Chemistry:Relamorelin
 | |
| Clinical data | |
|---|---|
| Routes of administration | Subcutaneous |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
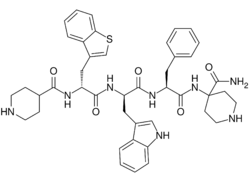
| Formula | C43H50N8O5S |
| Molar mass | 790.98 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Relamorelin (INN, USAN) (developmental code names RM-131, BIM-28131, BIM-28163) is a synthetic peptide, centrally penetrant, selective agonist of the ghrelin/growth hormone secretagogue receptor (GHSR) which is under development by Allergan pharmaceuticals for the treatment of diabetic gastroparesis, chronic idiopathic constipation, and anorexia nervosa.[1][2][3] It is a pentapeptide and an analogue of ghrelin with improved potency and pharmacokinetics.[1][2] In humans, relamorelin produces increases in plasma growth hormone, prolactin, and cortisol levels,[2][4] and, like other GHSR agonists, increases appetite.[3] As of June 2015, relamorelin is in phase II clinical trials for diabetic gastroparesis and constipation.[5] The United States Food and Drug Administration (FDA) has granted Fast Track designation to relamorelin for diabetic gastroparesis.[6] The development of the drug is uncertain as the most recent mention of it was in a 2019 SEC filing from the drug manufacturer lists the drug's expected launch year as 2024, but not in subsequent filings or press releases.[7][8]
See also
- List of growth hormone secretagogues
References
- ↑ 1.0 1.1 "Relamorelin. Ghrelin receptor agonist, treatment of constipation, treatment of anorexia nervosa, treatment of diabetic gastroparesis". Drugs of the Future 39 (11): 775. 2014. doi:10.1358/dof.2014.039.011.2231856. ISSN 0377-8282.
- ↑ 2.0 2.1 2.2 "Emerging treatments in Neurogastroenterology: relamorelin: a novel gastrocolokinetic synthetic ghrelin agonist". Neurogastroenterology and Motility 27 (3): 324–32. March 2015. doi:10.1111/nmo.12490. PMID 25545036.
- ↑ 3.0 3.1 "Relamorelin Relieves Constipation and Accelerates Colonic Transit in a Phase 2, Placebo-Controlled, Randomized Trial". Clinical Gastroenterology and Hepatology 13 (13): 2312–9.e1. December 2015. doi:10.1016/j.cgh.2015.04.184. PMID 26001337.
- ↑ "Randomized controlled phase Ib study of ghrelin agonist, RM-131, in type 2 diabetic women with delayed gastric emptying: pharmacokinetics and pharmacodynamics". Diabetes Care 36 (1): 41–8. January 2013. doi:10.2337/dc12-1128. PMID 22961573.
- ↑ "Early investigational therapeutics for gastrointestinal motility disorders: from animal studies to Phase II trials". Expert Opinion on Investigational Drugs 24 (6): 769–79. June 2015. doi:10.1517/13543784.2015.1025132. PMID 25971881.
- ↑ Rhythm Pharmaceuticals (2014). "Rhythm Presents Positive Phase 2 Study Results for Relamorelin for Chronic Constipation". PRNewsire. http://www.prnewswire.com/news-releases/rhythm-presents-positive-phase-2-study-results-for-relamorelin-for-chronic-constipation-719659361.html.
- ↑ "AbbVie Inc SEC filing 2019". November 7, 2019. https://investors.abbvie.com/node/13386/html?_ga=2.259876234.1226232138.1686226211-949662417.1686226211.
- ↑ "AbbVie Reports First-Quarter 2023 Financial Results". https://investors.abbvie.com/news-releases/news-release-details/abbvie-reports-first-quarter-2023-financial-results.
External links
- Relamorelin (RM-131): Ghrelin Peptide Agonist: A New Drug Class for the Treatment of GI Functional Disorders – Rhythm Pharmaceuticals
- Relamorelin – AdisInsight
 |
