Chemistry:Cerium phosphide
From HandWiki
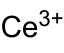 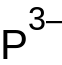
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CeP | |
| Molar mass | 171.090 g·mol−1 |
| Appearance | grey crystals[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Cerium phosphide is an inorganic compound with the chemical formula CeP. It is one of the phosphides of cerium. It can be obtained by reacting cerium dioxide and phosphine at 1300 °C in the presence of hydrogen,[2] or by reacting sodium phosphide and cerium chloride at 700~800 °C.[1] It will be oxidized to monoclinic CePO4 in air above 900 °C.[2] It reacts with iodine in a quartz ampoule to obtain CeSiP3.[3]
References
- ↑ 1.0 1.1 Rowley, Adrian T.; Parkin, Ivan P. (1993). "Convenient synthesis of lanthanide and mixed lanthanide phosphides by solid-state routes involving sodium phosphide". Journal of Materials Chemistry (Royal Society of Chemistry (RSC)) 3 (7): 689. doi:10.1039/jm9930300689. ISSN 0959-9428.
- ↑ 2.0 2.1 Vasil'eva, I. G.; Mironov, K. E.; Mironov, Yu. I. Properties of cerium monophosphideScript error: The function "in_lang" does not exist.. Redkozemel. Metal. Ikh Soedin., Mater. Vses. Simp., 1970: 160-165. Template:Coden.
- ↑ Mironov, K. E.; Abdullin, R. V. Formation and chemical transfer of cerium silicon phosphide (CeSiP3)Script error: The function "in_lang" does not exist.. Zhurnal Neorganicheskoi Khimii, 1980. 25 (8): 2062-2066.
External reading
- Pavel S. Chizhov, Nellie R. Khasanova, Michael Baitinger, Walter Schnelle, Yuri Prots, Ulrich Burkhardt, Evgeny V. Antipov, Yuri Grin (2006-09-01). "Ce 4 (P 1- x Si x ) 3- z : A First Example for the Stabilization of the Anti-Th 3 P 4 Type Structure by Substitution in the Non-Metal Substructure" (in en). Inorganic Chemistry 45 (18): 7210–7216. doi:10.1021/ic060726h. ISSN 0020-1669. PMID 16933921. https://pubs.acs.org/doi/10.1021/ic060726h. Retrieved 2021-12-27.
 |
