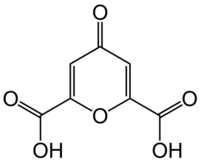
Chemistry:Chelidonic acid
| |
| Names | |
|---|---|
| Preferred IUPAC name
4-Oxo-4H-pyran-2,6-dicarboxylic acid | |
| Other names
Jerva acid; Jervaic acid; Jervasic acid; γ-Pyrone-2,6-dicarboxylic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C7H4O6 | |
| Molar mass | 184.103 g·mol−1 |
| Melting point | 257 °C (495 °F; 530 K)[1] (decomposes) |
| Related compounds | |
Related compounds
|
Meconic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Chelidonic acid is a heterocyclic organic acid with a pyran skeleton.
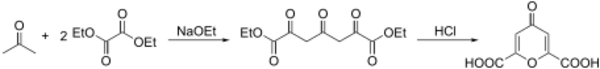
Preparation
Chelidonic acid can be prepared in two steps from diethyl oxalate and acetone:[1][2]
Uses
Chelidonic acid is used to synthesize 4-pyrone via thermal decarboxylation.[3]
Chelidonic acid is one of the most potent inhibitors of glutamate decarboxylase known in vitro, and is thus used in research purpose.[4]
Natural occurrence
Joseph M. A. Probst (1812–1842) discovered the acid in extracts of Chelidonium majus in 1839,[5] and it was first studied by Joseph Udo Lerch (1816–1892) in 1846.[6][7] It occurs naturally in plants of the Asparagales order.[8] Potassium chelidonate has been found to be responsible for nyctinasty in some plants; specifically, it has been found to regulate the closing of leaves of Cassia mimosoides at nightfall.[9][10]
See also
References
- ↑ 1.0 1.1 E. Raymond Riegel and F. Zwilgmeyer (1937). "Chelidonic acid". Organic Syntheses 17: 40. http://www.orgsyn.org/demo.aspx?prep=CV2P0126.; Collective Volume, 2, pp. 126
- ↑ G. Horvath; C. Russa; Z. Koentoes; J. Gerencser (1999). "A new Efficient Method for the Preparation of 2,6-Pyridinedihiethyl Ditosylates from Dimethyl 2,60-Pyridinedicarboxylates". Synth. Commun. 29 (21): 3719–3732. doi:10.1080/00397919908086011.
- ↑ Weygand, Conrad (1972). Hilgetag, G.; Martini, A.. eds. Weygand/Hilgetag Preparative Organic Chemistry (4th ed.). New York: John Wiley & Sons, Inc.. p. 1009. ISBN 0471937495.
- ↑ Porter, Terence G.; Martin, David L. (1985). "Chelidonic acid and other conformationally restricted substrate analogues as inhibitors of rat brain glutamate decarboxylase". Biochemical Pharmacology 34 (23): 4145–4150. doi:10.1016/0006-2952(85)90207-2. https://linkinghub.elsevier.com/retrieve/pii/0006295285902072. Retrieved 2026-01-29.
- ↑ Probst (1839). "Beschreibung und Darstellungsweise einiger bei der Analyse des Chelidonium majus neu aufgefundenen Stoffe" (in de). Annalen der Pharmacie 29 (2): 113–131. doi:10.1002/jlac.18390290202. ISSN 1099-0690. https://books.google.com/books?id=17E9AQAAMAAJ&pg=PA116.
- ↑ Lerch, J. Ud. (1846). "Untersuchung der Chelidonsäure" (in de). Annalen der Chemie und Pharmacie 57 (3): 273–318. doi:10.1002/jlac.18460570302. ISSN 1099-0690. https://books.google.com/books?id=LrQ9AQAAMAAJ&pg=RA1-PA273.
- ↑ Roscoe, H.E.; Schorlemmer, C. (1890). A Treatise on Chemistry, Volume 3, Part 2 (1st ed.). New York: D Appleton and Company. pp. 624. https://books.google.com/books?id=xM4cAQAAIAAJ&pg=PA624.
- ↑ "Asparagales". Angiosperm Phylogeny Group. http://www.mobot.org/MOBOT/research/APweb/orders/asparagalesweb.htm#Asparagales. Retrieved 30 August 2017.
- ↑ Ueda, Minoru; Ohnuki, Takashi; Yamamura, Shosuke (1998). "Leaf-opening substance of a nyctinastic plant, Cassia mimosoides". Phytochemistry 49 (3): 633. doi:10.1016/S0031-9422(98)00134-4.
- ↑ Ueda, Minoru; Yamamura, Shosuke (1998). "Chemical studies on plant movement". Current Organic Chemistry 2 (4): 437–461. https://books.google.com/books?id=aI_3BDcYKXgC&pg=PA437.
 |