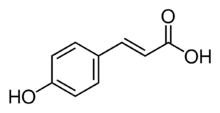
Chemistry:P-Coumaric acid
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
(2E)-3-(4-Hydroxyphenyl)prop-2-enoic acid | |
| Other names
(E)-3-(4-Hydroxyphenyl)-2-propenoic acid
(E)-3-(4-Hydroxyphenyl)acrylic acid para-Coumaric acid 4-Hydroxycinnamic acid trans-4-Hydroxycinnamic acid β-(4-Hydroxyphenyl)acrylic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| 2207383 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| 2245630 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C9H8O3 | |
| Molar mass | 164.160 g·mol−1 |
| Melting point | 210 to 213 °C (410 to 415 °F; 483 to 486 K) |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Danger |
| H315, H319, H335[1] | |
| P261, P264, P271, P280, P302+352, P305+351+338 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
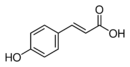
p-Coumaric acid is an organic compound with the formula HOC6H4CH=CHCO2H. It is one of the three isomers of coumaric acid. It is a white solid that is only slightly soluble in water but very soluble in ethanol and diethyl ether.
Natural occurrences
It is a precursor to many natural products, especially lignols, precursors to the woody mass that comprise many plants.[2] Of the myriad occurrences, p-coumaric acid can be found in Gnetum cleistostachyum.[3]
In food
p-Coumaric acid can be found in a wide variety of edible plants and fungi such as peanuts, navy beans, tomatoes, carrots, basil and garlic. It is found in wine and vinegar.[4] It is also found in barley grain.[5]
p-Coumaric acid from pollen is a constituent of honey.[6]
Derivatives
p-Coumaric acid glucoside can also be found in commercial breads containing flaxseed.[7] Diesters of p-coumaric acid can be found in carnauba wax.
Biosynthesis
It is biosynthesized from cinnamic acid by the action of the P450-dependent enzyme 4-cinnamic acid hydroxylase (C4H).
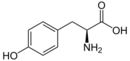
It is also produced from L-tyrosine by the action of tyrosine ammonia lyase (TAL).
Biosynthetic building block
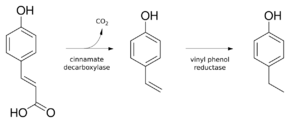
p-Coumaric acid is the precursor of 4-ethylphenol produced by the yeast Brettanomyces in wine. The enzyme cinnamate decarboxylase catalyzes the conversion of p-coumaric acid into 4-vinylphenol.[8] Vinyl phenol reductase then catalyzes the reduction of 4-vinylphenol to 4-ethylphenol. Coumaric acid is sometimes added to microbiological media, enabling the positive identification of Brettanomyces by smell.
cis-p-Coumarate glucosyltransferase is an enzyme that uses uridine diphosphate glucose and cis-p-coumarate to produce 4′-O-β-D-glucosyl-cis-p-coumarate and uridine diphosphate (UDP). This enzyme belongs to the family of glycosyltransferases, specifically the hexosyltransferases.[9]
Phloretic acid, found in the rumen of sheep fed with dried grass, is produced by hydrogenation of the 2-propenoic side chain of p-coumaric acid.[10]
The enzyme, resveratrol synthase, also known as stilbene synthase, catalyzes the synthesis of resveratrol ultimately from a tetraketide derived from 4-coumaroyl CoA.[11]
p-Coumaric acid is a cofactor of photoactive yellow proteins (PYP), a homologous group of proteins found in many eubacteria.[12]
p-Coumaric acid is found as the base moiety of caleicine, one of many sesquiterpenes in Calea ternifolia.
See also
- o-coumaric acid
- m-coumaric acid
- Coumarin
- Coumaroyl-Coenzyme A
- Ferulic acid
- Cinnamic acid
- Phenolic content in wine
- p-Coumaroylated anthocyanins
References
- ↑ Sigma-Aldrich Co., p-Coumaric acid. Retrieved on 2022-02-15.
- ↑ Wout Boerjan, John Ralph, Marie Baucher "Lignin Biosynthesis" Annu. Rev. Plant Biol. 2003, vol. 54, pp. 519–46. doi:10.1146/annurev.arplant.54.031902.134938
- ↑ "Stilbene derivatives from Gnetum cleistostachyum". Journal of Asian Natural Products Research 7 (2): 131–7. April 2005. doi:10.1080/10286020310001625102. PMID 15621615.
- ↑ "Analysis of polyphenolic compounds of different vinegar samples". Zeitschrift für Lebensmittel-Untersuchung und -Forschung 199: 29–31. 1994. doi:10.1007/BF01192948.
- ↑ "Phenolic compounds of barley grain and their implication in food product discoloration". Journal of Agricultural and Food Chemistry 54 (26): 9978–84. December 2006. doi:10.1021/jf060974w. PMID 17177530.
- ↑ "Honey constituents up-regulate detoxification and immunity genes in the western honey bee Apis mellifera". Proceedings of the National Academy of Sciences of the United States of America 110 (22): 8842–6. May 2013. doi:10.1073/pnas.1303884110. PMID 23630255. Bibcode: 2013PNAS..110.8842M.
- ↑ "Phenolic glucosides in bread containing flaxseed". Food Chemistry 110 (4): 997–9. October 2008. doi:10.1016/j.foodchem.2008.02.088. PMID 26047292.
- ↑ "Brettanomyces Monitoring by Analysis of 4-ethylphenol and 4-ethylguaiacol". http://www.etslabs.com/scripts/ets/pagetemplate/blank.asp?pageid=193.
- ↑ "Isolation, purification and characterization of UDP-glucose: cis-p-coumaric acid-β-D-glucosyltransferase from sphagnum fallax". Phytochemistry 46 (3): 449–453. 1997. doi:10.1016/S0031-9422(97)00337-3.
- ↑ "Influence of plant phenolic acids on growth and cellulolytic activity of rumen bacteria". Applied and Environmental Microbiology 44 (3): 597–603. September 1982. doi:10.1128/aem.44.3.597-603.1982. PMID 16346090. Bibcode: 1982ApEnM..44..597C.
- ↑ Wang, Chuanhong; Zhi, Shuang; Liu, Changying; Xu, Fengxiang; Zhao, Aichun; Wang, Xiling; Ren, Yanhong; Li, Zhengang et al. (2017). "Characterization of Stilbene Synthase Genes in Mulberry (Morus atropurpurea) and Metabolic Engineering for the Production of Resveratrol in Escherichia coli". Journal of Agricultural and Food Chemistry 65 (8): 1659–1668. doi:10.1021/acs.jafc.6b05212. PMID 28168876.
- ↑ "Thiol ester-linked p-coumaric acid as a new photoactive prosthetic group in a protein with rhodopsin-like photochemistry". Biochemistry 33 (47): 13959–62. November 1994. doi:10.1021/bi00251a001. PMID 7947803. http://dare.uva.nl/personal/pure/en/publications/pcoumaric-acid-a-new-photoactive-chromophore-of-a-yellow-photoreceptor-protein-with-rhodopsinlike-characteristics(6bd029db-bd7c-439b-af3a-46357240b242).html.
 |