Chemistry:Sodium tert-butoxide
| |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium tert-butoxide | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C4H9NaO | |
| Molar mass | 96.105 g·mol−1 |
| Density | 1.025 g/cm3 |
| Acidity (pKa) | 19[1] |
| Hazards | |
| Safety data sheet | [1] |
| Flash point | 14 °C (57 °F; 287 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
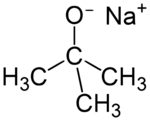
Sodium tert-butoxide (or sodium t-butoxide) is a chemical compound with the formula (CH3)3CONa (abbr. NaOtBu).[2] It is a strong, non-nucleophilic base. It is flammable and moisture sensitive. It is sometimes written in the chemical literature as sodium t-butoxide. It is similar in reactivity to the more common potassium tert-butoxide.
The compound can be produced by treating tert-butyl alcohol with sodium hydride.[3]
Reactions
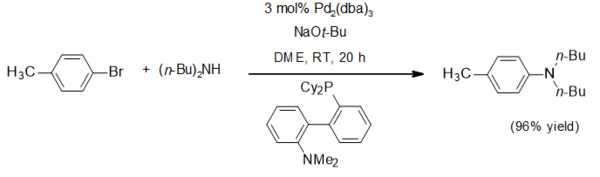
One application for sodium tert-butoxide is as a non-nucleophilic base. It has been widely used in the Buchwald–Hartwig amination, as in this typical example:[4]
Sodium tert-butoxide is used to prepare tert-butoxide complexes. For example hexa(tert-butoxy)ditungsten(III) is thus obtained by the salt metathesis reaction from a ditungsten heptachloride:[5]
- NaW2Cl7(THF)5 + 6 NaOBu-t → W2(OBu-t)6 + 7 NaCl + 5 THF
Structure
Sodium tert-butoxide forms clusters in the solid state, both hexamers[6] and nonamers.[7]
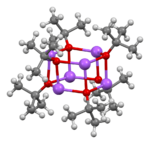 |
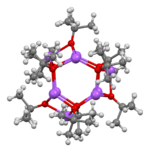
|
| hexamer | nonamer |
Related compounds
References
- ↑ Dewick, Paul M. (2013-03-20). Essentials of Organic Chemistry: For Students of Pharmacy, Medicinal Chemistry and Biological Chemistry. John Wiley & Sons. ISBN 978-1-118-68196-1. https://books.google.com/books?id=RrKgfuRwsqsC&dq=sodium+tert-butoxide+pKA&pg=PA157.
- ↑ http://www.sigmaaldrich.com/catalog/ProductDetail.do?lang=en&N4=359270%7CALDRICH&N5=SEARCH_CONCAT_PNO%7CBRAND_KEY&F=SPEC [bare URL]
- ↑ PM. Dewick, 2013. Essentials of Organic Chemistry: For Students of Pharmacy, Medicinal Chemistry and Biological Chemistry. John Wiley & Sons; p. 157. ISBN:978-1-118-68196-1
- ↑ Yang, Bryant H.; Buchwald, Stephen L. (1999). "Palladium-catalyzed amination of aryl halides and sulfonates". Journal of Organometallic Chemistry 576 (1–2): 125–146. doi:10.1016/S0022-328X(98)01054-7.
- ↑ Broderick, Erin M.; Browne, Samuel C.; Johnson, Marc J. A. (2014). "Dimolybdenum and Ditungsten Hexa(Alkoxides)". Inorganic Syntheses: Volume 36. 36. pp. 95–102. doi:10.1002/9781118744994.ch18. ISBN 978-1-118-74499-4.
- ↑ E. Østreng; H. H. Sønsteby; S. Øien; O. Nilsen; H. Fjellvåg (2014). "Atomic layer deposition of sodium and potassium oxides: evaluation of precursors and deposition of thin films". Dalton Trans. 43 (44): 16666–16672. doi:10.1039/C4DT01930J. PMID 25265332.
- ↑ H. Nekola; F. Olbrich; U. Behrens (2002). "Kristall- und Molekülstrukturen von Lithium- und Natrium-tert-butoxid". Z. Anorg. Allg. Chem. 628 (9–10): 2067–2070. doi:10.1002/1521-3749(200209)628:9/10<2067::AID-ZAAC2067>3.0.CO;2-N.
 |