Medicine:Acute kidney injury
| Acute kidney injury | |
|---|---|
| Other names | Acute renal failure (ARF), acute kidney failure (AKF) |
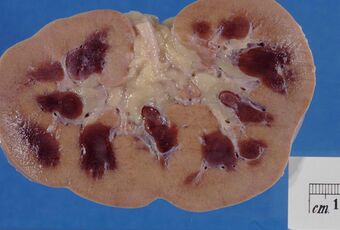 | |
| Pathologic kidney specimen showing marked pallor of the cortex, contrasting to the darker areas of surviving medullary tissue. The patient died with acute kidney injury. | |
| Specialty | Nephrology, Urology |
Acute kidney injury (AKI), previously called acute renal failure (ARF),[1][2] is a sudden decrease in kidney function that develops within 7 days,[3] as shown by an increase in serum creatinine or a decrease in urine output, or both.[4]
Causes of AKI are classified as either prerenal (due to decreased blood flow to the kidney), intrinsic renal (due to damage to the kidney itself), or postrenal (due to blockage of urine flow).[5] Prerenal causes of AKI include sepsis, dehydration, excessive blood loss, cardiogenic shock, heart failure, cirrhosis, and certain medications like ACE inhibitors or NSAIDs.[5] Intrinsic renal causes of AKI include glomerulonephritis, lupus nephritis, acute tubular necrosis, certain antibiotics, and chemotherapeutic agents.[5] Postrenal causes of AKI include kidney stones, bladder cancer, neurogenic bladder, enlargement of the prostate, narrowing of the urethra, and certain medications like anticholinergics.[5]
The diagnosis of AKI is made based on a person's signs and symptoms, along with lab tests for serum creatinine and measurement of urine output. Other tests include urine microscopy and urine electrolytes. Renal ultrasound can be obtained when a postrenal cause is suspected. A kidney biopsy may be obtained when intrinsic renal AKI is suspected and the cause is unclear.[5]
AKI is seen in 10-15% of people admitted to the hospital and in more than 50% of people admitted to the intensive care unit (ICU).[4] AKI may lead to a number of complications, including metabolic acidosis, high potassium levels, uremia, changes in body fluid balance, effects on other organ systems, and death. People who have experienced AKI are at increased risk of developing chronic kidney disease in the future.[4] Management includes treatment of the underlying cause and supportive care, such as renal replacement therapy.
Signs and symptoms
The clinical presentation is often dominated by the underlying cause. The various symptoms of acute kidney injury result from the various disturbances of kidney function that are associated with the disease. Accumulation of urea and other nitrogen-containing substances in the bloodstream lead to a number of symptoms, such as fatigue, loss of appetite, headache, nausea, and vomiting.[6] Marked increases in the potassium level can lead to abnormal heart rhythms, which can be severe and life-threatening.[7] Fluid balance is frequently affected, though blood pressure can be high, low, or normal.[8]
Pain in the flanks may be encountered in some conditions (such as clotting of the kidneys' blood vessels or inflammation of the kidney). This is the result of stretching of the fibrous tissue capsule surrounding the kidney.[9] If the kidney injury is the result of dehydration, there may be thirst as well as evidence of fluid depletion on physical examination.[9] Physical examination may also provide other clues as to the underlying cause of the kidney problem, such as a rash in interstitial nephritis (or vasculitis) and a palpable bladder in obstructive nephropathy.[9]
Causes
Prerenal
Prerenal causes of AKI ("pre-renal azotemia") are those that decrease effective blood flow to the kidney and cause a decrease in the glomerular filtration rate (GFR). Both kidneys need to be affected as one kidney is still more than adequate for normal kidney function. Notable causes of prerenal AKI include low blood volume (e.g., dehydration), low blood pressure, heart failure (leading to cardiorenal syndrome), hepatorenal syndrome in the context of liver cirrhosis, and local changes to the blood vessels supplying the kidney. The latter include renal artery stenosis, or the narrowing of the renal artery which supplies the kidney with blood, and renal vein thrombosis, which is the formation of a blood clot in the renal vein that drains blood from the kidney.[citation needed]
Intrinsic or renal
Intrinsic AKI refers to disease processes which directly damage the kidney itself. Intrinsic AKI can be due to one or more of the kidney's structures including the glomeruli, kidney tubules, or the interstitium. Common causes of each are glomerulonephritis, acute tubular necrosis (ATN), and acute interstitial nephritis (AIN), respectively. Other causes of intrinsic AKI are rhabdomyolysis and tumor lysis syndrome.[10] Certain medication classes such as calcineurin inhibitors (e.g., tacrolimus) can also directly damage the tubular cells of the kidney and result in a form of intrinsic AKI.[11]
Postrenal
Postrenal AKI refers to acute kidney injury caused by disease states downstream of the kidney and most often occurs as a consequence of urinary tract obstruction. This may be related to benign prostatic hyperplasia, kidney stones, obstructed urinary catheter, bladder stones, or cancer of the bladder, ureters, or prostate.
Diagnosis
Definition
Introduced by the KDIGO in 2012,[12] specific criteria exist for the diagnosis of AKI.
AKI can be diagnosed if any one of the following is present:
- Increase in SCr by ≥0.3 mg/dl (≥26.5 μmol/L) within 48 hours; or
- Increase in SCr to ≥1.5 times baseline, which has occurred within the prior 7 days; or
- Urine volume < 0.5 mL/kg/h for 6 hours.
Staging
The RIFLE criteria, proposed by the Acute Dialysis Quality Initiative (ADQI) group, aid in assessment of the severity of a person's acute kidney injury. The acronym RIFLE is used to define the spectrum of progressive kidney injury seen in AKI:[13][14]
- Risk: 1.5-fold increase in the serum creatinine, or glomerular filtration rate (GFR) decrease by 25 percent, or urine output <0.5 mL/kg per hour for six hours.
- Injury: Two-fold increase in the serum creatinine, or GFR decrease by 50 percent, or urine output <0.5 mL/kg per hour for 12 hours.
- Failure: Three-fold increase in the serum creatinine, or GFR decrease by 75 percent, or urine output of <0.3 mL/kg per hour for 24 hours, or no urine output (anuria) for 12 hours.
- Loss: Complete loss of kidney function (e.g., need for renal replacement therapy) for more than four weeks.
- End-stage kidney disease: Complete loss of kidney function (e.g., need for renal replacement therapy) for more than three months.
Evaluation
The deterioration of kidney function may be signaled by a measurable decrease in urine output. Often, it is diagnosed on the basis of blood tests for substances normally eliminated by the kidney: urea and creatinine. Additionally, the ratio of BUN to creatinine is used to evaluate kidney injury. Both tests have their disadvantages. For instance, it takes about 24 hours for the creatinine level to rise, even if both kidneys have ceased to function. A number of alternative markers have been proposed (such as NGAL, KIM-1, IL18 and cystatin C), but none of them are established enough as of 2018 to replace creatinine as a marker of kidney function.[15]
Once the diagnosis of AKI is made, further testing is often required to determine the underlying cause. It is useful to perform a bladder scan or a post void residual to rule out urinary retention. In post void residual, a catheter is inserted into the urinary tract immediately after urinating to measure fluid still in the bladder. 50–100 mL suggests neurogenic bladder dysfunction.[citation needed]
These may include urine sediment analysis, renal ultrasound and/or kidney biopsy. Indications for kidney biopsy in the setting of AKI include the following:[16]
- Unexplained AKI, in a patient with two non-obstructed normal sized kidneys.
- AKI in the presence of the nephritic syndrome.
- Systemic disease associated with AKI.
- Kidney transplant dysfunction.
In medical imaging, the acute changes in the kidney are often examined with renal ultrasonography as the first-line modality, where CT scan and magnetic resonance imaging (MRI) are used for the follow-up examinations and when US fails to demonstrate abnormalities. In evaluation of the acute changes in the kidney, the echogenicity of the renal structures, the delineation of the kidney, the renal vascularity, kidney size and focal abnormalities are observed.[17] CT is preferred in renal traumas, but US is used for follow-up, especially in the patients suspected for the formation of urinomas. A CT scan of the abdomen will also demonstrate bladder distension or hydronephrosis.[18]
Renal ultrasonograph of acute pyelonephritis with increased cortical echogenicity and blurred delineation of the upper pole.[17]
Renal ultrasonograph in renal failure after surgery with increased cortical echogenicity and kidney size. Biopsy showed acute tubular necrosis.[17]
Renal ultrasonograph in renal trauma with laceration of the lower pole and subcapsular fluid collection below the kidney.[17]
Classification
Acute kidney injury is diagnosed on the basis of clinical history and laboratory data. A diagnosis is made when there is a rapid reduction in kidney function, as measured by serum creatinine, or based on a rapid reduction in urine output, termed oliguria (less than 400 mLs of urine per 24 hours).[citation needed]
| Type | UOsm | UNa | FeNa | BUN/Cr |
|---|---|---|---|---|
| Prerenal | >500 | <10 | <1% | >20[19] |
| Intrinsic | <350 | >20 | >2% | <10-15[19] |
| Postrenal | <350 | >40 | >4% | >20[19] |
AKI can be caused by systemic disease (such as a manifestation of an autoimmune disease, e.g., lupus nephritis), crush injury, contrast agents, some antibiotics, and more. AKI often occurs due to multiple processes. The most common cause is dehydration and sepsis combined with nephrotoxic drugs, especially following surgery or contrast agents.[citation needed]
The causes of acute kidney injury are commonly categorized into prerenal, intrinsic, and postrenal.
Acute kidney injury occurs in up to 30% of patients following cardiac surgery.[20] Mortality increases by 60-80% in post-cardiopulmonary bypass patients who go on to require renal replacement therapy. Preoperative creatinine greater than 1.2 mg/dL, combined valve and bypass procedures, emergency surgery, and preoperative intra-aortic balloon pump are risk factors most strongly correlated with post-cardiopulmonary bypass acute kidney injury. Other well-known minor risk factors include female gender, congestive heart failure, chronic obstructive pulmonary disease, insulin-requiring diabetes, and depressed left ventricular ejection fraction.[20] Volatile anesthetic agents have been shown to increase renal sympathetic nerve activity (RSNA), which causes retention of salts and water, diminished renal blood flow (RBF) and an increase in serum renin levels, but not in antidiuretic hormone (ADH).[21]
Treatment
The management of AKI hinges on identification and treatment of the underlying cause. The main objectives of initial management are to prevent cardiovascular collapse and death and to call for specialist advice from a nephrologist. In addition to treatment of the underlying disorder, management of AKI routinely includes the avoidance of substances that are toxic to the kidneys, called nephrotoxins. These include NSAIDs such as ibuprofen or naproxen, iodinated contrasts such as those used for CT scans, many antibiotics such as gentamicin, and a range of other substances.[22]
Monitoring of kidney function, by serial serum creatinine measurements and monitoring of urine output, is routinely performed. In the hospital, insertion of a urinary catheter helps monitor urine output and relieves possible bladder outlet obstruction, such as with an enlarged prostate.[citation needed]
Prerenal
In prerenal AKI without fluid overload, administration of intravenous fluids is typically the first step to improving kidney function. Volume status may be monitored with the use of a central venous catheter to avoid over- or under-replacement of fluid.[citation needed]
If low blood pressure persists despite providing a person with adequate amounts of intravenous fluid, medications that increase blood pressure (vasopressors) such as norepinephrine, and in certain circumstances medications that improve the heart's ability to pump (known as inotropes) such as dobutamine may be given to improve blood flow to the kidney. While a useful vasopressor, there is no evidence to suggest that dopamine is of any specific benefit and may in fact be harmful.[23]
Intrinsic
The myriad causes of intrinsic AKI require specific therapies. For example, intrinsic AKI due to vasculitis or glomerulonephritis may respond to steroid medication, cyclophosphamide, and (in some cases) plasma exchange. Toxin-induced prerenal AKI often responds to discontinuation of the offending agent, such as ACE inhibitors, ARB antagonists, aminoglycosides, penicillins, NSAIDs, or paracetamol.[9]
The use of diuretics such as furosemide, is widespread and sometimes convenient in improving fluid overload. It is not associated with higher mortality (risk of death),[24] nor with any reduced mortality or length of intensive care unit or hospital stay.[25]
Postrenal
If the cause is obstruction of the urinary tract, relief of the obstruction (with a nephrostomy or urinary catheter) may be necessary.[citation needed]
Renal replacement therapy
Renal replacement therapy, such as with hemodialysis, may be instituted in some cases of AKI. Renal replacement therapy can be applied intermittently (IRRT) and continuously (CRRT). Study results regarding differences in outcomes between IRRT and CRRT are inconsistent. A systematic review of the literature in 2008 demonstrated no difference in outcomes between the use of intermittent hemodialysis and continuous venovenous hemofiltration (CVVH) (a type of continuous hemodialysis).[26] Among critically ill patients, intensive renal replacement therapy with CVVH does not appear to improve outcomes compared to less intensive intermittent hemodialysis.[22][27] However, other clinical and health economic studies demonstrated that, initiation of CRRT is associated with a lower likelihood of chronic dialysis and was cost-effective compared with IRRT in patients with acute kidney injury.[28][29][30]
Complications
Metabolic acidosis, hyperkalemia, and pulmonary edema may require medical treatment with sodium bicarbonate, antihyperkalemic measures, and diuretics.[31]
Lack of improvement with fluid resuscitation, therapy-resistant hyperkalemia, metabolic acidosis, or fluid overload may necessitate artificial support in the form of dialysis or hemofiltration.[7] However, oliguria during anesthesia may predict AKI,[32][33] but the effect of a fluid load is highly variable. Striving toward a predefined urine output target to prevent AKI is futile.[21][34][35]
Early recovery of AKI
AKI recovery can be classified into three stages 1–3 on the basis of the inverse of the AKI KDIGO serum creatinine criteria.[36]
Prognosis
Mortality
Mortality after AKI remains high. AKI has a death rate as high as 20%, which may reach up to 50% in the intensive care unit (ICU). Each year, around two million people die of AKI worldwide.[37]
AKI develops in 5% to 30% of patients who undergo cardiothoracic surgery, depending on the definition used for AKI.[38] If AKI develops after major abdominal surgery (13.4% of all people who have undergone major abdominal surgery) the risk of death is markedly increased (over 12-fold).[39]
Kidney function
Depending on the cause, a proportion of patients (5–10%) will never regain full kidney function, thus entering end-stage kidney failure and requiring lifelong dialysis or a kidney transplant. Patients with AKI are more likely to die prematurely after being discharged from hospital, even if their kidney function has recovered.[2]
The risk of developing chronic kidney disease is increased (8.8-fold).[40]
Epidemiology
New cases of AKI are unusual but not rare, affecting approximately 0.1% of the UK population per year (2000 ppm/year), 20x incidence of new ESKD (end-stage kidney disease). AKI requiring dialysis (10% of these) is rare (200 ppm/year), 2x incidence of new ESKD.[41]
There is an increased incidence of AKI in agricultural workers because of occupational hazards such as dehydration and heat illness.[42] No other traditional risk factors, including age, BMI, diabetes, or hypertension, were associated with incident AKI.
Acute kidney injury is common among hospitalized patients. It affects some 3–7% of patients admitted to the hospital and approximately 25–30% of patients in the intensive care unit.[43]
Acute kidney injury was one of the most expensive conditions seen in U.S. hospitals in 2011, with an aggregated cost of nearly $4.7 billion for approximately 498,000 hospital stays.[44] This was a 346% increase in hospitalizations from 1997, when there were 98,000 acute kidney injury stays.[45] According to a review article of 2015, there has been an increase in cases of acute kidney injury in the last 20 years which cannot be explained solely by changes to the manner of reporting.[46]
History
Before the advancement of modern medicine, acute kidney injury was referred to as uremic poisoning while uremia was contamination of the blood with urine. Starting around 1847, uremia came to be used for reduced urine output, a condition now called oliguria, which was thought to be caused by the urine's mixing with the blood instead of being voided through the urethra.[47]
Acute kidney injury due to acute tubular necrosis (ATN) was recognized in the 1940s in United Kingdom , where crush injury victims during the London Blitz developed patchy necrosis of kidney tubules, leading to a sudden decrease in kidney function.[48] During the Korean and Vietnam wars, the incidence of AKI decreased due to better acute management and administration of intravenous fluids.[49]
See also
- BUN-to-creatinine ratio
- Chronic kidney disease
- Dialysis
- Genitourinary tract injury
- Kidney failure
- Rhabdomyolysis
- Contrast-induced nephropathy
- Ischemia-reperfusion injury of the appendicular musculoskeletal system
- Kidney Ischemia
References
- ↑ "ARF, ATN or AKI? It's now acute kidney injury". Anaesthesia and Intensive Care 35 (6): 843–44. December 2007. doi:10.1177/0310057X0703500601. PMID 18084974.
- ↑ 2.0 2.1 Dan Longo; Anthony Fauci; Dennis Kasper; Stephen Hauser; J. Jameson; Joseph Loscalzo (July 21, 2011). Harrison's Principles of Internal Medicine, 18 edition. McGraw-Hill Professional.
- ↑ "Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury". Critical Care 11 (2): R31. 2007. doi:10.1186/cc5713. PMID 17331245.
- ↑ 4.0 4.1 4.2 "Acute kidney injury". The Lancet 394 (10212): 1949–64. 23 November 2019. doi:10.1016/S0140-6736(19)32563-2. PMID 31777389.
- ↑ 5.0 5.1 5.2 5.3 5.4 "Acute Kidney Injury: Diagnosis and Management". American Family Physician 100 (11): 687–694. 1 December 2019. PMID 31790176.
- ↑ "Chronic renal failure". Harrison's Principles of Internal Medicine (16th ed.). New York, NY: McGraw-Hill. 2005. pp. 1653–63. ISBN 978-0-07-139140-5. https://archive.org/details/harrisonsprincip00kasp.
- ↑ 7.0 7.1 Weisberg LS (December 2008). "Management of severe hyperkalemia". Crit. Care Med. 36 (12): 3246–51. doi:10.1097/CCM.0b013e31818f222b. PMID 18936701.
- ↑ Tierney, Lawrence M.; Stephen J. McPhee; Maxine A. Papadakis (2004). "22". CURRENT Medical Diagnosis and Treatment 2005 (44th ed.). McGraw-Hill. p. 871. ISBN 978-0-07-143692-2. https://archive.org/details/currentmedicaldi0000unse.
- ↑ 9.0 9.1 9.2 9.3 "Chronic renal failure". Harrison's Principles of Internal Medicine (16th ed.). New York, NY: McGraw-Hill. 2005. pp. 1644–53. ISBN 978-0-07-139140-5. https://archive.org/details/harrisonsprincip00kasp.
- ↑ Jim Cassidy; Donald Bissett; Roy A. J. Spence; Miranda Payne (1 January 2010). Oxford Handbook of Oncology. Oxford University Press. p. 706. ISBN 978-0-19-956313-5. https://books.google.com/books?id=WKoQW4G0g14C&pg=PA706.
- ↑ Shirali, Anushree; Pazhayattil, George Sunny (December 2014). "Drug-induced impairment of renal function". International Journal of Nephrology and Renovascular Disease 7: 457–468. doi:10.2147/ijnrd.s39747. ISSN 1178-7058. PMID 25540591.
- ↑ "Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomes 2012 clinical practice guideline". Ann Intern Med 158 (11): 825–30. June 2013. doi:10.7326/0003-4819-158-11-201306040-00007. PMID 23732715.
- ↑ "Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group". Crit Care 8 (4): R204–12. 2004. doi:10.1186/cc2872. PMID 15312219.
- ↑ "Acute renal failure". Lancet 365 (9457): 417–30. 2005. doi:10.1016/S0140-6736(05)17831-3. PMID 15680458.
- ↑ Hall, P (2018). "The future for diagnostic tests of acute kidney injury in critical care: evidence synthesis, care pathway analysis and research prioritisation.". Health Technology Assessment 22 (32): 1–274. doi:10.3310/hta22320. PMID 29862965. PMC 6004543. https://doi.org/10.3310%2Fhta22320.
- ↑ Current Medical Diagnosis and Treatment. McGraw-Hill Professional. 2008. ISBN 978-0-07-159124-9.
- ↑ 17.0 17.1 17.2 17.3 Content initially copied from: Hansen, Kristoffer; Nielsen, Michael; Ewertsen, Caroline (2015). "Ultrasonography of the Kidney: A Pictorial Review". Diagnostics 6 (1): 2. doi:10.3390/diagnostics6010002. ISSN 2075-4418. PMID 26838799. (CC-BY 4.0)
- ↑ Silverman, Stuart G.; Leyendecker, John R.; Amis, E. Stephen (February 2009). "What Is the Current Role of CT Urography and MR Urography in the Evaluation of the Urinary Tract?" (in en). Radiology 250 (2): 309–323. doi:10.1148/radiol.2502080534. ISSN 0033-8419. PMID 19188307. http://pubs.rsna.org/doi/10.1148/radiol.2502080534.
- ↑ 19.0 19.1 19.2 Goldman-Cecil medicine (25th ed.). Philadelphia, PA. 15 April 2015. pp. 781. ISBN 978-1455750177. OCLC 899727756.
- ↑ 20.0 20.1 "AKI associated with cardiac surgery". Clin J Am Soc Nephrol 10 (3): 500–14. March 2015. doi:10.2215/CJN.07830814. PMID 25376763.
- ↑ 21.0 21.1 Taavo, Micael; Rundgren, Mats; Frykholm, Peter; Larsson, Anders; Franzén, Stephanie; Vargmar, Karin; Valarcher, Jean F; DiBona, Gerald F et al. (2021-10-14). "Role of Renal Sympathetic Nerve Activity in Volatile Anesthesia's Effect on Renal Excretory Function" (in en). Function 2 (6): zqab042. doi:10.1093/function/zqab042. ISSN 2633-8823. PMID 35330795. PMC 8788708. https://academic.oup.com/function/article/doi/10.1093/function/zqab042/6355435.
- ↑ 22.0 22.1 "Intensity of renal support in critically ill patients with acute kidney injury". The New England Journal of Medicine 359 (1): 7–20. July 2008. doi:10.1056/NEJMoa0802639. PMID 18492867.
- ↑ "Bad medicine: low-dose dopamine in the ICU". Chest 123 (4): 1266–75. 2003. doi:10.1378/chest.123.4.1266. PMID 12684320.
- ↑ "Diuretics and mortality in acute renal failure". Crit. Care Med. 32 (8): 1669–77. 2004. doi:10.1097/01.CCM.0000132892.51063.2F. PMID 15286542.
- ↑ "The use of loop diuretics in acute renal failure in critically ill patients to reduce mortality, maintain renal function, or avoid the requirements for renal support". Emergency Medicine Journal 23 (7): 569–70. 2006. doi:10.1136/emj.2006.038513. PMID 16794108.
- ↑ "Renal replacement therapy in patients with acute renal failure: a systematic review". JAMA: The Journal of the American Medical Association 299 (7): 793–805. February 2008. doi:10.1001/jama.299.7.793. PMID 18285591. http://jama.ama-assn.org/cgi/pmidlookup?view=long&pmid=18285591.
- ↑ "Intensity of continuous renal-replacement therapy in critically ill patients". The New England Journal of Medicine 361 (17): 1627–38. October 2009. doi:10.1056/NEJMoa0902413. PMID 19846848. http://espace.library.uq.edu.au/view/UQ:694105/UQ694105_OA.pdf.
- ↑ Schoenfelder, T; Chen, X; Bless, HH (March 2017). "Effects of continuous and intermittent renal replacement therapies among adult patients with acute kidney injury". GMS Health Technol Assess 13 (Doc01 (20170301)). http://www.egms.de/static/en/journals/hta/2017-13/hta000127.shtml. Retrieved 8 August 2017.
- ↑ Schneider, AG; Bellomo, R; Bagshaw, SM; Glassford, NJ; Lo, S; Jun, M; Cass, A; Gallagher, M (June 2013). "Choice of renal replacement therapy modality and dialysis dependence after acute kidney injury: a systematic review and meta-analysis.". Intensive Care Medicine 39 (6): 987–97. doi:10.1007/s00134-013-2864-5. PMID 23443311. http://minerva-access.unimelb.edu.au/bitstream/11343/218110/1/Choice%20of%20renal%20replacement%20therapy%20modality%20and%20dialysis%20dependence%20after%20acute%20kidney%20injury.pdf.
- ↑ Singh, Ambrish; Hussain, Salman; Kher, Vijay; Palmer, Andrew J.; Jose, Matthew; Antony, Benny (2022). "A systematic review of cost-effectiveness analyses of continuous versus intermittent renal replacement therapy in acute kidney injury". Expert Review of Pharmacoeconomics & Outcomes Research 22 (1): 27–35. doi:10.1080/14737167.2021.1916471. PMID 33934689. https://doi.org/10.1080/14737167.2021.1916471.
- ↑ Mushiyakh, Yelena; Dangaria, Harsh; Qavi, Shahbaz; Ali, Noorjahan; Pannone, John; Tompkins, David (2011). "Treatment and pathogenesis of acute hyperkalemia". Journal of Community Hospital Internal Medicine Perspectives 1 (4): 7372. doi:10.3402/jchimp.v1i4.7372. ISSN 2000-9666. PMID 23882341.
- ↑ Myles, Paul S.; McIlroy, David R.; Bellomo, Rinaldo; Wallace, Sophie (June 2019). "Importance of intraoperative oliguria during major abdominal surgery: findings of the Restrictive versus Liberal Fluid Therapy in Major Abdominal Surgery trial" (in en). British Journal of Anaesthesia 122 (6): 726–733. doi:10.1016/j.bja.2019.01.010. PMID 30916001.
- ↑ Mizota, T.; Yamamoto, Y.; Hamada, M.; Matsukawa, S.; Shimizu, S.; Kai, S. (December 2017). "Intraoperative oliguria predicts acute kidney injury after major abdominal surgery" (in en). British Journal of Anaesthesia 119 (6): 1127–1134. doi:10.1093/bja/aex255. PMID 29136086. https://linkinghub.elsevier.com/retrieve/pii/S0007091217541409.
- ↑ Egal, Mohamud; Erler, Nicole S.; de Geus, Hilde R. H.; van Bommel, Jasper; Groeneveld, A. B. Johan (January 2016). "Targeting Oliguria Reversal in Goal-Directed Hemodynamic Management Does Not Reduce Renal Dysfunction in Perioperative and Critically Ill Patients: A Systematic Review and Meta-Analysis" (in en). Anesthesia & Analgesia 122 (1): 173–185. doi:10.1213/ANE.0000000000001027. ISSN 0003-2999. PMID 26505575. https://journals.lww.com/00000539-201601000-00027.
- ↑ Egal, Mohamud; de Geus, Hilde R.H.; van Bommel, Jasper; Groeneveld, A.B. Johan (June 2016). "Targeting oliguria reversal in perioperative restrictive fluid management does not influence the occurrence of renal dysfunction: A systematic review and meta-analysis" (in en). European Journal of Anaesthesiology 33 (6): 425–435. doi:10.1097/EJA.0000000000000416. ISSN 0265-0215. PMID 26840829. http://journals.lww.com/00003643-201606000-00006.
- ↑ Duff, Stephen; Murray, Patrick T. (7 September 2020). "Defining Early Recovery of Acute Kidney Injury" (in en). Clinical Journal of the American Society of Nephrology 15 (9): 1358–1360. doi:10.2215/CJN.13381019. ISSN 1555-9041. PMID 32238366.
- ↑ Duan, Shao-Bin; Liu, Fu-You; Liu, Hong; Tang, Mi-Mi; Li, Xu-Wei; Cheng, Wei; Xu, Jun; Yang, Yuan et al. (2017-08-11). "A new scoring model for the prediction of mortality in patients with acute kidney injury" (in en). Scientific Reports 7 (1): 7862. doi:10.1038/s41598-017-08440-w. ISSN 2045-2322. PMID 28801674. Bibcode: 2017NatSR...7.7862L.
- ↑ Hobson Charles E.; Yavas Sinan; Segal Mark S.; Schold Jesse D.; Tribble Curtis G.; Layon A. Joseph; Bihorac Azra (2009-05-12). "Acute Kidney Injury Is Associated With Increased Long-Term Mortality After Cardiothoracic Surgery". Circulation 119 (18): 2444–2453. doi:10.1161/CIRCULATIONAHA.108.800011. PMID 19398670.
- ↑ O'Connor, M. E.; Kirwan, C. J.; Pearse, R. M.; Prowle, J. R. (24 November 2015). "Incidence and associations of acute kidney injury after major abdominal surgery". Intensive Care Medicine 42 (4): 521–30. doi:10.1007/s00134-015-4157-7. PMID 26602784. http://qmro.qmul.ac.uk/xmlui/handle/123456789/10969.
- ↑ Coca, SG; Singanamala, S; Parikh, CR (March 2012). "Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis.". Kidney International 81 (5): 442–48. doi:10.1038/ki.2011.379. PMID 22113526.
- ↑ "Renal Medicine: Acute Kidney Injury (AKI)". Renalmed.co.uk. 2012-05-23. http://www.renalmed.co.uk/database/acute-kidney-injury-aki.
- ↑ "Cumulative Incidence of Acute Kidney Injury in California's Agricultural Workers". J Occup Environ Med 58 (4): 391–7. April 2016. doi:10.1097/JOM.0000000000000668. PMID 27058480.
- ↑ Brenner and Rector's The Kidney. Philadelphia: Saunders. 2007. ISBN 978-1-4160-3110-9.
- ↑ Torio CM, Andrews RM. National Inpatient Hospital Costs: The Most Expensive Conditions by Payer, 2011. HCUP Statistical Brief #160. Agency for Healthcare Research and Quality, Rockville, MD. August 2013. "Statistical Brief #160". http://hcup-us.ahrq.gov/reports/statbriefs/sb160.jsp.
- ↑ Pfuntner A., Wier L.M., Stocks C. Most Frequent Conditions in U.S. Hospitals, 2011. HCUP Statistical Brief #162. September 2013. Agency for Healthcare Research and Quality, Rockville, MD. "Most Frequent Conditions in U.S. Hospitals, 2011 #162". http://www.hcup-us.ahrq.gov/reports/statbriefs/sb162.jsp.
- ↑ "The growth of acute kidney injury: a rising tide or just closer attention to detail?". Kidney International 87 (1): 46–61. 2015. doi:10.1038/ki.2014.293. PMID 25229340.
- ↑ "oliguria | Origin and meaning of oliguria by Online Etymology Dictionary" (in en). https://www.etymonline.com/word/oliguria.
- ↑ "Crush injuries with impairment of renal function.". Br Med J 1 (4185): 427–32. 1941. doi:10.1136/bmj.1.4185.427. PMID 20783577.
- ↑ "Acute renal failure: definitions, diagnosis, pathogenesis, and therapy". J. Clin. Invest. 114 (1): 5–14. 2004. doi:10.1172/JCI22353. PMID 15232604.
External links
| Classification | |
|---|---|
| External resources |
 |





