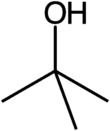
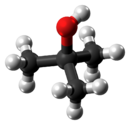
Chemistry:Tert-Butyl alcohol
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Methylpropan-2-ol | |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 906698 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| EC Number |
| ||
| 1833 | |||
| MeSH | tert-Butyl+Alcohol | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1120 | ||
| |||
| |||
| Properties | |||
| C4H10O | |||
| Molar mass | 74.123 g·mol−1 | ||
| Appearance | Colorless solid | ||
| Odor | Camphorous | ||
| Density | 0.775 g/mL | ||
| Melting point | 25 to 26 °C; 77 to 79 °F; 298 to 299 K | ||
| Boiling point | 82 to 83 °C; 179 to 181 °F; 355 to 356 K | ||
| miscible[2] | |||
| log P | 0.584 | ||
| Vapor pressure | 4.1 kPa (at 20 °C) | ||
| Acidity (pKa) | 16.54[3] | ||
| 5.742×10−5 cm3/mol | |||
Refractive index (nD)
|
1.387 | ||
| 1.31 D | |||
| Thermochemistry | |||
Heat capacity (C)
|
215.37 J K−1 mol−1 | ||
Std molar
entropy (S |
189.5 J K−1 mol−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
−360.04 to −358.36 kJ mol−1 | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−2.64479 to −2.64321 MJ mol−1 | ||
| Hazards | |||
| Safety data sheet | inchem.org | ||
| GHS pictograms |  
| ||
| GHS Signal word | DANGER | ||
| H225, H319, H332, H335 | |||
| P210, P261, P305+351+338 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 11 °C (52 °F; 284 K) | ||
| 480 °C (896 °F; 753 K) | |||
| Explosive limits | 2.4–8.0% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
3559 mg/kg (rabbit, oral) 3500 mg/kg (rat, oral)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 100 ppm (300 mg/m3)[1] | ||
REL (Recommended)
|
TWA 100 ppm (300 mg/m3) ST 150 ppm (450 mg/m3)[1] | ||
IDLH (Immediate danger)
|
1600 ppm[1] | ||
| Related compounds | |||
Related butanols
|
2-Butanol n-Butanol | ||
Related compounds
|
2-Methyl-2-butanol Trimethylsilanol | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
tert-Butyl alcohol is the simplest tertiary alcohol, with a formula of (CH3)3COH (sometimes represented as t-BuOH). Its isomers are 1-butanol, isobutanol, and 2-butanol. tert-Butyl alcohol is a colorless solid, which melts near room temperature and has a camphor-like odor. It is miscible with water, ethanol and diethyl ether.
Natural occurrence
tert-Butyl alcohol has been identified in beer and chickpeas.[5] It is also found in cassava,[6] which is used as a fermentation ingredient in certain alcoholic beverages.
Preparation
tert-Butyl alcohol is derived commercially from isobutane as a coproduct of propylene oxide production. It can also be produced by the catalytic hydration of isobutylene, or by a Grignard reaction between acetone and methylmagnesium chloride.
Purification cannot be performed by simple distillation due to formation of an azeotrope with water, although initial drying of the solvent containing large amounts of water is performed by adding benzene to form a tertiary azeotrope and distilling off the water. Smaller amounts of water are removed by drying with calcium oxide (CaO), potassium carbonate (K2CO3), calcium sulfate (CaSO4), or magnesium sulfate (MgSO4), followed by fractional distillation. Anhydrous tert-butyl alcohol is obtained by further refluxing and distilling from magnesium activated with iodine, or alkali metals such as sodium or potassium. Other methods include the use of 4 Å molecular sieves, aluminium tert-butylate, calcium hydride (CaH2), or fractional crystallization under inert atmosphere.[7]
Applications
tert-Butyl alcohol is used as a solvent, ethanol denaturant, paint remover ingredient, and gasoline octane booster and oxygenate. It is a chemical intermediate used to produce methyl tert-butyl ether (MTBE) and ethyl tert-butyl ether (ETBE) by reaction with methanol and ethanol, respectively, and tert-butyl hydroperoxide (TBHP) by reaction with hydrogen peroxide.
Reactions
Unlike other isomers of butanol, tert-butyl alcohol, as a tertiary alcohol, has no hydrogen atom next to hydroxy-group, which makes it resistant to oxidation to carbonyl compounds.
tert-Butyl alcohol is deprotonated with a strong base to give the alkoxide. Particularly common is potassium tert-butoxide, which is prepared by treating tert-butanol with potassium metal.[8]
- K + t-BuOH → t-BuO−K+ + 1/2 H2
The tert-butoxide is a strong, non-nucleophilic base in organic chemistry. It readily abstracts acidic protons from substrates, but its steric bulk inhibits the group from participating in nucleophilic substitution, such as in a Williamson ether synthesis or an SN2 reaction.
tert-Butyl alcohol reacts with hydrogen chloride to form tert-butyl chloride.
O-Chlorination of tert-butyl alcohol with hypochlorous acid to give tert-butyl hypochlorite:[9]
- (CH3)3COH + HOCl → (CH3)3COCl + H2O
Pharmacology and toxicology
There is limited data on the pharmacology and toxicology of tert-butanol in humans and other animals.[10] Human exposure may occur due to fuel oxygenate metabolism. Tert-butanol is poorly absorbed through skin but rapidly absorbed if inhaled or ingested. Tert-butanol is irritating to skin or eyes. Toxicity of single doses is usually low but high doses of tert-Butyl alcohol can produce a sedative or anesthetic effect.
References
- ↑ 1.0 1.1 1.2 1.3 NIOSH Pocket Guide to Chemical Hazards. "#0078". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0078.html.
- ↑ "ICSC 0114 – tert-Butanol". http://www.inchem.org/documents/icsc/icsc/eics0114.htm.
- ↑ Reeve, W.; Erikson, C. M.; Aluotto, P. F. (1979). "tert-Butyl alcohol". Can. J. Chem. 57: 2747. doi:10.1139/v79-444.
- ↑ "tert-Butyl alcohol". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/idlh/75650.html.
- ↑ "t-Butyl Alcohol". National Institute for Health. http://toxnet.nlm.nih.gov/cgi-bin/sis/search/a?dbs+hsdb:@term+@DOCNO+50.
- ↑ "Archived copy". http://www.sc.mahidol.ac.th/scbc/bc_internet/publication/696.pdf.
- ↑ Perrin, D. D.; Armarego, W. L. F. (1988). Purification of Laboratory Chemicals (3rd ed.). Pergamon Press. ISBN 9780080347141. https://archive.org/details/purificationofla0000perr_n7w5.
- ↑ Johnson, W. S.; Schneider, W. P. (1950). "β-Carbethoxy-γ,γ-diphenylvinylacetic acid". Organic Syntheses 30: 18. doi:10.15227/orgsyn.030.0018.
- ↑ Mintz, H. M.; Walling, C. (1969). "t-Butyl Hypochlorite". Org. Synth. 49: 9. doi:10.15227/orgsyn.049.0009.
- ↑ Douglas McGregor (2010). "Tertiary-Butanol: a toxicological review". Critical Reviews in Toxicology 40 (8): 697–727. doi:10.3109/10408444.2010.494249. PMID 20722584.
External links
- International Chemical Safety Card 0114
- NIOSH Pocket Guide to Chemical Hazards. "#0078". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0078.html.
- IPCS Environmental Health Criteria 65: Butanols: four isomers
- IPCS Health and Safety Guide 7: tert-Butanol
 |