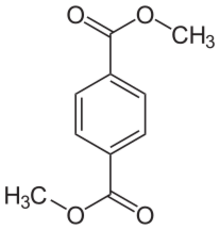
Chemistry:Dimethyl terephthalate
| |
| Names | |
|---|---|
| Preferred IUPAC name
Dimethyl benzene-1,4-dicarboxylate | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | DMT |
| 1107185 | |
| ChEBI | |
| ChemSpider | |
| EC Number |
|
| MeSH | Dimethyl+4-phthalate |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| C10H10O4 | |
| Molar mass | 194.186 g·mol−1 |
| Appearance | white solid |
| Density | 1.2 g/cm3 |
| Melting point | 142 °C (288 °F; 415 K) |
| Boiling point | 288 °C (550 °F; 561 K) |
| Acidity (pKa) | −7.21 |
| Basicity (pKb) | −6.60 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Dimethyl terephthalate (DMT) is an organic compound with the formula C
6H
4(COOCH
3)
2. It is the diester formed from terephthalic acid and methanol. It is a white solid that melts to give a distillable colourless liquid.[1][2] it is an intermediate in some schemes for the recycling of PET, e.g. from plastic bottles.[3]
Production
Dimethyl terephthalate (DMT) can be prepared via a direct esterification of terephthalic acid with methanol:
- C
8H
6O
4 + 2 CH
3OH → C
10H
10O
4 + 2 H
2O
On a commercial scale, this reaction is usually done at 250–300 °C (482–572 °F) using o-xylene as a solvent. The dimethyl terephthalate that is formed is then purified by distillation. Even terephthalic acid of low purity may be used in this method.
Alternatively, it can be prepared from para-xylene by alternating oxidation and methyl-esterification steps via methyl para-toluate (PT).[1]
Uses
DMT is used in the production of polyesters, including polyethylene terephthalate (PET), polytrimethylene terephthalate (PTT), and polybutylene terephthalate (PBT). These polymers are produced by transesterification with a diol. In the production of PET, the transesterification initially produces 2-hydroxyethyl methyl terephthalate.
Hydrogenation of DMT affords the diol cyclohexanedimethanol, which is a useful monomer.[4]
Hazards
DMT is not directly dangerous to humans, but it is harmful to aquatic organisms, so environmental releases are avoided. It is flammable and the dust may form explosive mixtures with air.[5]
References
- ↑ 1.0 1.1 J. Sheehan, Richard. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a26_193.
- ↑ Elliott, John H.; Chris, Marjorie D. (1968-10-01). "Thermodynamic properties of high-purity dimethyl terephthalate" (in en). Journal of Chemical & Engineering Data 13 (4): 475–479. doi:10.1021/je60039a005. ISSN 0021-9568. https://pubs.acs.org/doi/abs/10.1021/je60039a005.
- ↑ Zhang, Hui; Xia, Qing; Yang, Ying; Zhang, Feng-Bao; Zhang, Guo-Liang (2013-04-10). "Solubility of Dimethyl Terephthalate and Monomethyl Terephthalate in the Methanol Aqueous Solution and Its Application To Recycle Monomethyl Terephthalate from Crude Dimethyl Terephthalate" (in en). Industrial & Engineering Chemistry Research 52 (14): 5230–5234. doi:10.1021/ie400421e. ISSN 0888-5885. https://pubs.acs.org/doi/10.1021/ie400421e.
- ↑ Baikerikar, Kiran K.; Tulchinsky, Michael L.; Argyropoulos, John (March 2010). "UV curable, liquid diacrylate monomers based on (cis,trans)-1,3/1,4-cyclohexanedimethanol" (in en). Journal of Coatings Technology and Research 7 (2): 175–188. doi:10.1007/s11998-009-9185-1. ISSN 1547-0091. http://link.springer.com/10.1007/s11998-009-9185-1.
- ↑ International Chemical Safety Card 0262
External links
 |
