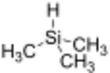
Chemistry:Trimethylsilane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Trimethylsilane | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| |||
| Properties | |||
| C3H10Si | |||
| Molar mass | 74.198 g·mol−1 | ||
| Density | 0.638 g cm−3 | ||
| Melting point | −135.9 °C (−212.6 °F; 137.2 K) | ||
| Boiling point | 6.7 °C (44.1 °F; 279.8 K) | ||
| Hazards | |||
| GHS pictograms |   
| ||
| GHS Signal word | Danger | ||
| H220, H224, H280, H315, H319, H335 | |||
| P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P302+352, P303+361+353, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P370+378, P377, P381, P403, P403+233, P403+235 | |||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Trimethylsilane is the organosilicon compound with the formula (CH3)3SiH. It is a trialkylsilane. The Si-H bond is reactive. It is less commonly used as a reagent than the related triethylsilane, which is a liquid at room temperature.
Trimethylsilane is used in the semiconductor industry as precursor to deposit dielectrics and barrier layers via plasma-enhanced chemical vapor deposition (PE-CVD).[1] It is also used a source gas to deposit TiSiCN hard coatings via plasma-enhanced magnetron sputtering (PEMS). It has also been used to deposit silicon carbide hard coatings via low-pressure chemical vapor deposition (LP-CVD) at relatively low temperatures under 1000 °C. It is an expensive gas but safer to use than silane (SiH4); and produces properties in the coatings that cannot be undertaken by multiple source gases containing silicon and carbon.
See also
- Dimethylsilane
- Trimethylsilyl functional group
References
- ↑ Chen, Sheng-Wen; Wang, Yu-Sheng; Hu, Shao-Yu; Lee, Wen-Hsi; Chi, Chieh-Cheng; Wang, Ying-Lang (2012). "A Study of Trimethylsilane (3MS) and Tetramethylsilane (4MS) Based α-SiCN:H/α-SiCO:H Diffusion Barrier Films". Materials 5 (3): 377–384. doi:10.3390/ma5030377. PMID 28817052. Bibcode: 2012Mate....5..377C.
 |