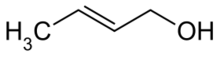
Chemistry:Crotyl alcohol
From HandWiki
| |
| Names | |
|---|---|
| Preferred IUPAC name
(2E)-But-2-en-1-ol | |
| Other names
(E)-But-2-en-1-ol
Crotyl alcohol Crotonyl alcohol 2-Butenol | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 2614 |
| |
| |
| Properties | |
| C4H8O | |
| Molar mass | 72.10 g/mol |
| Density | 0.8454 g/cm3 |
| Melting point | < 25 °C (77 °F; 298 K) |
| Boiling point | 121.2 °C (250.2 °F; 394.3 K) |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| H226, H302, H312, H315, H319 | |
| P210, P233, P240, P241, P242, P243, P264, P270, P280, P301+312, P302+352, P303+361+353, P305+351+338, P312, P321, P322, P330, P332+313, P337+313, P362, P363, P370+378, P403+235, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Crotyl alcohol, or crotonyl alcohol, is an unsaturated alcohol. It is a colourless liquid that is moderately soluble in water and miscible with most organic solvents. It exhibits cis-trans isomerism about the alkene group, and is a structural isomer of butanone.
It can be synthesized by the hydrogenation of crotonaldehyde. The compound is of little commercial interest,[2] but can be used as a reagent in laboratory organic synthesis.[3]
See also
References
- ↑ Merck Index, 11th Edition, 2604
- ↑ Falbe, Jürgen; Bahrmann, Helmut; Lipps, Wolfgang; Mayer, Dieter. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a01_279..
- ↑ C. John Blankley, Frederick J. Sauter, and Herbert O. House (1969). "Crotyl diazoacetate". Organic Syntheses 49: 22. doi:10.15227/orgsyn.049.0022. http://www.orgsyn.org/demo.aspx?prep=CV5P0258.
 |
