Chemistry:Tyrosine
L-Tyrosine or tyrosine (symbol Tyr or Y)[1] or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a conditionally essential amino acid with a polar side group. The word "tyrosine" is from the Greek tyrós, meaning cheese, as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese.[2][3] It is called tyrosyl when referred to as a functional group or side chain. While tyrosine is generally classified as a hydrophobic amino acid, it is more hydrophilic than phenylalanine.[4] It is encoded by the codons UAC and UAU in messenger RNA.
The one-letter symbol Y was assigned to tyrosine for being alphabetically nearest of those letters available. Note that T was assigned to the structurally simpler threonine, U was avoided for its similarity with V for valine, W was assigned to tryptophan, while X was reserved for undetermined or atypical amino acids.[5] The mnemonic tYrosine was also proposed.[6]
Functions
Aside from being a proteinogenic amino acid, tyrosine has a special role by virtue of the phenol functionality. Its hydroxy group is able to form the ester linkage, with phosphate in particular. Phosphate groups are transferred to tyrosine residues by way of protein kinases. This is one of the post-translational modifications. Phosphorylated tyrosine occurs in proteins that are part of signal transduction processes.
Similar functionality is also presented in serine and threonine, whose side chains have a hydroxy group, but are alcohols. Phosphorylation of these three amino acids' moieties (including tyrosine) creates a negative charge on their ends, which is greater than the negative charge of the only negatively charged aspartic and glutamic acids. Phosphorylated proteins keep these same properties—which are useful for more reliable protein-protein interactions—by means of phosphotyrosine, phosphoserine and phosphothreonine.[7]
Binding sites for a signalling phosphoprotein may be diverse in their chemical structure.[8]
Phosphorylation of the hydroxyl group can change the activity of the target protein, or may form part of a signaling cascade via SH2 domain binding.[9]
A tyrosine residue also plays an important role in photosynthesis. In chloroplasts (photosystem II), it acts as an electron donor in the reduction of oxidized chlorophyll. In this process, it loses the hydrogen atom of its phenolic OH-group. This radical is subsequently reduced in the photosystem II by the four core manganese clusters.[10]
Dietary requirements and sources
The Dietary Reference Intake for tyrosine is usually estimated together with phenylalanine. It varies depending on an estimate method, however the ideal proportion of these two amino acids is considered to be 60:40 (phenylalanine:tyrosine) as a human body has such composition.[11] Tyrosine, which can also be synthesized in the body from phenylalanine, is found in many high-protein food products such as meat, fish, cheese, cottage cheese, milk, yogurt, peanuts, almonds, pumpkin seeds, sesame seeds, soy protein and lima beans.[12][13] For example, the white of an egg has about 250 mg per egg,[14] while beef, lamb, pork, tuna, salmon, chicken, and turkey contain about 500–1000 mg per 3 ounces (85 g) portion.[14][15]
Biosynthesis
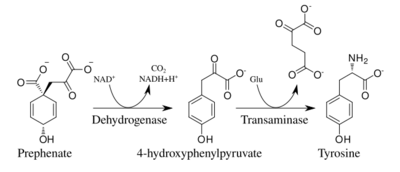
In plants and most microorganisms, tyrosine is produced via prephenate, an intermediate on the shikimate pathway. Prephenate is oxidatively decarboxylated with retention of the hydroxyl group to give p-hydroxyphenylpyruvate, which is transaminated using glutamate as the nitrogen source to give tyrosine and α-ketoglutarate.
Mammals synthesize tyrosine from the essential amino acid phenylalanine (Phe), which is derived from food. The conversion of Phe to Tyr is catalyzed by the enzyme phenylalanine hydroxylase, a monooxygenase. This enzyme catalyzes the reaction causing the addition of a hydroxyl group to the end of the 6-carbon aromatic ring of phenylalanine, such that it becomes tyrosine.
Metabolism

Phosphorylation and sulfation
Some of the tyrosine residues can be tagged (at the hydroxyl group) with a phosphate group (phosphorylated) by protein kinases. In its phosphorylated form, tyrosine is called phosphotyrosine. Tyrosine phosphorylation is considered to be one of the key steps in signal transduction and regulation of enzymatic activity. Phosphotyrosine can be detected through specific antibodies. Tyrosine residues may also be modified by the addition of a sulfate group, a process known as tyrosine sulfation.[16] Tyrosine sulfation is catalyzed by tyrosylprotein sulfotransferase (TPST). Like the phosphotyrosine antibodies mentioned above, antibodies have recently been described that specifically detect sulfotyrosine.[17]
Precursor to neurotransmitters and hormones
In dopaminergic cells in the brain, tyrosine is converted to L-DOPA by the enzyme tyrosine hydroxylase (TH). TH is the rate-limiting enzyme involved in the synthesis of the neurotransmitter dopamine. Dopamine can then be converted into other catecholamines, such as norepinephrine (noradrenaline) and epinephrine (adrenaline).
The thyroid hormones triiodothyronine (T3) and thyroxine (T4) in the colloid of the thyroid are also derived from tyrosine. {{Annotated image 4 | caption = {{{caption|In humans, catecholamines and phenethylaminergic trace amines are derived from the amino acid {{nowrap|L-phenylalanine}}.}}} | header_background = #F0F8FF | header = Biosynthetic pathways for catecholamines and trace amines in the human brain<ref name="Trace amine template 1">Broadley KJ (March 2010). "The vascular effects of trace amines and amphetamines". Pharmacol. Ther. 125 (3): 363–375. doi:10.1016/j.pharmthera.2009.11.005. PMID 19948186.</ref>[18][19] | alt = Graphic of catecholamine and trace amine biosynthesis | image = Catecholamine and trace amine biosynthesis.png | image-width = 580 | image-left = 5 | image-top = 0 | align = center | width = 590 | height = 585 | annot-font-size = 14 | annot-text-align = center | annotations =
{{annotation|50|565|{{if pagename|Adrenaline=Adrenaline|Epinephrine=Epinephrine|Catecholamine=Epinephrine|other=Epinephrine}}}}
{{annotation|245|60|{{if pagename|Phenethylamine=Phenethylamine|Trace amine=Phenethylamine|Neurobiological effects of physical exercise={{highlight|Phenethylamine}}|other=Phenethylamine}}}}
{{annotation|245|565|{{if pagename|Norepinephrine=Norepinephrine|Adrenaline=Noradrenaline|Catecholamine=Norepinephrine|other=Norepinephrine}}}}
{{annotation|440|295|p-Octopamine}}}}
pathway
CYP2D6
pathway
Precursor to other compounds
The latex of Papaver somniferum, the opium poppy, has been shown to convert tyrosine into the alkaloid morphine and the bio-synthetic pathway has been established from tyrosine to morphine by using Carbon-14 radio-labelled tyrosine to trace the in-vivo synthetic route.[20]Tyrosine ammonia lyase (TAL) is an enzyme in the natural phenols biosynthesis pathway. It transforms L-tyrosine into p-coumaric acid. Tyrosine is also the precursor to the pigment melanin. Tyrosine (or its precursor phenylalanine) is needed to synthesize the benzoquinone structure which forms part of coenzyme Q10.[21][22]
Degradation
The decomposition of L-tyrosine (syn. para-hydroxyphenylalanine) begins with an α-ketoglutarate dependent transamination through the tyrosine transaminase to para-hydroxyphenylpyruvate. The positional description para, abbreviated p, mean that the hydroxyl group and side chain on the phenyl ring are across from each other (see the illustration below).
The next oxidation step catalyzes by p-hydroxyphenylpyruvate dioxygenase and splitting off CO2 homogentisate (2,5-dihydroxyphenyl-1-acetate).[23] In order to split the aromatic ring of homogentisate, a further dioxygenase, homogentisate 1,2-dioxygenase is required. Thereby, through the incorporation of a further O2 molecule, maleylacetoacetate is created.
Fumarylacetoacetate is created by maleylacetoacetate cis-trans-isomerase through rotation of the carboxyl group created from the hydroxyl group via oxidation. This cis-trans-isomerase contains glutathione as a coenzyme. Fumarylacetoacetate is finally split by the enzyme fumarylacetoacetate hydrolase through the addition of a water molecule.
Thereby fumarate (also a metabolite of the citric acid cycle) and acetoacetate (3-ketobutyroate) are liberated. Acetoacetate is a ketone body, which is activated with succinyl-CoA, and thereafter it can be converted into acetyl-CoA, which in turn can be oxidized by the citric acid cycle or be used for fatty acid synthesis.
Phloretic acid is also a urinary metabolite of tyrosine in rats.[24]
Ortho- and meta-tyrosine

Three structural isomers of L-tyrosine are known. In addition to the common amino acid L-tyrosine, which is the para isomer (para-tyr, p-tyr or 4-hydroxyphenylalanine), there are two additional regioisomers, namely meta-tyrosine (also known as 3-hydroxyphenylalanine, L-m-tyrosine, and m-tyr) and ortho-tyrosine (o-tyr or 2-hydroxyphenylalanine), that occur in nature. The m-tyr and o-tyr isomers, which are rare, arise through non-enzymatic free-radical hydroxylation of phenylalanine under conditions of oxidative stress.[25][26]
Medical use
Tyrosine is a precursor to neurotransmitters and increases plasma neurotransmitter levels (particularly dopamine and norepinephrine),[27] but has little if any effect on mood in normal subjects.[28][29][30]
A 2015 systematic review found that "tyrosine loading acutely counteracts decrements in working memory and information processing that are induced by demanding situational conditions such as extreme weather or cognitive load" and therefore "tyrosine may benefit healthy individuals exposed to demanding situational conditions".[31]
Industrial synthesis
L-Tyrosine is used in pharmaceuticals, dietary supplements, and food additives. Two methods were formerly used to manufacture L-tyrosine. The first involves the extraction of the desired amino acid from protein hydrolysates using a chemical approach. The second utilizes enzymatic synthesis from phenolics, pyruvate, and ammonia through the use of tyrosine phenol-lyase.[32] Advances in genetic engineering and the advent of industrial fermentation have shifted the synthesis of L-tyrosine to the use of engineered strains of E. coli.[33][32]
See also
References
- ↑ "Nomenclature and Symbolism for Amino Acids and Peptides". IUPAC-IUB Joint Commission on Biochemical Nomenclature. 1983. http://www.chem.qmul.ac.uk/iupac/AminoAcid/AA1n2.html.
- ↑ "Tyrosine". The Columbia Electronic Encyclopedia, 6th ed. Infoplease.com — Columbia University Press. 2007. http://www.infoplease.com/ce6/sci/A0849873.html. Retrieved 2008-04-20.
- ↑ "Tyrosine". Online Etymology Dictionary. 2001. http://www.etymonline.com/index.php?term=tyrosine.
- ↑ "Amino Acids - Tyrosine". http://www.biology.arizona.edu/biochemistry/problem_sets/aa/Tyrosine.html.
- ↑ "IUPAC-IUB Commission on Biochemical Nomenclature A One-Letter Notation for Amino Acid Sequences" (in en). Journal of Biological Chemistry 243 (13): 3557–3559. 10 July 1968. doi:10.1016/S0021-9258(19)34176-6. https://www.jbc.org/article/S0021-9258(19)34176-6/pdf.
- ↑ Saffran, M. (April 1998). "Amino acid names and parlor games: from trivial names to a one-letter code, amino acid names have strained students' memories. Is a more rational nomenclature possible?" (in en). Biochemical Education 26 (2): 116–118. doi:10.1016/S0307-4412(97)00167-2. http://linkinghub.elsevier.com/retrieve/pii/S0307441297001672.
- ↑ Hunter, Tony (2012-09-19). "Why nature chose phosphate to modify proteins" (in en). Philosophical Transactions of the Royal Society B: Biological Sciences 367 (1602): 2513–2516. doi:10.1098/rstb.2012.0013. ISSN 0962-8436. PMID 22889903.
- ↑ Lu, Zheng-Chang; Jiang, Fan; Wu, Yun-Dong (2021-12-11). "Phosphate binding sites prediction in phosphorylation-dependent protein-protein interactions". Bioinformatics 37 (24): 4712–4718. doi:10.1093/bioinformatics/btab525. ISSN 1367-4811. PMID 34270697.
- ↑ Liu, Bernard A.; Nash, Piers D. (2012-09-19). "Evolution of SH2 domains and phosphotyrosine signalling networks" (in en). Philosophical Transactions of the Royal Society B: Biological Sciences 367 (1602): 2556–2573. doi:10.1098/rstb.2012.0107. ISSN 0962-8436. PMID 22889907.
- ↑ Barry, Bridgette A. (January 2015). "Reaction dynamics and proton coupled electron transfer: studies of tyrosine-based charge transfer in natural and biomimetic systems". Biochimica et Biophysica Acta (BBA) - Bioenergetics 1847 (1): 46–54. doi:10.1016/j.bbabio.2014.09.003. ISSN 0006-3002. PMID 25260243.
- ↑ "Aromatic amino acid requirements in healthy human subjects". The Journal of Nutrition 137 (6 Suppl 1): 1576S-1578S; discussion 1597S-1598S. June 2007. doi:10.1093/jn/137.6.1576S. PMID 17513429. https://academic.oup.com/jn/article/137/6/1576S/4664888.
- ↑ Nutient Ranking Tool. MyFoodData.com. https://tools.myfooddata.com/nutrient-ranking-tool/tyrosine/all/highest
- ↑ "Tyrosine". University of Maryland Medical Center. http://www.umm.edu/altmed/articles/tyrosine-000329.htm.
- ↑ 14.0 14.1 Top 10 Foods Highest in Tyrosine
- ↑ Nutient Ranking Tool. MyFoodData.com. https://tools.myfooddata.com https://tools.myfooddata.com/nutrient-ranking-tool/tyrosine/meats/highest/ounces/common/no
- ↑ "Detection and purification of tyrosine-sulfated proteins using a novel anti-sulfotyrosine monoclonal antibody". The Journal of Biological Chemistry 281 (49): 37877–87. December 2006. doi:10.1074/jbc.M609398200. PMID 17046811.
- ↑ "Focus on molecules: sulfotyrosine". Experimental Eye Research 105: 85–6. December 2012. doi:10.1016/j.exer.2012.02.014. PMID 22406006.
- ↑ "A renaissance in trace amines inspired by a novel GPCR family". Trends Pharmacol. Sci. 26 (5): 274–281. May 2005. doi:10.1016/j.tips.2005.03.007. PMID 15860375.
- ↑ "The endogenous substrates of brain CYP2D". Eur. J. Pharmacol. 724: 211–218. February 2014. doi:10.1016/j.ejphar.2013.12.025. PMID 24374199.
- ↑ Battersby, A. R.; Binks, R.; Harper, B. J. T. (1962-01-01). "692. Alkaloid biosynthesis. Part II. The biosynthesis of morphine" (in en). Journal of the Chemical Society: 3534–3544. doi:10.1039/JR9620003534. ISSN 0368-1769. https://pubs.rsc.org/en/content/articlelanding/1962/jr/jr9620003534.
- ↑ "Coenzyme Q--biosynthesis and functions". Biochemical and Biophysical Research Communications 396 (1): 74–9. May 2010. doi:10.1016/j.bbrc.2010.02.147. PMID 20494114. Bibcode: 2010BBRC..396...74B.
- ↑ Acosta, Manuel Jesús; Vazquez Fonseca, Luis; Desbats, Maria Andrea; Cerqua, Cristina; Zordan, Roberta; Trevisson, Eva; Salviati, Leonardo (2016). "Coenzyme Q biosynthesis in health and disease". Biochimica et Biophysica Acta (BBA) - Bioenergetics 1857 (8): 1079–1085. doi:10.1016/j.bbabio.2016.03.036. PMID 27060254.
- ↑ "The Incidence of Transient Neonatal Tyrosinemia Within a Mexican Population". Journal of Inborn Errors of Metabolism and Screening 5: 232640981774423. 27 November 2017. doi:10.1177/2326409817744230.
- ↑ "Urinary phenolic acid metabolities of tyrosine". Journal of Biological Chemistry 235 (9): 2649–2652. 1960. doi:10.1016/S0021-9258(19)76930-0.
- ↑ "Urinary ortho-tyrosine excretion in diabetes mellitus and renal failure: evidence for hydroxyl radical production". Kidney International 68 (5): 2281–7. November 2005. doi:10.1111/j.1523-1755.2005.00687.x. PMID 16221230.
- ↑ "Accumulation of the hydroxyl free radical markers meta-, ortho-tyrosine and DOPA in cataractous lenses is accompanied by a lower protein and phenylalanine content of the water-soluble phase". Free Radical Research 39 (12): 1359–66. December 2005. doi:10.1080/10715760500307107. PMID 16298866.
- ↑ "Effects of tyrosine and tryptophan ingestion on plasma catecholamine and 3,4-dihydroxyphenylacetic acid concentrations". The Journal of Clinical Endocrinology and Metabolism 57 (4): 760–3. October 1983. doi:10.1210/jcem-57-4-760. PMID 6885965.
- ↑ "Diet-induced mood changes in normal populations". Journal of Psychiatric Research 17 (2): 147–54. 1982. doi:10.1016/0022-3956(82)90016-4. PMID 6764931.
- ↑ "Effect of tyrosine on cognitive function and blood pressure under stress". Brain Research Bulletin 33 (3): 319–23. 1994. doi:10.1016/0361-9230(94)90200-3. PMID 8293316.
- ↑ "The effects of dietary neurotransmitter precursors on human behavior". The American Journal of Clinical Nutrition 42 (2): 366–70. August 1985. doi:10.1093/ajcn/42.2.366. PMID 4025206.
- ↑ Jung, Sophie E; Hase, Adrian; ann het Rot, Marije (2015). "Behavioral and cognitive effects of tyrosine intake in healthy human adults". Pharmacology Biochemistry and Behavior 133: 1–6. doi:10.1016/j.pbb.2015.03.008. PMID 25797188.
- ↑ 32.0 32.1 "Perspectives of biotechnological production of L-tyrosine and its applications". Applied Microbiology and Biotechnology 77 (4): 751–62. December 2007. doi:10.1007/s00253-007-1243-y. PMID 17968539.
- ↑ "Biotechnological production of L-tyrosine and derived compounds". Process Biochemistry 47 (7): 1017–1026. 2012. doi:10.1016/j.procbio.2012.04.005.
External links
- Tyrosine MS Spectrum
- Tyrosine metabolism
- Phenylalanine and tyrosine biosynthesis
- Phenylalanine, Tyrosine, and tryptophan biosynthesis
{{Navbox
| name = Neurotransmitter metabolism intermediates | title = Neurotransmitter metabolic intermediates | state = autocollapse| | listclass = hlist
| group1 = catecholamines | list1 = {{Navbox|child
| group1 = Anabolism
(tyrosine→epinephrine) | list1 =
- Tyrosine → Levodopa → [[Chemistry:Dop[[Chemistry:Dopamine → Norepinephrine|Norepinephrine]] → Epinephrine
| group2 = Catabolism/
metabolites
| list2 =
| dopamine: | |
|---|---|
| norepinephrine: | |
| epinephrine: |
}}
| group3 = tryptophan→serotonin| list3 =
| anabolism: | |
|---|---|
| catabolism: |