Chemistry:Bis(pyridine)iodonium(I) tetrafluoroborate
From HandWiki
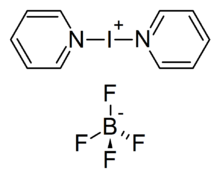
| |
| |
| Names | |
|---|---|
| IUPAC name
bis(pyridin-1-ium-1-yl)iodanuide tetrafluoroborate
| |
| Other names
Barluenga's Reagent
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C10H10BF4IN2 | |
| Molar mass | 371.91 g·mol−1 |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Bis(pyridine)iodonium(I) tetrafluoroborate or Barluenga's reagent, named after José Barluenga,[1] is a mild iodinating reagent. Commercially available, it may be prepared by reacting iodine with pyridine in the presence of silver tetrafluoroborate supported on silica gel.[2]
References
- ↑ Martín, Nazario; Muñiz, Kilian (2010). "Congratulations to Professor José Barluenga on his 70th Birthday". Chemistry: A European Journal 16 (32): 9696–9697. doi:10.1002/chem.201001986.
- ↑ Justin M. Chalker; Amber L. Thompson; Benjamin G. Davis (2010). "Safe and Scalable Preparation of Barluenga's Reagent". Organic Syntheses 87: 288. http://www.orgsyn.org/demo.aspx?prep=v87p0288.
 |

