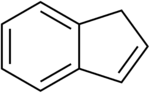
Chemistry:Indene
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1H-Indene[1] | |
| Other names
Benzocyclopentadiene
Indonaphthene Bicyclo[4.3.0]nona-1,3,5,7-tetraene | |
| Identifiers | |
3D model (JSmol)
|
|
| 635873 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| 27265 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C 9H 8 | |
| Molar mass | 116.16 |
| Appearance | Colorless liquid[2] |
| Density | 0.997 g/mL |
| Melting point | −1.8 °C (28.8 °F; 271.3 K) |
| Boiling point | 181.6 °C (358.9 °F; 454.8 K) |
| Insoluble | |
| Acidity (pKa) | 20.1 (in DMSO)[3] |
| −80.89×10−6 cm3/mol | |
| Hazards | |
| Main hazards | Flammable |
| Flash point | 78.3 °C (172.9 °F; 351.4 K) |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
none[2] |
REL (Recommended)
|
TWA 10 ppm (45 mg/m3)[2] |
IDLH (Immediate danger)
|
N.D.[2] |
| Related compounds | |
Related compounds
|
Benzofuran, Benzothiophene, Indole |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Indene is an aromatic, polycyclic hydrocarbon with chemical formula C
9H
8. It is composed of a benzene ring fused with a cyclopentene ring. This flammable liquid is colorless although samples often are pale yellow. The principal industrial use of indene is in the production of indene/coumarone thermoplastic resins. Substituted indenes and their closely related indane derivatives are important structural motifs found in many natural products and biologically active molecules, such as sulindac.[4]
Isolation
Indene occurs naturally in coal-tar fractions boiling around 175–185 °C. It can be obtained by heating this fraction with sodium to precipitate solid "sodio-indene". This step exploits indene's weak acidity evidenced by its deprotonation by sodium to give the indenyl derivative. The sodio-indene is converted back to indene by steam distillation.[5]
Reactivity
Indene readily polymerises. Oxidation of indene with acid dichromate yields homophthalic acid (o-carboxylphenylacetic acid). It condenses with diethyl oxalate in the presence of sodium ethoxide to form indene–oxalic ester, and with aldehydes or ketones in the presence of alkali to form benzofulvenes, which are highly coloured. Treatment of indene with organolithium reagents gives lithium indenyl compounds:
Indenyl is a ligand in organometallic chemistry, giving rise to many transition metal indenyl complexes.[6]
See also
References
- ↑ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. pp. 207. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ↑ 2.0 2.1 2.2 2.3 NIOSH Pocket Guide to Chemical Hazards. "#0340". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0340.html.
- ↑ Bordwell FG (1988). "Equilibrium acidities in dimethyl sulfoxide solution". Accounts of Chemical Research 21 (12): 456–463. doi:10.1021/ar00156a004. Bordwell pKa Table in DMSO
- ↑ Wu, Jie; Qiu, Guanyinsheng (2014). "Generation of Indene Derivatives by Tandem Reactions". Synlett 25 (19): 2703–2713. doi:10.1055/s-0034-1379318.
- ↑ Collin, Gerd; Mildenberg, Rolf; Zander, Mechthild; Höke, Hartmut; McKillip, William; Freitag, Werner; Imöhl, Wolfgang. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH.
- ↑ O'Connor, Joseph M.; Casey, Charles P. (1987). "Ring-Slippage Chemistry of Transition Metal Cyclopentadienyl and Indenyl Complexes". Chemical Reviews 87 (2): 307–318. doi:10.1021/cr00078a002.
External links
- W. v. Miller, Rohde (1890). "Zur Synthese von Indenderivaten". Berichte der Deutschen Chemischen Gesellschaft 23 (2): 1881–1886. doi:10.1002/cber.18900230227. https://zenodo.org/record/1425600.
- W. v. Miller, Rohde (1890). "Zur Synthese von Indenderivaten". Berichte der Deutschen Chemischen Gesellschaft 23 (2): 1887–1902. doi:10.1002/cber.18900230228. https://zenodo.org/record/1425559.
- Finar, I. L. (1985). Organic Chemistry. Longman Scientific & Technical. ISBN 0-582-44257-5.
 |
