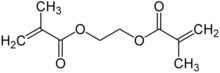
Chemistry:Ethylene glycol dimethacrylate
From HandWiki
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Ethane-1,2-diyl bis(2-methylprop-2-enoate) | |
| Other names
Methacrylic acid, ethylene ester; 1,2-Bis(methacryloyloxy)ethane; 1,2-Ethanediol dimethacrylate; Diglycol dimethacrylate; Ethanediol dimethacrylate; Ethylene dimethacrylate; Ethylene glycol bis(methacrylate); Ethylene glycol dimethacrylate; Ethylene methacrylate; 2-(Methacryloyloxy)ethyl methacrylate
| |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | EGDMA |
| 1776663 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| 637376 | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| C10H14O4 | |
| Molar mass | 198.218 g·mol−1 |
| Density | 1.051 g/mL |
| Melting point | −40 °C (−40 °F; 233 K) |
| Boiling point | 98 to 100 °C (208 to 212 °F; 371 to 373 K) (5 mmHg) |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H317, H319, H335, H412 | |
| P261, P264, P271, P272, P273, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P333+313, P337+313, P362, P363, P403+233, P405, P501 | |
| Flash point | 101 °C (214 °F; 374 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Ethylene glycol dimethylacrylate (EGDMA) is a diester formed by condensation of two equivalents of methacrylic acid and one equivalent of ethylene glycol.[2]
EGDMA can be used in free radical copolymer crosslinking reactions. When used with methyl methacrylate, it leads to gel point at relatively low concentrations because of the nearly equivalent reactivities of all the double bonds involved.
It is used as a monomer to prepare Hydroxyapatite/Poly methyl methacrylate composites. EGDMA can be used in free radical copolymer crosslinking reactions.
Its toxicity profile has been fairly well studied.[3] It is sometimes called ethylene dimethacrylate.[4]
References
- ↑ |ALDRICH&N5=SEARCH_CONCAT_PNO|BRAND_KEY&F=SPEC Ethylene glycol dimethacrylate at Sigma-Aldrich
- ↑ Bielstein 2, IV, 1532
- ↑ Bielecka-Kowalska, Anna; Czarny, Piotr; Wigner, Paulina; Synowiec, Ewelina; Kowalski, Bartosz; Szwed, Marzena; Krupa, Renata; Toma, Monika et al. (March 2018). "Ethylene glycol dimethacrylate and diethylene glycol dimethacrylate exhibits cytotoxic and genotoxic effect on human gingival fibroblasts via induction of reactive oxygen species" (in en). Toxicology in Vitro 47: 8–17. doi:10.1016/j.tiv.2017.10.028. https://linkinghub.elsevier.com/retrieve/pii/S0887233317303363.
- ↑ PubChem. "Ethylene glycol dimethacrylate" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/7355.
 |

