Chemistry:Copper(I) fluoride
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
Copper(I) fluoride
| |
| Systematic IUPAC name
Fluorocopper[1] | |
| Other names
Cuprous fluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| CuF | |
| Molar mass | 82.544 g·mol−1 |
| Density | 7.1 g cm−3 |
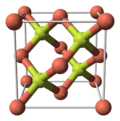
| Structure | |
| sphalerite | |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| NFPA 704 (fire diamond) | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 1 mg/m3 (as Cu)[2] |
REL (Recommended)
|
TWA 1 mg/m3 (as Cu)[2] |
IDLH (Immediate danger)
|
TWA 100 mg/m3 (as Cu)[2] |
| Related compounds | |
Other anions
|
Copper(I) chloride Copper(I) bromide Copper(I) iodide |
Other cations
|
Silver(I) fluoride Gold(I) fluoride |
Related compounds
|
Copper(II) fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Copper(I) fluoride or cuprous fluoride is an inorganic compound with the chemical formula CuF. Its existence is uncertain. It was reported in 1933 to have a sphalerite-type crystal structure.[3] Modern textbooks state that CuF is not known,[4] since fluorine is so electronegative that it will always oxidise copper to its +2 oxidation state.[5] Complexes of CuF such as [(Ph3P)3CuF] are, however, known and well characterised.[6]
Synthesis and reactivity
Unlike other copper(I) halides like copper(I) chloride, copper(I) fluoride tends to disproportionate into copper(II) fluoride and copper in a one-to-one ratio at ambient conditions, unless it is stabilised through complexation as in the example of [Cu(N2)F].[7]
- 2CuF → Cu + CuF2
See also
- Copper(II) fluoride, the other simple fluoride of copper
References
- ↑ "Copper Monofluoride - PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information. https://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=3084153&loc=ec_rcs.
- ↑ 2.0 2.1 2.2 NIOSH Pocket Guide to Chemical Hazards. "#0150". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0150.html.
- ↑ Ebert, F.; Woitinek, H. (1933). "Kristallstrukturen von Fluoriden. II. HgF, HgF2, CuF und CuF2". Z. anorg. allg. Chem. 210 (3): 269–272. doi:10.1002/zaac.19332100307.
- ↑ Housecroft, C. E.; Sharpe, A. G. (2008). Inorganic Chemistry (3rd ed.). Prentice Hall. pp. 737–738. ISBN 978-0-13-175553-6.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1183–1185. ISBN 978-0-08-037941-8.
- ↑ Gulliver, D. J.; Levason, W.; Webster, M. (1981). "Coordination Stabilised Copper(I) Fluoride. Crystal and Molecular Structure of Fluorotris(triphenylphosphine)copper(I)·Ethanol (1/2), Cu(PPh3)3F·2EtOH". Inorg. Chim. Acta 52: 153–159. doi:10.1016/S0020-1693(00)88590-4.
- ↑ Francis, Simon G.; Matthews, Steven L.; Poleshchuk, Oleg Kh; Walker, Nicholas R.; Legon, Anthony C. (2006-09-25). "N2-Cu-F: A Complex of Dinitrogen and Cuprous Fluoride Characterized by Rotational Spectroscopy". Angewandte Chemie 118 (38): 6489–6491. doi:10.1002/ange.200601988. PMID 16937427.
 |

