Chemistry:Copper(II) lactate
From HandWiki

| |
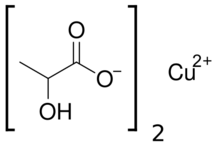
| |
| Names | |
|---|---|
| IUPAC name
copper;2-hydroxypropanoate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Cu(C3H5O3)2 (anhydrous) Cu(C3H5O3)2·2H2O (dihydrate) | |
| Molar mass | 241.69 g/mol (anhydrous) 277.72 g/mol (dihydrate)[1] |
| Appearance | green powder |
| 167 g/L (dihydrate, cold water)[1] 450 g/L (dihydrate, hot water)[1] | |
| Solubility | insoluble in acetone and isopropyl alcohol |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302 | |
| P264, P270, P301+312, P330, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Copper(II) lactate, also referred to as cupric lactate, is the chemical compound with the formula Cu(C3H5O3)2. It is a green powder that readily dissolves in hot water to form a green solution, often more blue than the acetate.
Preparation
Copper(II) lactate can be prepared by heating copper(II) oxide with lactic acid.[citation needed] The reaction does not usually go to completion, and excess reactants must be removed from the product.[citation needed]
Bibliography
- ↑ 1.0 1.1 1.2 Lide, David R., ed (1991). CRC Handbook of Chemistry and Physics (72 ed.).. Boca Raton, FL: CRC Press. ISBN 0849304725.
 |
