Chemistry:Copper(I) iodide

| |
| Names | |
|---|---|
| IUPAC name
Copper(I) iodide
| |
| Other names
Cuprous iodide, marshite
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| CuI | |
| Molar mass | 190.45 g/mol |
| Appearance | White solid |
| Odor | odorless |
| Density | 5.67 g/cm3 [1] |
| Melting point | 606 °C (1,123 °F; 879 K) |
| Boiling point | 1,290 °C (2,350 °F; 1,560 K) (decomposes) |
| 0.000042 g/100 mL | |
Solubility product (Ksp)
|
1.27 x 10−12 [2] |
| Solubility | soluble in ammonia and iodide solutions insoluble in dilute acids |
| Vapor pressure | 10 mm Hg (656 °C) |
| -63.0·10−6 cm3/mol | |
Refractive index (nD)
|
2.346 |
| Structure | |
| zincblende | |
| Tetrahedral anions and cations | |
| Hazards | |
| Safety data sheet | Sigma Aldrich |
| GHS pictograms |   
|
| GHS Signal word | DANGER |
| H302, H315, H319, H335, H410 | |
| P261, P273, P305+351+338, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 1 mg/m3 (as Cu)[3] |
REL (Recommended)
|
TWA 1 mg/m3 (as Cu)[3] |
IDLH (Immediate danger)
|
TWA 100 mg/m3 (as Cu)[3] |
| Related compounds | |
Other anions
|
|
Other cations
|
silver iodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Copper(I) iodide is the inorganic compound with the formula CuI. It is also known as cuprous iodide. It is useful in a variety of applications ranging from organic synthesis to cloud seeding.
Copper(I) iodide is white, but samples often appear tan or even, when found in nature as rare mineral marshite, reddish brown, but such color is due to the presence of impurities. It is common for samples of iodide-containing compounds to become discolored due to the facile aerobic oxidation of the iodide anion to molecular iodine.[4][5][6]
Structure
Copper(I) iodide, like most binary (containing only two elements) metal halides, is an inorganic polymer. It has a rich phase diagram, meaning that it exists in several crystalline forms. It adopts a zinc blende structure below 390 °C (γ-CuI), a wurtzite structure between 390 and 440 °C (β-CuI), and a rock salt structure above 440 °C (α-CuI). The ions are tetrahedrally coordinated when in the zinc blende or the wurtzite structure, with a Cu-I distance of 2.338 Å. Copper(I) bromide and copper(I) chloride also transform from the zinc blende structure to the wurtzite structure at 405 and 435 °C, respectively. Therefore, the longer the copper–halide bond length, the lower the temperature needs to be to change the structure from the zinc blende structure to the wurtzite structure. The interatomic distances in copper(I) bromide and copper(I) chloride are 2.173 and 2.051 Å, respectively.[7] Consistent with its covalency, CuI is a p-type semiconductor.[8]
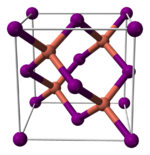 |
150px | 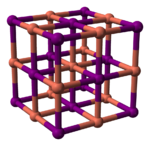
|
| γ-CuI | β-CuI | α-CuI |
Preparation
Copper(I) iodide can be prepared by heating iodine and copper in concentrated hydriodic acid.[9]
In the laboratory however, copper(I) iodide is prepared by simply mixing an aqueous solution of potassium iodide and a soluble copper(II) salt such as copper(II) sulfate.[4]
- Cu2+ + 2I− → CuI + 0.5 I2
Reactions
Copper(I) iodide reacts with mercury vapors to form copper tetraiodomercurate:
- 4 CuI + Hg → Cu2HgI4 + 2 Cu
This reaction can be used for the detection of mercury since the white (CuI) to brown (Cu2HgI4) color change is dramatic.
Copper(I) iodide is used in the synthesis of Cu(I) clusters such as Cu
6I−
7.[10]
Copper(I) iodide dissolves in acetonitrile, yielding diverse complexes. Upon crystallization, molecular[11] or polymeric[12][13] compounds can be isolated. Dissolution is also observed when a solution of the appropriate complexing agent in acetone or chloroform is used. For example, thiourea and its derivatives can be used. Solids that crystallize out of those solutions are composed of hybrid inorganic chains.[14]
Uses
In combination with 1,2- or 1,3-diamine ligands, CuI catalyzes the conversion of aryl, heteroaryl, and vinyl bromides into the corresponding iodides. NaI is the typical iodide source and dioxane is a typical solvent (see aromatic Finkelstein reaction).[15]
CuI is used as a co-catalyst with palladium catalyst in the Sonogashira coupling.[16]
CuI is used in cloud seeding,[17] altering the amount or type of precipitation of a cloud, or their structure by dispersing substances into the atmosphere which increase water's ability to form droplets or crystals. CuI provides a sphere for moisture in the cloud to condense around, causing precipitation to increase and cloud density to decrease.
The structural properties of CuI allow CuI to stabilize heat in nylon in commercial and residential carpet industries, automotive engine accessories, and other markets where durability and weight are a factor.[citation needed]
CuI is used as a source of dietary iodine in table salt and animal feed.[17]
References
- ↑ Lide, David R., ed (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. ISBN 0-8493-0487-3.
- ↑ John Rumble (June 18, 2018) (in English). CRC Handbook of Chemistry and Physics (99th ed.). CRC Press. pp. 4–47. ISBN 978-1138561632.
- ↑ 3.0 3.1 3.2 NIOSH Pocket Guide to Chemical Hazards. "#0150". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0150.html.
- ↑ 4.0 4.1 George B. Kauffman, Lawrence W. Fang (1983). Copper(I) Iodide. Inorganic Syntheses. 22. p. 101. doi:10.1002/9780470132531.ch20.
- ↑ "Verification". https://www.mindat.org/min-2580.html.
- ↑ "List of Minerals". 21 March 2011. https://www.ima-mineralogy.org/Minlist.htm.
- ↑ Structural Inorganic Chemistry (5th ed.). Oxford: Oxford University Press. 1984. pp. 410 and 444.
- ↑ Bidikoudi, Maria; Kymakis, Emmanuel (2019). "Novel approaches and scalability prospects of copper based hole transporting materials for planar perovskite solar cells". Journal of Materials Chemistry C 7 (44): 13680–13708. doi:10.1039/c9tc04009a.
- ↑ Inorganic Chemistry. San Diego: Academic Press. 2001. ISBN 0-12-352651-5.
- ↑ Yu, Muxin; Chen, Lian; Jiang, Feilong; Zhou, Kang; Liu, Caiping; Sun, Cai; Li, Xingjun; Yang, Yan et al. (2017). "Cation-Induced Strategy toward an Hourglass-Shaped Cu6I7– Cluster and its Color-Tunable Luminescence". Chemistry of Materials 29 (19): 8093–8099. doi:10.1021/acs.chemmater.7b01790.
- ↑ "Crystal structure of di-μ-iodido-bis-[bis(aceto-nitrile-κN)copper(I)"]. Acta Crystallographica Section E 71 (Pt 11): m189-90. November 2015. doi:10.1107/S2056989015018149. PMID 26594527.
- ↑ "Lewis-Base Adducts of Group 11 Metal(I) Compounds. XL. Conformational Systematics of [(N-base)1(CuX)1]∞ Orthogonal' Stair' Polymers (N-base = 'One-Dimensional Aceto-nitrile, Benzo-nitrile Ligand)" (in en). Australian Journal of Chemistry 42 (1): 79. 1989. doi:10.1071/CH9890079. ISSN 0004-9425.
- ↑ "Copper(I) complexes with metal-metal (d10–d10) bond. Crystal and molecular structures of adducts of tantalocene trihydride with copper(I) iodide of composition: (η5-C5H5)2TaH[(μ2-H)Cu(μ2-I)2Cu(μ2-H)]2HTa(η5-C5H5)2, (η5-C5H4But)2TaH(μ2-H)2Cu(μ2-I)2Cu(μ2-H)2HTa(η5-C5H4But)2·CH3CN and {Cu(μ3-I)·P[N(CH3)2]3}4" (in en). Inorganica Chimica Acta 169 (1): 109–118. 1990. doi:10.1016/S0020-1693(00)82043-5.
- ↑ "Copper(I) iodide ribbons coordinated with thiourea derivatives". Acta Crystallographica Section C 74 (Pt 12): 1650–1655. December 2018. doi:10.1107/S2053229618015620. PMID 30516149.
- ↑ "Copper-catalyzed halogen exchange in aryl halides: an aromatic Finkelstein reaction". Journal of the American Chemical Society 124 (50): 14844–5. December 2002. doi:10.1021/ja028865v. PMID 12475315.
- ↑ Sonogashira, K. (2002), "Development of Pd-Cu catalyzed cross-coupling of terminal acetylenes with sp2-carbon halides", J. Organomet. Chem. 653 (1–2): 46–49, doi:10.1016/s0022-328x(02)01158-0
- ↑ 17.0 17.1 "Copper compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. June 2000. pp. 1–31. doi:10.1002/14356007.a07_567. ISBN 3527306730.
Further reading
- Dictionary of Inorganic Compounds. 3. London: Chapman and Hall. 1992. pp. 3103.
External links
| HI | He | ||||||||||||||||
| LiI | BeI2 | BI3 | CI4 | NI3 | I2O4, I2O5, I4O9 |
IF, IF3, IF5, IF7 |
Ne | ||||||||||
| NaI | MgI2 | AlI3 | SiI4 | PI3, P2I4 |
S | ICl, ICl3 |
Ar | ||||||||||
| KI | CaI2 | Sc | TiI4 | VI3 | CrI3 | MnI2 | FeI2 | CoI2 | NiI2 | CuI | ZnI2 | Ga2I6 | GeI2, GeI4 |
AsI3 | Se | IBr | Kr |
| RbI | SrI2 | YI3 | ZrI4 | NbI5 | Mo | Tc | Ru | Rh | Pd | AgI | CdI2 | InI3 | SnI4, SnI2 |
SbI3 | TeI4 | I | Xe |
| CsI | BaI2 | HfI4 | TaI5 | W | Re | Os | Ir | Pt | AuI | Hg2I2, HgI2 |
TlI | PbI2 | BiI3 | Po | AtI | Rn | |
| Fr | RaI2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | Ce | Pr | Nd | Pm | SmI2 | Eu | Gd | TbI3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | ThI4 | Pa | UI3, UI4 |
Np | Pu | Am | Cm | Bk | Cf | EsI3 | Fm | Md | No | Lr | |||
 |

