four locations of the cathinone molecule:
- R1 = hydrogen, or any combination of one or more alkyl, alkoxy, alkylenedioxy, haloalkyl or halide substituents
- R2 = hydrogen or any alkyl group
- R3 = hydrogen, any alkyl group, or incorporation in a cyclic structure
- R4 = hydrogen, any alkyl group, or incorporation in a cyclic structure
The following table displays notable derivatives that have been reported:[7][8][9][10][11][12][13][14][15][16][17][18][19][20][21][22][23][24][25][26][27][28]
| Structure | Compound | R1 | R2 | R3 | R4 | CAS number |
|---|---|---|---|---|---|---|
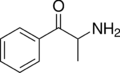 |
Cathinone | H | Me | H | H | 71031-15-7 |
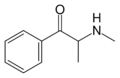 |
Methcathinone | H | Me | H | Me | 5650-44-2 |
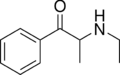 |
Ethcathinone | H | Me | H | Et | 51553-17-4 |
 |
Propylcathinone | H | Me | H | nPr | 52597-14-5 |
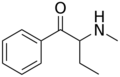 |
Buphedrone | H | Et | H | Me | 408332-79-6 |
| Error creating thumbnail: Unable to save thumbnail to destination | N-Ethylbuphedrone (NEB) | H | Et | H | Et | 1354631-28-9 |
 |
N-Methyl-N-ethylbuphedrone | H | Et | Me | Et | |
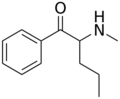 |
Pentedrone | H | nPr | H | Me | 879722-57-3 |
 |
N-Ethylpentedrone | H | nPr | H | Et | 18268-16-1 |
 |
N-Isopropylpentedrone | H | nPr | H | iPr | 18268-14-9 |
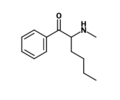 |
Hexedrone | H | nBu | H | Me | 2169446-41-5 |
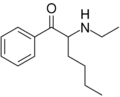 |
N-Ethylhexedrone | H | nBu | H | Et | 18410-62-3 |
 |
N-Butylhexedrone | H | nBu | H | nBu | 18296-66-7 |
 |
N-Isobutylhexedrone (NDH) | H | nBu | H | i-Bu | |
 |
Isohexedrone | H | iBu | H | Me | |
 |
N-Ethylheptedrone | H | nPe | H | Et | |
 |
Octedrone | H | hexyl | H | Me | |
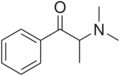 |
Dimethylcathinone | H | Me | Me | Me | 15351-09-4 |
 |
Diethylpropion | H | Me | Et | Et | 134-80-5 |
 |
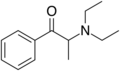
N-Methyl-N-ethylcathinone | H | Me | Me | Et | 1157739-24-6 |
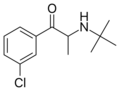 |
Bupropion | 3-Cl | Me | H | t-Bu | 34911-55-2 |
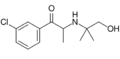 |
Hydroxybupropion | 3-Cl | Me | H | 2-Me-3-OH-propan-2-yl | 357399-43-0 |
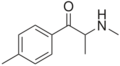 |
Mephedrone | 4-Me | Me | H | Me | 1189805-46-6 |
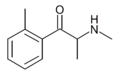 |
2-MMC | 2-Me | Me | H | Me | 1246911-71-6 |
 |
2-MEC | 2-Me | Me | H | Et | 1439439-84-5 |
 |
2-EMC | 2-Et | Me | H | Me | |
 |
2-EEC | 2-Et | Me | H | Et | 2446466-59-5 |
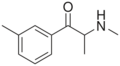 |
3-MMC | 3-Me | Me | H | Me | 1246816-62-5 |
 |
3-MEC | 3-Me | Me | H | Et | 1439439-83-4 |
 |
3-MPC | 3-Me | Me | H | nPr | |
 |
3-EMC | 3-Et | Me | H | Me | |
 |
3-EEC | 3-Et | Me | H | Et | 2446466-61-9 |
 |
4-EMC | 4-Et | Me | H | Me | 1225622-14-9 |
 |
4-EEC | 4-Et | Me | H | Et | 2446466-62-0 |
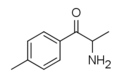 |
4-MC | 4-Me | Me | H | H | 31952-47-3 |
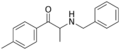 |
Benzedrone | 4-Me | Me | H | Bn | 1225617-75-3 |
| Error creating thumbnail: | 2'-MeO-Benzedrone | 4-Me | Me | H | 2-MeO-Bn | |
| File:2,N-DM-Benzedrone structure.png | 2,N-Dimethylbenzedrone | 2-Me | Me | Me | Bn | |
| File:3,N-DM-Benzedrone structure.png | 3,N-Dimethylbenzedrone | 3-Me | Me | Me | Bn | |
| File:4,N-DM-Benzedrone structure.png | 4,N-Dimethylbenzedrone | 4-Me | Me | Me | Bn | |
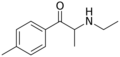 |
4-MEC | 4-Me | Me | H | Et | 1225617-18-4 |
| File:4-methyl-propylcathinone structure.png | 4-MPC | 4-Me | Me | H | nPr | |
| File:NN-DMMC structure.png | N,N-DMMC | 4-Me | Me | Me | Me | 1448845-14-4 |
| File:NN-MEMC structure.png | N,N-MEMC | 4-Me | Me | Me | Et | |
 |
N,N-DEMC | 4-Me | Me | Et | Et | 676316-90-8 |
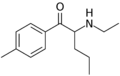 |
4-MEAP | 4-Me | Pr | H | Et | 746540-82-9 |
 |
EDMC | 4-Et | Me | Me | Me | |
 |
2,3-DMMC | 2,3-dimethyl | Me | H | Me | |
 |
2,3-DMEC | 2,3-dimethyl | Me | H | Et | |
 |
2,4-DMMC | 2,4-dimethyl | Me | H | Me | 1225623-63-1 |
 |
2,4-DMEC | 2,4-dimethyl | Me | H | Et | 1225913-88-1 |
 |
2,5-DMMC | 2,5-dimethyl | Me | H | Me | |
 |
2,5-DMEC | 2,5-dimethyl | Me | H | Et | |
 |
2,6-DMMC | 2,6-dimethyl | Me | H | Me | |
 |
2,6-DMEC | 2,6-dimethyl | Me | H | Et | |
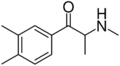 |
3,4-DMMC | 3,4-dimethyl | Me | H | Me | 1082110-00-6 |
 |
3,4-DMEC | 3,4-dimethyl | Me | H | Et | 1225811-81-3 |
 |
3,5-DMEC | 3,5-dimethyl | Me | H | Et | |
 |
2,4,5-TMMC | 2,4,5-trimethyl | Me | H | Me | 1368603-85-3 |
 |
2,4,5-TMOMC | 2,4,5-trimethoxy | Me | H | Me | |
 |
3,4,5-TMOMC | 3,4,5-trimethoxy | Me | H | Me | |
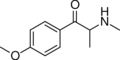 |
Methedrone | 4-MeO | Me | H | Me | 530-54-1 |
 |
Dimethedrone | 4-MeO | Me | Me | Me | 91564-39-5 |
 |
Ethedrone | 4-MeO | Me | H | Et | |
 |
2-MOMC | 2-MeO | Me | H | Me | |
 |
3-MOMC | 3-MeO | Me | H | Me | 1435933-70-2 |
 |
3-FC | 3-F | Me | H | H | 1082949-91-4 |
 |
4-FC | 4-F | Me | H | H | 80096-51-1 |
 |
2-FMC | 2-F | Me | H | Me | 1186137-35-8 |
 |
2-FEC | 2-F | Me | H | Et | |
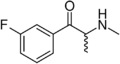 |
3-FMC | 3-F | Me | H | Me | 1049677-77-1 |
 |
3-FEC | 3-F | Me | H | Et | |
 |
2-CMC | 2-Cl | Me | H | Me | |
 |
2-BMC | 2-Br | Me | H | Me | |
 |
2-IMC | 2-I | Me | H | Me | |
 |
2-TFMAP | 2-CF3 | Me | H | Me | |
 |
Clophedrone | 3-Cl | Me | H | Me | 1049677-59-9 |
 |
3-CEC | 3-Cl | Me | H | Et | 2150476-60-9 |
 |
3-BMC | 3-Br | Me | H | Me | 676487-42-6 |
 |
3-IMC | 3-I | Me | H | Me | |
 |
3-TFMAP | 3-CF3 | Me | H | Me | |
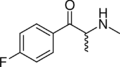 |
Flephedrone | 4-F | Me | H | Me | 447-40-5 |
 |
4-FEC | 4-F | Me | H | Et | 1225625-74-0 |
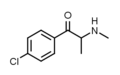 |
Clephedrone | 4-Cl | Me | H | Me | 1225843-86-6 |
 |
2-CEC | 2-Cl | Me | H | Et | |
 |
4-CEC | 4-Cl | Me | H | Et | 14919-85-8 |
 |
2-CiPC | 2-Cl | Me | H | iPr | |
 |
3-CiPC | 3-Cl | Me | H | iPr | |
 |
4-CiPC | 4-Cl | Me | H | iPr | |
| File:4-CBC structure.png | 4-CBC | 4-Cl | Me | H | nBu | 1225621-71-5 |
| File:2Cl-DMC structure.png | 2-CDMC | 2-Cl | Me | Me | Me | |
| File:3Cl-DMC structure.png | 3-CDMC | 3-Cl | Me | Me | Me | |
| File:4-CDMC structure.png | 4-CDMC | 4-Cl | Me | Me | Me | 1157667-29-2 |
 |
Brephedrone | 4-Br | Me | H | Me | 486459-03-4 |
| File:4-BEC structure.png | 4-BEC | 4-Br | Me | H | Et | 135333-26-5 |
| File:4-IMC structure.png | 4-IMC | 4-I | Me | H | Me | |
| File:4-TFMMC structure.png | 4-TFMAP | 4-CF3 | Me | H | Me | |
| File:4-EFMC structure.png | 4-EFMC | 4-(2-fluoroethyl) | Me | H | Me | |
| File:4-MTMC structure.png | 4-MTMC | 4-SCH3 | Me | H | Me | |
| File:4-MSMC structure.png | 4-MSMC | 4-SO2CH3 | Me | H | Me | |
| File:4-PHMC structure.png | 4-PHMC | 4-phenyl | Me | H | Me | |
 |
Mexedrone | 4-Me | methoxymethyl | H | Me | |
| File:3,4-FMMC structure.png | FMMC | 3-F-4-Me | Me | H | Me | 1696642-00-8 |
| File:3,4-MFMC structure.png | MFMC | 3-Me-4-F | Me | H | Me | 1368943-21-8 |
| File:4-Cl-3-MMC structure.png | 4-Cl-3-MMC | 3-Me-4-Cl | Me | H | Me | |
| File:3,4-MMOMC structure.png | MMOMC | 3-Me-4-MeO | Me | H | Me | |
| File:3,4-DCMC structure.png | 3,4-DCMC | 3,4-dichloro | Me | H | Me | 802281-39-6 |
| File:3,4-DCEC structure.png | 3,4-DCEC | 3,4-dichloro | Me | H | Et | 1225618-63-2 |
| File:3,5-DCMC structure.png | 3,5-DCMC | 3,5-dichloro | Me | H | Me | |
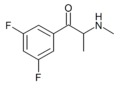 |
3,5-DFMC | 3,5-difluoro | Me | H | Me | 1430343-55-7 |
| File:2,5-DMOMC structure.png | 2,5-DMOMC | 2,5-dimethoxy | Me | H | Me | |
| File:Bk2CC structure.png | βk-2C-C | 2,5-dimethoxy-4-chloro | H | H | H | 1538191-15-9 |
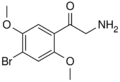 |
βk-2C-B | 2,5-dimethoxy-4-bromo | H | H | H | 807631-09-0 |
| File:Bk2CI structure.png | βk-2C-I | 2,5-dimethoxy-4-iodo | H | H | H | |
| File:Bk2CD structure.png | βk-2C-D | 2,5-dimethoxy-4-methyl | H | H | H | 1368627-25-1 |
| File:Bk2CE structure.png | βk-2C-E | 2,5-dimethoxy-4-ethyl | H | H | H | 1517021-02-1 |
| File:Bk2CP structure.png | βk-2C-P | 2,5-dimethoxy-4-propyl | H | H | H | |
| File:Bk2CiP structure.png | βk-2C-iP | 2,5-dimethoxy-4-isopropyl | H | H | H | 1511033-62-7 |
| File:BkDOB structure.png | βk-DOB | 2,5-dimethoxy-4-bromo | Me | H | H | |
| File:BkMDOM structure.png | βk-MDOM | 2,5-dimethoxy-4-methyl | Me | H | Me | |
| Error creating thumbnail: Unable to save thumbnail to destination | βk-MDA | 3,4-methylenedioxy | Me | H | H | 80535-73-5 |
| File:BkMDAc structure.png | N-Acetyl-βk-MDA | 3,4-methylenedioxy | Me | H | acetyl | |
| File:2,3-MDMC structure.png | 2,3-MDMC | 2,3-methylenedioxy | Me | H | Me | 1427205-87-5 |
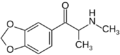 |
Methylone | 3,4-methylenedioxy | Me | H | Me | 186028-79-5 |
 |
Dimethylone | 3,4-methylenedioxy | Me | Me | Me | 109367-07-9 |
| File:NAc-Methylone structure.png | N-Acetylmethylone | 3,4-methylenedioxy | Me | acetyl | Me | |
| File:NOH-Methylone structure.png | N-Hydroxymethylone | 3,4-methylenedioxy | Me | hydroxy | Me | |
| Error creating thumbnail: Unable to save thumbnail to destination | Ethylone | 3,4-methylenedioxy | Me | H | Et | 1112937-64-0 |
 |
Diethylone | 3,4-methylenedioxy | Me | Et | Et | |
| File:NAc-Ethylone structure.png | N-Acetylethylone | 3,4-methylenedioxy | Me | acetyl | Et | |
| File:BkMDiP structure.png | N-Isopropyl-βk-MDA | 3,4-methylenedioxy | Me | H | iPr | |
| File:BkMDtB structure.png | MDPT | 3,4-methylenedioxy | Me | H | t-Bu | 186028-84-2 |
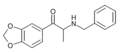 |
Benzylone (BMDP) | 3,4-methylenedioxy | Me | H | Bn | 1823274-68-5 |
| File:N-Cyclohexylmethylone structure.png | N-Cyclohexylmethylone | 3,4-methylenedioxy | Me | H | cyclohexyl | |
| File:3,4-EDMC structure.png | 3,4-EDMC | 3,4-ethylenedioxy | Me | H | Me | 30253-44-2 |
| File:BkIMP structure.png | βk-IMP | 3,4-trimethylene | Me | H | Me | 100608-69-3 |
| File:BkIEB structure.png | βk-IBP | 3,4-trimethylene | Et | H | Et | |
| File:BkIEV structure.png | βk-IVP | 3,4-trimethylene | nPr | H | Et | |
| File:3-fluorobuphedrone structure.png | 3-Fluorobuphedrone | 3-F | Et | H | Me | |
| File:4-fluorobuphedrone structure.png | 4-Fluorobuphedrone | 4-F | Et | H | Me | 1368599-12-5 |
| File:4-bromobuphedrone structure.png | 4-Bromobuphedrone | 4-Br | Et | H | Me | |
| File:3-Methylbuphedrone structure.png | 3-Methylbuphedrone | 3-Me | Et | H | Me | 1797911-07-9 |
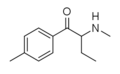 |
4-Me-MABP | 4-Me | Et | H | Me | 1336911-98-8 |
| File:4-Me-NEB structure.png | 4-Me-NEB | 4-Me | Et | H | Et | 18268-19-4 |
| File:2F-NEB structure.png | 2-F-NEB | 2-F | Et | H | Et | |
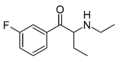 |
3F-NEB | 3-F | Et | H | Et | |
| File:4-F-NEB structure.png | 4-F-NEB | 4-F | Et | H | Et | |
| File:4-Me-DMB structure.png | 4-Me-DMB | 4-Me | Et | Me | Me | |
| File:3,4-DMEB structure.png | 3,4-DMEB | 3,4-dimethyl | Et | H | Et | |
| File:4-methoxybuphedrone structure.png | 4-Methoxybuphedrone | 4-MeO | Et | H | Me | |
 |
Butylone | 3,4-methylenedioxy | Et | H | Me | 802575-11-7 |
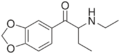 |
Eutylone | 3,4-methylenedioxy | Et | H | Et | 802855-66-9 |
| File:BkPBDB structure.png | βk-PBDB | 3,4-methylenedioxy | Et | H | nPr | |
| File:Bn-4-Me-MABP structure.png | Bn-4-MeMABP | 4-Me | Et | H | Bn | 1445751-39-2 |
| File:BMDB structure.png | BMDB | 3,4-methylenedioxy | Et | H | Bn | 1445751-47-2 |
| File:N-Cyclohexylbutylone structure.png | N-Cyclohexylbutylone | 3,4-methylenedioxy | Et | H | cyclohexyl | |
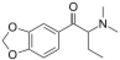 |
βk-DMBDB | 3,4-methylenedioxy | Et | Me | Me | 802286-83-5 |
 |
βk-MMDMA | 3,4-methylenedioxy-5-MeO | Me | H | Me | 2230716-98-8 |
| File:2-methoxymethylone structure.png | βk-MMDMA-2 | 2-MeO-3,4-methylenedioxy | Me | H | Me | |
| File:BkDMMDA structure.png | βk-DMMDA | 2,5-diMeO-3,4-methylenedioxy | Me | H | H | |
| File:5-methylmethylone structure.png | 5-Methylmethylone | 3,4-methylenedioxy-5-Me | Me | H | Me | 1364933-83-4 |
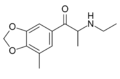 |
5-Methylethylone | 3,4-methylenedioxy-5-Me | Me | H | Et | 1364933-82-3 |
| File:2-methylbutylone structure.png | 2-Methylbutylone | 2-Me-3,4-methylenedioxy | Et | H | Me | 1364933-86-7 |
| File:5-methylbutylone structure.png | 5-Methylbutylone | 3,4-methylenedioxy-5-Me | Et | H | Me | 1354631-29-0 |
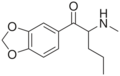 |
Pentylone | 3,4-methylenedioxy | nPr | H | Me | 698963-77-8 |
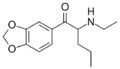 |
N-Ethylpentylone | 3,4-methylenedioxy | nPr | H | Et | 727641-67-0 |
| File:N-propylpentylone structure.png | N-propylpentylone | 3,4-methylenedioxy | nPr | H | nPr | |
| File:N-butylpentylone structure.png | N-butylpentylone | 3,4-methylenedioxy | nPr | H | nBu | |
| File:2,3-Dipentylone structure.png | 2,3-Dipentylone | 2,3-methylenedioxy | nPr | Me | Me | |
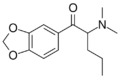 |
Dipentylone | 3,4-methylenedioxy | nPr | Me | Me | 17763-13-2 |
| File:NN-diethyl-pentylone structure.png | N,N-Diethylnorpentylone | 3,4-methylenedioxy | nPr | Et | Et | |
| File:Hexylone structure.png | Hexylone | 3,4-methylenedioxy | nBu | H | Me | |
 |
Isohexylone | 3,4-methylenedioxy | iBu | H | Me | 1157947-89-1 |
| File:Isoheptylone structure.png | Isoheptylone | 3,4-methylenedioxy | iPe | H | Me | |
 |
N-Ethylhexylone | 3,4-methylenedioxy | nBu | H | Et | 27912-41-0 |
 |
N-Ethylheptylone | 3,4-methylenedioxy | nPe | H | Et | |
 |
4-MEAP | 4-Me | nPr | H | Et | 746540-82-9 |
| File:3,4-DMEP structure.png | 3,4-DMEP | 3,4-dimethyl | nPr | H | Et | |
| File:2F-Pentedrone structure.png | 2-F-Pentedrone | 2-F | nPr | H | Me | |
| File:3F-Pentedrone structure.png | 3-F-Pentedrone | 3-F | nPr | H | Me | |
| File:4-fluoropentedrone structure.png | 4-F-Pentedrone | 4-F | nPr | H | Me | |
| File:4-chloropentedrone structure.png | 4-Cl-Pentedrone | 4-Cl | nPr | H | Me | 2167949-43-9 |
 |
4-Methylpentedrone | 4-Me | nPr | H | Me | 1373918-61-6 |
| File:DL-4662 structure.png | DL-4662 | 3,4-dimethoxy | nPr | H | Et | 1674389-55-9 |
| File:4F-NiP-pentedrone structure.png | 4-F-iPr-norpentedrone | 4-F | nPr | H | iPr | |
| File:4Cl-NtB-pentedrone structure.png | 3-CBV | 3-Cl | nPr | H | tBu | |
| File:4-methylhexedrone structure.png | 4-methylhexedrone | 4-Me | nBu | H | Me | |
| File:4-methyl-N-ethylhexedrone structure.png | MEH | 4-Me | nBu | H | Et | |
| Error creating thumbnail: Unable to save thumbnail to destination | 3F-NEH | 3-F | nBu | H | Et | |
| File:4-fluorohexedrone structure.png | 4-F-hexedrone | 4-F | nBu | H | Me | |
| File:4-fluorooctedrone structure.png | 4-F-octedrone | 4-F | hexyl | H | Me | |
| File:Alpha-phenylmephedrone structure.png | α-phenylmephedrone | 4-Me | phenyl | H | Me | |
 |
βk-Ephenidine | H | phenyl | H | Et | 22312-16-9 |
 |
BMAPN | β-naphthyl instead of phenyl | Me | H | Me | |
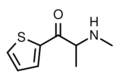 |
βk-Methiopropamine | thiophen-2-yl instead of phenyl | Me | H | Me | 24065-17-6 |
| Error creating thumbnail: Unable to save thumbnail to destination | 5-Cl-bk-MPA | 5-chlorothiophen-2-yl instead of phenyl | Me | H | Me | |
| File:Bk-5-MAPB structure.png | βk-5-MAPB | benzofuran-5-yl instead of phenyl | Me | H | Me | |
| File:Bk-6-MAPB structure.png | βk-6-MAPB | benzofuran-6-yl instead of phenyl | Me | H | Me | |
| File:Bk-5-IT structure.png | βk-5-IT | indol-5-yl instead of phenyl | Me | H | H | 1369231-36-6 |
| File:BK-5F-NM-AMT structure.png | βk-5F-NM-AMT[29] | 5-fluoroindol-3-yl instead of phenyl | Me | H | Me | |
| Error creating thumbnail: Unable to save thumbnail to destination | α-Phthalimidopropiophenone | H | Me | phthalimido | 19437-20-8 | |
| File:PPPO structure.png | PPPO | H | Me | piperidinyl | ||
| File:PPBO structure.png | PPBO | H | Et | piperidinyl | 92728-82-0 | |
| File:FPPVO structure.png | FPPVO | 4-F | nPr | piperidinyl | ||
| File:MDPV-azepane structure.png | MDPV-azepane | 3,4-methylenedioxy | nPr | azepane | ||
| File:Caccure907 structure.png | Caccure 907 | 4-SCH3 | α,α-di-Me | morpholinyl | ||
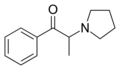 |
α-PPP | H | Me | pyrrolidinyl | 19134-50-0 | |
 |
α-PBP | H | Et | pyrrolidinyl | 13415-54-8 | |
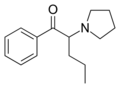 |
α-PVP (O-2387) | H | nPr | pyrrolidinyl | 14530-33-7 | |
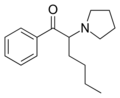 |
α-PHP | H | nBu | pyrrolidinyl | 13415-86-6 | |
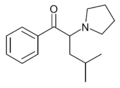 |
α-PHiP | H | iBu | pyrrolidinyl | ||
 |
α-PEP (α-PHPP) | H | nPe | pyrrolidinyl | 13415-83-3 | |
| File:Alpha-POP structure.png | α-POP | H | hexyl | pyrrolidinyl | ||
| File:Alpha-PNP structure.png | α-PNP | H | heptyl | pyrrolidinyl | ||
| File:Diphenylpyrrolidinylethanone structure.png | DPPE (A-D2PV) | H | phenyl | pyrrolidinyl | 27590-61-0 | |
| File:Alpha-PcPeP structure.png | α-PcPeP | H | cyclopentyl | pyrrolidinyl | ||
 |
α-PCYP | H | cyclohexyl | pyrrolidinyl | 1803168-11-7 | |
| File:2-Me-PPP structure.png | 2-MePPP | 2-Me | Me | pyrrolidinyl | 2092429-83-7 | |
| File:3-Me-PPP structure.png | 3-MePPP | 3-Me | Me | pyrrolidinyl | 1214940-01-8 | |
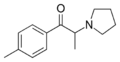 |
4-MePPP | 4-Me | Me | pyrrolidinyl | 1313393-58-6 | |
| File:3MeO-PPP structure.png | 3-MeO-PPP | 3-MeO | Me | pyrrolidinyl | ||
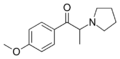 |
MOPPP | 4-MeO | Me | pyrrolidinyl | 478243-09-3 | |
| File:3-F-PPP structure.png | 3-F-PPP | 3-F | Me | pyrrolidinyl | 1214939-99-7 | |
| File:4-F-PPP structure.png | FPPP | 4-F | Me | pyrrolidinyl | 28117-76-2 | |
| File:4-Cl-PPP structure.png | Cl-PPP | 4-Cl | Me | pyrrolidinyl | 93307-24-5 | |
| File:3-Br-PPP structure.png | 3-Br-PPP | 3-Br | Me | pyrrolidinyl | ||
| File:4Br-PPP structure.png | Br-PPP | 4-Br | Me | pyrrolidinyl | ||
| File:2,3-DMPPP structure.png | 2,3-DMPPP | 2,3-dimethyl | Me | pyrrolidinyl | ||
| File:2,4-DMPPP structure.png | 2,4-DMPPP | 2,4-dimethyl | Me | pyrrolidinyl | ||
| File:3,4-DMPPP structure.png | 3,4-DMPPP | 3,4-dimethyl | Me | pyrrolidinyl | ||
| File:3-Me-PBP structure.png | 3-MPBP | 3-Me | Et | pyrrolidinyl | 1373918-60-5 | |
| File:3-F-PBP structure.png | 3-F-PBP | 3-F | Et | pyrrolidinyl | 1373918-59-2 | |
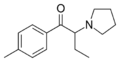 |
MPBP | 4-Me | Et | pyrrolidinyl | 732180-91-5 | |
| File:4-F-PBP structure.png | FPBP | 4-F | Et | pyrrolidinyl | 1373918-67-2 | |
| File:4-Et-PBP structure.png | EPBP | 4-Et | Et | pyrrolidinyl | ||
| File:4-MeO-PBP structure.png | MOPBP | 4-MeO | Et | pyrrolidinyl | ||
| File:MMOPBP structure.png | MMOPBP | 3-Me-4-MeO | Et | pyrrolidinyl | ||
| File:O-2384 structure.png | O-2384 | 3,4-dichloro | Et | pyrrolidinyl | 850352-65-7 | |
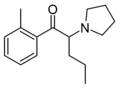 |
2-Me-PVP | 2-Me | nPr | pyrrolidinyl | ||
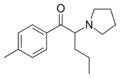 |
Pyrovalerone (O-2371) | 4-Me | nPr | pyrrolidinyl | 3563-49-3 | |
 |
4-Et-PVP | 4-Et | nPr | pyrrolidinyl | ||
| File:3-F-PVP structure.png | 3F-PVP | 3-F | nPr | pyrrolidinyl | ||
 |
FPVP | 4-F | nPr | pyrrolidinyl | 850352-31-7 | |
| File:2Cl-PVP structure.png | 2-Cl-PVP | 2-Cl | nPr | pyrrolidinyl | ||
| File:3Cl-PVP structure.png | 3-Cl-PVP | 3-Cl | nPr | pyrrolidinyl | ||
| File:4-Cl-PVP structure.png | 4-Cl-PVP | 4-Cl | nPr | pyrrolidinyl | 5537-17-7 | |
| File:3-Br-PVP structure.png | 3-Br-PVP | 3-Br | nPr | pyrrolidinyl | ||
| File:4-Br-PVP structure.png | 4-Br-PVP | 4-Br | nPr | pyrrolidinyl | ||
 |
MOPVP | 4-MeO | nPr | pyrrolidinyl | 5537-19-9 | |
 |
DMOPVP | 3,4-dimethoxy | nPr | pyrrolidinyl | 850442-84-1 | |
| File:3,4-DMPVP structure.png | DMPVP | 3,4-dimethyl | nPr | pyrrolidinyl | ||
 |
O-2390 | 3,4-dichloro | nPr | pyrrolidinyl | 850352-61-3 | |
 |
MFPVP | 3-methyl-4-fluoro | nPr | pyrrolidinyl | ||
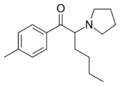 |
MPHP | 4-Me | nBu | pyrrolidinyl | 34138-58-4 | |
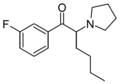 |
3F-PHP | 3-F | nBu | pyrrolidinyl | ||
 |
4F-PHP | 4-F | nBu | pyrrolidinyl | 2230706-09-7 | |
 |
4-Cl-PHP | 4-Cl | nBu | pyrrolidinyl | 2748592-29-0 | |
| File:DMOPHP structure.png | DMOPHP | 3,4-dimethoxy | nBu | pyrrolidinyl | ||
| File:MFPHP structure.png | MFPHP | 3-Me-4-F | nBu | pyrrolidinyl | ||
| Error creating thumbnail: Unable to save thumbnail to destination | 3F-PiHP | 3-F | iBu | pyrrolidinyl | ||
| File:4-F-PiHP structure.png | 4F-PiHP | 4-F | iBu | pyrrolidinyl | ||
| File:O-2494 structure.png | O-2394 | 4-Me | iBu | pyrrolidinyl | 850352-51-1 | |
| File:4-Me-PEP structure.png | MPEP | 4-Me | pentyl | pyrrolidinyl | ||
| File:4-F-PEP structure.png | 4F-PV8 | 4-F | pentyl | pyrrolidinyl | ||
| File:4-MeO-PEP structure.png | 4-MeO-PV8 | 4-MeO | pentyl | pyrrolidinyl | ||
| File:MFPEP structure.png | MFPEP | 3-Me-4-F | pentyl | pyrrolidinyl | ||
| File:MCPEP structure.png | MCPEP | 3-Me-4-Cl | pentyl | pyrrolidinyl | ||
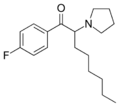 |
4F-PV9 | 4-F | hexyl | pyrrolidinyl | ||
| File:4-MeO-POP structure.png | 4-MeO-PV9 | 4-MeO | hexyl | pyrrolidinyl | ||
| File:Alpha-phenylpyrovalerone structure.png | α-Phenylpyrovalerone | 4-Me | phenyl | pyrrolidinyl | ||
 |
MDPPP | 3,4-methylenedioxy | Me | pyrrolidinyl | 783241-66-7 | |
| File:MDMPP structure.png | MDMPP | 3,4-methylenedioxy | α,α-di-Me | pyrrolidinyl | ||
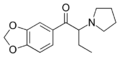 |
MDPBP | 3,4-methylenedioxy | Et | pyrrolidinyl | 784985-33-7 | |
 |
MDPV | 3,4-methylenedioxy | nPr | pyrrolidinyl | 687603-66-3 | |
| File:2,3-MDPV structure.png | 2,3-MDPV | 2,3-methylenedioxy | nPr | pyrrolidinyl | ||
| File:5-Me-MDPV structure.png | 5-Me-MDPV | 3,4-methylenedioxy-5-Me | nPr | pyrrolidinyl | ||
| File:6-Me-MDPV structure.png | 6-Me-MDPV | 2-Me-4,5-methylenedioxy | nPr | pyrrolidinyl | ||
| File:6-MeO-MDPV structure.png | 6-MeO-MDPV | 2-MeO-4,5-methylenedioxy | nPr | pyrrolidinyl | ||
| File:4-MeO-5-Br-2,3-MDPV structure.png | Br-MeO-MDPV | 2,3-methylenedioxy-4-MeO-5-Br | nPr | pyrrolidinyl | ||
| File:MDPiVP structure.png | MDPiVP | 3,4-methylenedioxy | iPr | pyrrolidinyl | ||
 |
MDPHP | 3,4-methylenedioxy | nBu | pyrrolidinyl | 776994-64-0 | |
| File:MDPHiP structure.png | MDPHiP | 3,4-methylenedioxy | iBu | pyrrolidinyl | ||
| File:MDPEP structure.png | MDPEP (MD-PV8) | 3,4-methylenedioxy | pentyl | pyrrolidinyl | 24646-39-7 | |
| File:MDPOP structure.png | MDPOP (MD-PV9) | 3,4-methylenedioxy | hexyl | pyrrolidinyl | 24646-40-0 | |
| File:5-PPDI structure.png | 5-PPDI | 3,4-trimethylene | Et | pyrrolidinyl | ||
| File:5-BPDI structure.png | 5-BPDI | 3,4-trimethylene | nPr | pyrrolidinyl | ||
| File:5-HPDI structure.png | 5-HPDI | 3,4-trimethylene | nBu | pyrrolidinyl | ||
 |
IPPV | 3,4-trimethylene | phenyl | pyrrolidinyl | ||
| File:TH-PBP structure.png | TH-PBP | 3,4-tetramethylene | Et | pyrrolidinyl | ||
 |
TH-PVP | 3,4-tetramethylene | nPr | pyrrolidinyl | 2304915-07-7 | |
| File:TH-PHP structure.png | TH-PHP | 3,4-tetramethylene | nBu | pyrrolidinyl | ||
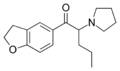 |
5-DBFPV | 2,3-dihydrobenzofuran-5-yl instead of Ph | nPr | pyrrolidinyl | 1620807-94-4 | |
| File:3-BF-PVP structure.png | 3-BF-PVP | benzofuran-3-yl instead of Ph | nPr | pyrrolidinyl | ||
 |
Naphyrone (O-2482) | β-naphthyl instead of phenyl | nPr | pyrrolidinyl | 850352-53-3 | |
| File:Alpha-naphyrone structure.png | α-Naphyrone | α-naphthyl instead of phenyl | nPr | pyrrolidinyl | ||
| File:Alpha-PPT structure.png | α-PPT | thiophen-2-yl instead of phenyl | Me | pyrrolidinyl | ||
| File:Alpha-PBT structure.png | α-PBT | thiophen-2-yl instead of phenyl | Et | pyrrolidinyl | ||
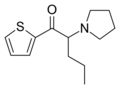 |
α-PVT | thiophen-2-yl instead of phenyl | nPr | pyrrolidinyl | 1400742-66-6 | |
Legality
On 2 April 2010, the Advisory Council on the Misuse of Drugs in the United Kingdom announced that a broad structure-based ban of this entire class of compounds would be instituted, following extensive publicity around grey-market sales and recreational use of mephedrone, a common member of the family. This ban covers compounds with the aforementioned general structure, with 28 compounds specifically named.[30]
"Any compound (not being bupropion or a substance for the time being specified in paragraph 2.2) structurally derived from 2-amino-1-phenyl-1-propanone by modification in any of the following ways, that is to say,(i) by substitution in the phenyl ring to any extent with alkyl, alkoxy, alkylenedioxy, haloalkyl or halide substituents, whether or not further substituted in the phenyl ring by one or more other univalent substituents;
(ii) by substitution at the 3-position with an alkyl substituent;
(iii) by substitution at the nitrogen atom with alkyl or dialkyl groups, or by inclusion of the nitrogen atom in a cyclic structure."—ACMD, 2 April 2010
This text was added as an amendment to the Misuse of Drugs Act 1971, to come into force on 16 April 2010.[31] Note that four of the above compounds (cathinone, methcathinone, diethylpropion and pyrovalerone) were already illegal in the UK at the time the ACMD report was issued. Two compounds were specifically excluded from the ban, these being bupropion because of its common use in medicine and relative lack of abuse potential, and naphyrone because its structure falls outside the generic definition and not enough evidence was yet available to justify a ban.
Naphyrone analogues were subsequently banned in July 2010 following a further review by the ACMD,[32][33] along with a further broad based structure ban even more expansive than the last.[34][35]
"Any compound structurally derived from 2–aminopropan–1–one by substitution at the 1-position with any monocyclic, or fused-polycyclic ring system (not being a phenyl ring or alkylenedioxyphenyl ring system), whether or not the compound isfurther modified in any of the following ways, that is to say—
(i) by substitution in the ring system to any extent with alkyl, alkoxy, haloalkyl or halide substituents, whether or not further substituted in the ring system by one or more other univalent substituents;
(ii) by substitution at the 3–position with an alkyl substituent;
(iii) by substitution at the 2-amino nitrogen atom with alkyl or dialkyl groups, or
by inclusion of the 2-amino nitrogen atom in a cyclic structure".—Home Office, 13 July 2010.
The substitutions in the general structure for naphyrone analogues subject to the ban may be described as follows:
- Cyc = any monocyclic, or fused-polycyclic ring system (not being a phenyl ring or alkylenedioxyphenyl ring system), including analogues where the ring system is substituted to any extent with alkyl, alkoxy, haloalkyl or halide substituents, whether or not further substituted in the ring system by one or more other univalent substituents
- R1 = hydrogen or any alkyl group
- R2 = hydrogen, any alkyl group, or incorporation in a cyclic structure
- R3 = hydrogen, any alkyl group, or incorporation in a cyclic structure
More new derivatives have however continued to appear, with the UK reporting more novel cathinone derivatives detected in 2010 than any other country in Europe, with most of them first identified after the generic ban had gone into effect and thus already being illegal despite never having been previously reported.[36]
In the United States, substituted cathinones are the psychoactive ingredients in "bath salts" which as of July 2011 were banned by at least 28 states, but not by the federal government.[37]
See also
- Markush structure
- Substituted amphetamines
- Substituted methylenedioxyphenethylamines
- Substituted phenethylamines
- Substituted phenylmorpholines
- Structural scheduling of synthetic cannabinoids
- Arylcyclohexylamines
- List of aminorex analogues
- List of fentanyl analogues
- List of methylphenidate analogues
References
- ↑ "1-(4-Methylphenyl)-2-pyrrolidin-1-yl-pentan-1-one (Pyrovalerone) analogues: a promising class of monoamine uptake inhibitors". Journal of Medicinal Chemistry 49 (4): 1420–32. February 2006. doi:10.1021/jm050797a. PMID 16480278.
- ↑ "Emerging drugs of abuse: current perspectives on substituted cathinones". Substance Abuse and Rehabilitation 5: 37–52. 2014. doi:10.2147/SAR.S37257. PMID 24966713.
- ↑ "DARK Classics in Chemical Neuroscience: Cathinone-Derived Psychostimulants". ACS Chemical Neuroscience 9 (10): 2379–2394. October 2018. doi:10.1021/acschemneuro.8b00147. PMID 29714473.
- ↑ "Intoxications in the STRIDA project involving a panorama of psychostimulant pyrovalerone derivatives, MDPV copycats". Clinical Toxicology 56 (4): 256–263. April 2018. doi:10.1080/15563650.2017.1370097. PMID 28895757.
- ↑ "The newest cathinone derivatives as designer drugs: an analytical and toxicological review". Forensic Toxicology 36 (1): 33–50. 2018. doi:10.1007/s11419-017-0385-6. PMID 29367861.
- ↑ "Khat use is associated with impaired working memory and cognitive flexibility". PLOS ONE 6 (6): e20602. 2011. doi:10.1371/journal.pone.0020602. PMID 21698275. Bibcode: 2011PLoSO...620602C.
- ↑ Europol 2008 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2009 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2010 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2011 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2012 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2013 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2014 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2015 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2016 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ Europol 2017 Annual Report on the implementation of Council Decision 2005/387/JHA
- ↑ European Monitoring Center for Drugs and Drug Addiction (December 2020). New psychoactive substances: global markets, glocal threats and the COVID-19 pandemic. An update from the EU Early Warning System. Luxembourg: Publications Office of the European Union. doi:10.2810/921262. ISBN 9789294975584. https://www.emcdda.europa.eu/system/files/publications/13464/20205648_TD0320796ENN_PDF_rev.pdf.
- ↑ "Chemistry, pharmacology, toxicology, and hepatic metabolism of designer drugs of the amphetamine (ecstasy), piperazine, and pyrrolidinophenone types: a synopsis". Therapeutic Drug Monitoring 26 (2): 127–31. April 2004. doi:10.1097/00007691-200404000-00007. PMID 15228152.
- ↑ "The characterisation of two halogenated cathinone analogues: 3,5-difluoromethcathinone and 3,5-dichloromethcathinone". Forensic Science International 217 (1–3): 139–45. April 2012. doi:10.1016/j.forsciint.2011.10.042. PMID 22088945.
- ↑ "Identification and analytical characterization of nine synthetic cathinone derivatives N-ethylhexedrone, 4-Cl-pentedrone, 4-Cl-α-EAPP, propylone, N-ethylnorpentylone, 6-MeO-bk-MDMA, α-PiHP, 4-Cl-α-PHP, and 4-F-α-PHP". Drug Testing and Analysis 9 (8): 1162–1171. August 2017. doi:10.1002/dta.2136. PMID 27863142.
- ↑ "Identification and structural characterization of synthetic cathinones: N-propylcathinone, 2,4-dimethylmethcathinone, 2,4-dimethylethcathinone, 2,4-dimethyl-α-pyrrolidinopropiophenone, 4-bromo-α-pyrrolidinopropiophenone, 1-(2,3-dihydro-1H-inden-5-yl)-2-(pyrrolidin-1-yl)hexan-1-one and 2,4-dimethylisocathinone.". Forensic Toxicol 37 (2): 288–307. 2019. doi:10.1007/s11419-018-00463-w.
- ↑ "Analytische Daten neuer 2-aminosubstituierter Methylendioxyvalerophenonderivate.". Toxichem Krimtech 83 (1): 3–29. January 2016.
- ↑ "The newest cathinone derivatives as designer drugs: an analytical and toxicological review". Forensic Toxicology 36 (1): 33–50. 2018. doi:10.1007/s11419-017-0385-6. PMID 29367861.
- ↑ "Prediction of designer drugs: Synthesis and spectroscopic analysis of synthetic cathinone analogs that may appear on the Swedish drug market". Drug Testing and Analysis 10 (7): 1076–1098. February 2018. doi:10.1002/dta.2366. PMID 29426062.
- ↑ "Forensic drug analysis of chloro-N,N-dimethylcathinone (CDC) and chloroethcathinone (CEC): Identification of 4-CDC and 4-CEC in drug seizures and differentiation from their ring-substituted positional isomers". Forensic Science International 298: 268–277. May 2019. doi:10.1016/j.forsciint.2019.03.002. PMID 30925345.
- ↑ "New psychoactive versus conventional stimulants - a ten-year review of casework in Hungary". Legal Medicine 47: 101780. November 2020. doi:10.1016/j.legalmed.2020.101780. PMID 32882537.
- ↑ "Interpol review of controlled substances 2016-2019". Forensic Science International. Synergy 2: 608–669. 2020. doi:10.1016/j.fsisyn.2020.01.019. PMID 33385148.
- ↑ "Valtioneuvoston asetus kuluttajamarkkinoilta kielletyistä psykoaktiivisista aineista" (in Finnish). Finlex Data Bank. https://finlex.fi/fi/laki/ajantasa/2014/20141130#a12.11.2020-764.
- ↑ Baggott M. Advantageous Tryptamine Compositions For Mental Disorders or Enhancement. Patent WO 2022/061242
- ↑ Advisory Council on the Misuse of Drugs (UK). Consideration of the cathinones. 31 March 2010. Retrieved 2011-07-17.
- ↑ "The Misuse of Drugs (Amendment) (England, Wales and Scotland) Regulations 2010 No. 1144". Opsi.gov.uk. http://www.opsi.gov.uk/si/si2010/uksi_20101144_en_1.
- ↑ "NRG-1 'legal high' drug is banned". BBC News. 2010-07-12. http://news.bbc.co.uk/1/hi/uk/10602398.stm.
- ↑ "Advisory Council on the Misuse of Drugs Naphyrone Report (2010)". Home Office. 2010-07-07. http://www.homeoffice.gov.uk/publications/drugs/acmd1/naphyrone-report.
- ↑ "Explanatory Memorandum To The Misuse of Drugs (Amendment No. 2) (England, Wales and Scotland) Regulations 2010 No. 1799". Opsi.gov.uk. http://www.opsi.gov.uk/si/si2010/em/uksiem_20101799_en.pdf.
- ↑ "The Misuse of Drugs (Amendment No. 2) (England, Wales and Scotland) Regulations 2010 No. 1799". Opsi.gov.uk. http://www.opsi.gov.uk/si/si2010/pdf/uksi_20101799_en.pdf.
- ↑ European Monitoring Centre on Drugs and Drug Addiction. EMCDDA–Europol 2010 Annual Report on the implementation of Council Decision 2005/387/JHA. Retrieved 2011-07-17.
- ↑ "An Alarming New Stimulant, Legal in Many States.". The New York Times. 16 July 2011. https://www.nytimes.com/2011/07/17/us/17salts.html.
 |
