Chemistry:4-Pyrone
From HandWiki
| |
| Names | |
|---|---|
| Preferred IUPAC name
4H-Pyran-4-one | |
| Other names
γ-Pyrone
4-Pyranone | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C5H4O2 | |
| Molar mass | 96.08 |
| Melting point | 32 to 34 °C (90 to 93 °F; 305 to 307 K) |
| Boiling point | 210 to 215 °C (410 to 419 °F; 483 to 488 K) |
| Hazards | |
| Flash point | 101 °C (214 °F; 374 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
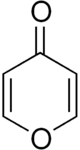
4-Pyrone (γ-pyrone or pyran-4-one) is an unsaturated cyclic chemical compound with the molecular formula C5H4O2.It is isomeric with 2-pyrone.
Preparation
4-Pyrone is prepared via the thermal decarboxylation of chelidonic acid.[2]
Reactions
4-Pyrone and its derivatives react with amines in protic solvents to form 4-Pyridones.[2][3][4]
Derivatives
4-Pyrone forms the central core of several natural chemical compounds,[5] including maltol, meconic acid, kojic acid, and of the important class of the Flavones.
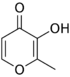
Error creating thumbnail: Unable to save thumbnail to destination Maltol Kojic acid
See also
- Pyrone
- 4-Pyridone
- Dehydroacetic acid
References
- ↑ 4H-Pyran-4-one at Sigma-Aldrich
- ↑ 2.0 2.1 Weygand, Conrad (1972). Hilgetag, G.; Martini, A.. eds. Weygand/Hilgetag Preparative Organic Chemistry (4th ed.). New York: John Wiley & Sons, Inc.. pp. 533–534, & 1009. ISBN 0471937495.
- ↑ Van Allan, J. A.; Reynolds, G. A.; Alessi, J. T.; Chie Chang, S.; C. Joines, R. (1971). "Reactions of 4-pyrones with primary amines. A new class of ionic associates". Journal of Heterocyclic Chemistry 8 (6): 919–922. doi:10.1002/jhet.5570080606.
- ↑ Cook, Denys (1963). "The Preparation, Properties, and Structure of 2,6-bis-(Alkyamino)-2,5-heptadien-4-ones". Canadian Journal of Chemistry 41 (6): 1435–1440. doi:10.1139/v63-195.
- ↑ Wilk, Wolfram; Waldmann, Herbert; Kaiser, Markus (2009). "Γ-Pyrone natural products—A privileged compound class provided by nature". Bioorganic & Medicinal Chemistry 17 (6): 2304–2309. doi:10.1016/j.bmc.2008.11.001. PMID 19042133.
 |
