Medicine:Intranasal drug delivery
Intranasal drug delivery occurs when particles are inhaled into the nasal cavity and transported directly into the nervous system. Though pharmaceuticals can be injected into the nose, some concerns include injuries, infection, and safe disposal. Studies demonstrate improved patient compliance with inhalation. Treating brain diseases has been a challenge due to the blood brain barrier. Previous studies evaluated the efficacy of delivery therapeutics through intranasal route for brain diseases and mental health conditions. Intranasal administration is a potential route associated with high drug transfer from nose to brain and drug bioavailability.[1]
History of drug delivery
Drug delivery is a process of administering therapeutics to treat human diseases. The first drug delivery system often dates back to the 1950s, where Smith Kline & French Laboratories introduced the Spansule technology.[2] Between 1950s and 1980's, there were four drug release systems developed for oral and transdermal applications: dissolution, diffusion, osmosis, and ion-exchange controlled release.[3] Later in the 1980s, the Lupron Depot technology further advanced the field by offering zero-order and long-term release systems. Over the past few decades, the field has seen breathtaking advancements with improved drug formulations, release mechanisms, delivery materials, and administration routes. The intranasal route gained interest towards the end of the 20th century with treating cardiovascular and respiratory diseases. During the late 1980s, William Frey II studied the intranasal route for treating brain diseases. Ever since, it has become a potential route for nose-to-brain delivery.
Anatomy
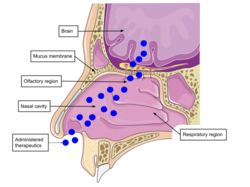
Intranasal delivery pathway
The nasal cavity is highly vascularized which enables efficient transfer of molecules directly to the nervous system. When utilizing other routes of administration to target the brain, drug adsorption is low due to issues with systemic circulation effects and blood brain barrier. Drug delivery through nasal cavity increases drug bioavailability and lowers risk of systemic exposure. Nasal cavity is a slightly acidic environment with enzymes which can alter degradation of drug delivery systems. The delivery systems between neutral to acidic pH is ideal for high drug bioavailability following intranasal administration. The respiratory region of the cavity is the most vascularized with large surface area. These properties make it the primary site for drug transfer and absorption for systemic circulation. Targeting the olfactory region increases efficiency of nose-to-brain drug delivery. The olfactory region consists of the mucosal membrane consisting of the olfactory nerve connected to olfactory bulb. When particles are transmitted via the nerve fibers to olfactory bulb, they have direct access to the brain. Intranasal particles have to pass through the mucus layer, the first barrier, to then travel from nasal epithelium to brain. The mucosal membrane is negatively charged and contains hydrophobic domains. These properties affect drug diffusion. Overall, this delivery route is a potential route to treat brain diseases and mental health conditions.
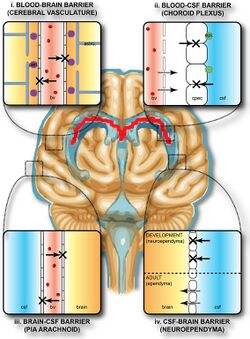
Blood brain barrier
The blood brain barrier (BBB) is the semipermeable membrane lining the vascular network of the brain. More specifically, it is a barrier between blood and interstitial fluid of the brain. It is formed by the tight junctions between endothelial cells, astrocytes, and pericytes in the capillaries. The BBB has high electrical resistance. Some diseases can damage this barrier and cause leakage. It is crucial to maintain integrity. Researches suggest increased intake of vitamins and antioxidants as well as stress reduction can restore the blood brain barrier. This is the most highly selective barrier in the body that restricts passive diffusion of solutes, large and hydrophilic molecules, and immune factors. Two molecule properties that affect movement through BBB are surface activity and size.[4] The main purpose of this structure is to protect the brain from pathogens or toxic substances. The restrictive barrier maintains homeostasis and prevents any alterations to neuronal functions. Therefore, it is challenging to deliver pharmaceuticals directly to the brain.
Recent studies on nose-to-brain drug delivery
Alzheimer's
Neurodegenerative diseases occur from loss of neuronal structure and function. This progressive degeneration of neurons is irreversible. Alzheimer's is a neurodegenerative disease that begins with short-term memory loss progressing to loss of control over heartbeat and breathing. It has been over 100 years since Alois Alzheimer first presented the world disease to the world in 1906. There is evidence for the efficacy of intranasal delivery to treat Alzheimer's. Intranasal delivery of insulin showed greater memory improvement in patients with Alzheimer's than in healthy individuals.[5] Increased microglial activation inflammation are characteristics of Alzheimer's. Animal studies show intranasal administration of pro-resolving lipid mediators decreased both factors, slowing pathogenesis of this disease.[6] Delivering a novel peptide via intranasal route reduced amyloid beta plaques, a defining trait of Alzheimer's and enhanced cognitive functions.[7] Intranasal delivery of anti-Alzheimer's drug dispersed through hydrogel in rabbits demonstrated higher bioavailability compared to oral tablets.[8] MiR132 is an RNA molecule that regulates neuronal morphology and maintains survival. This molecule is downregulated with Alzheimer's. A study administered PEG-PLA nanoparticles loaded with this miRNA to mice through the nasal route. This novel therapy showed increased expression of miR132 and improved memory function.[9] To strengthen the effectiveness of intranasal delivery, there are studies to develop permeation enhancers to better improve drug transport across the blood brain barrier.[10]
Glioblastoma
Abnormal cell growth and formation of mass in the brain tissue or nearby regions may cause brain cancer. Constant headaches, seizures, and blurred vision are common symptoms. Glioblastoma (GBM) is the most fast-growing and deadliest brain tumor. Though the main cause of glioblastoma remains unknown, it originates when astrocytes mutate and multiply uncontrollably forming tumors in the frontal and temporal lobes of the brain. The challenge with current therapeutics is to initiate tumor cell apoptosis with no toxic effects to healthy brain tissue. Nanoparticles loaded with chemotherapeutics delivered through the intranasal route show promising results in treating glioblastoma. PLGA-based nanoparticles loaded with paclitaxel or doxorubicin conjugated with a RGD sequence targeted the glioblastoma microenvironment and reduced tumor volume through cell death.[11][12] MicroRNA-21 (miR-21) inhibits pro-apoptotic genes increasing progression of glioblastoma. Self-assembling nanoparticles produced with anti-tumor peptides were administered intranasally and reduced miR-21 levels increasing tumor cell apoptosis.[13]
Epilepsy
Infection, head injury, or strokes can cause sudden bursts of neuronal activity leading to abnormal behaviors, muscle movement, and mood changes. This condition is known as seizure. Epilepsy is characterized by recurring seizures. Some possible causes of epilepsy include imbalance or disruption of neurotransmitters, strokes, or brain injury. Intranasal delivery of carbamazepine nanoparticles increase antiepileptic drug bioavailability.[14] Administering a self-assembling hydrogel with neuroactive drugs to treat Parkinson's disease appears to be biocompatible, low in toxicity, and have a good recovery capacity. Nasal delivery of this gel demonstrated increased drug concentration in the brain.[15] Oxytocin is a hormone which is observed to alleviate anxiety symptoms in people with autism. Intranasal administration indicated efficient transfer of pharmacologically active oxytocin from nasal cavity to brain.[16]
Parkinson's
Similar to Alzheimer's, Parkinson's is the most common neurodegenerative disease associated with balance and coordination issues, muscle stiffness, and tremors. During the early 1800s, James Parkinson medically defined this disease. A study observed improvement in locomotor abilities in rats with Parkinson's after intranasal delivery of conjugated mitochondrial systems.[17] Another study demonstrated delivery of neuroactive drugs in a hydrogel increased residence times in the nasal cavity and concentration in the brain.[18] Administering therapeutics combined with nanocarriers is shown to directly transfer drugs to the target cells and enhance accumulation. The observed effects include improved neuronal signaling and locomotion.[19] Furthermore, intranasal delivery of biodegradable nanoparticles surface-modified with lactoferrin increase accumulation in the brain and cellular uptake.[20]
Depression
Characterized by loss of neuroplasticity, depression is a common mood disorder causing persistent negative emotions and changes in lifestyle. Intranasal delivery of relaxin-3 mimetics demonstrated significant anti-depressant activity in behavior paradigms of rat models.[21] Delivering a thermoresponsive hydrogel loaded with berberine intranasally exhibited high bioavailability in hippocampus and anti-depressant activity.[22]
Anxiety
Anxiety can impair hippocampus function which increases risk of depression and dementia. Anxiolytic effects were observed in animal models post-intranasal delivery of a loaded polymeric nanoparticles.[23] Another study indicated intranasal delivery of neuropeptide Y lowered anxiety in rats.[24]
Anorexia nervosa
Anorexia nervosa (AN) is a common eating disorder characterized by low intake of food from fear of weight gain. Several complications are associated with this chronic disorder such as fatigue, insomnia, and low blood pressure. Intranasal administration of oxytocin in patients with AN significantly lowered food anticipation and eating concern.[25]
Substance use disorder
Uncontrolled and continuous use of a substance, drugs or alcohol, is known as substance use disorder. Substances can interfere with neuronal signaling and potentially disrupt the brain circuit. Addiction to these substances impairs thinking, behavior, and other biological functions. Intranasal delivery of insulin is associated with improvement in brain metabolic activities and alleviate impulsivity.[26] Opioid addiction is prevalent and associated with many substance abuse deaths. A study observed high biodistribution in the brain and reduction in opioid overdose in rats administered with naloxone-loaded lipid nanoparticles.[27]
Post-traumatic stress disorder
Witnessing a devastating or terrifying situation can lead to post-traumatic stress disorder (PTSD). This mental health condition triggers anxiety, depression, and extreme fear with memories. Intranasal administration of temperature-sensitive hydrogels loaded with PTSD medications showed enhanced brain targeting effects and tissue distribution.[28] Similarly, another study observed anti-PTSD effects with intranasal administration of loaded hydrogels.[29]
Schizophrenia
Schizophrenia is a chronic mental health condition caused by changes in brain chemistry and structure. Genetics and environment are hypothesized to play a key role in development of this disorder. Research suggests impaired gene expression or chemical imbalance may impact this condition. Anxiety can increase risk of schizophrenia and symptoms include hallucinations, disorganized speech, and abnormal behavior. Davunetide (NAP) is a segment of activity-dependent neuroprotective protein (ADNP). ADNP is reported be downregulated with schizophrenia. A study observed decreased hyperactivity in mice when treated with NAP via the intranasal route.[30]
Migraine
Migraine occurs with episodes of intense headache causing nausea and throbbing pain. Stress and hormonal changes can be a trigger migraine. A nasal spray containing sumatriptan demonstrated a significant reduction of migraine pain. Further clinical studies of intranasal administration of sumatriptan (ST) can help evaluate efficacy and safety of such delivery systems.[31]
Nanosystems for Intranasal Drug Delivery
Nanoparticles are drug delivery systems ranging from 1-1000 nm in diameter. Lipid-based and polymer-based nanocarriers are commonly used for nose-to-brain delivery as they exert high stability, solubility, and adherence.[32] Exosomes and dendrimers are other potential nanocarriers. Nanosystems can be synthesized either using physical or chemical methods. A few physical methods include evaporation-condensation reaction and laser ablation. Irradiation, microemulsion, and chemical reduction are common chemical techniques to develop nanoparticles. Sonication, electroporation, and incubation are common methods to load drugs into nanocarriers.
Coating these nanosystems with mucoadhesive agents, stimulus-sensitive materials, or antibodies can enhance biocompatibility, clearance rates, specificity, and bioavailability. Penetration and absorption enhancers can significantly increase the overall efficacy of the system. Imaging studies along with measurement of drug transfer efficiency and bioavailability can further support the role of these drug delivery systems.
Lipid-based nanoparticles
Lipid-based nanoparticles (LNP) can deliver molecules with low toxicity and controlled release. Liposomes, solid lipid nanoparticles (SLN), nanostructured lipid carriers (NLC), and nanoemulsions are examples.
Liposomes are made up of phospholipids forming spherical vesicles. This property enables liposomes to exhibit high biocompatibility and biodegradability. Studies report potential application of liposomes to treat brain diseases due to increased retention and absorption in nasal cavity, and high brain biodistribution.[33] A previous study developed a cationic liposome loaded with mRNA and green fluorescent protein (GFP). Intranasal delivery of this formulation in murine models demonstrated high brain biodistribution and expression of mRNA-GFP.[34]
Solid lipid nanoparticles (SLNs) are made up of solid lipids forming a matrix and stabilized by surfactants. They exhibit high physical stability and remain in solid state at different temperatures. Based on a study, intranasal delivery of SLNs loaded with rivastigmine tartrate (RT) exhibited no toxicity, stability, and improved bioavailability.[35] Sometimes burst release may occur due to rigidity and less flexibility in shape.
Nanostructured lipid carriers (NLC) are synthesized by a mixture of solid and aqueous lipids. NLC's are developed from SLNs, thus referred to as second generation LNPs. Intranasal administration of NLC loaded with curcumin (CRM) increased biodistribution and concentration in brain after emerging as a potential system for brain cancer.[36]
Small colloidal systems made of micelles containing oil, aqueous phases, and emulsifiers are called nanoemulsions. Intranasal delivery of gel nanoemulsion loaded with temozolomide is observed to exhibit sustained release and better permeation from nose to brain to treat glioblastoma.[37]
Polymer-based nanoparticles
Polymer-based nanoparticles can be made from either natural or synthetic sources. Nanospheres and nanocapsules are polymeric nanoparticle systems. Natural polymers can be found in the environment or human body. On the other hand, synthetic polymers do not occur naturally and are artificially developed polymers with chemical modifications. Natural polymer-based nanoparticles can be made up of chitosan, hyaluronic acid, alginate, and gelatin. Natural polymers exhibit excellent biocompatibility and biodegradability, and low toxicity. Synthetic polymer-based nanoparticles can consist of poly (glycolic acid) (PGA), poly (lactic acid) (PLA), and poly(L-lactide-co-glycolide) (PLGA).
A study evaluated chitosan nanoparticles loaded with an anti-epileptic drug, phenytoin (PHT), to treat epilepsy. Observations suggested high stability, sustained release, and bioavailability when these particles where administered via the intranasal route.[38] Similarly, administering PLGA nanoparticles loaded lamotrigine (LTG), polymer-based nanoparticle, showed better permeation through BBB and higher bioavailability.[39]
Exosomes
Exosomes are vesicular structures containing genetic information. Recently, exosomes are being utilized as drug carriers. These systems are observed to be stable, specific, and safe. Moreover, delivery of exosomes shows less immunogenic affects. Further surface modifications and conjugation with liposomes enhances the therapeutic effects. Based on a previous study, intranasal delivery of exosomes loaded with a Stat3 inhibitor reduced brain inflammation and slowed brain tumor growth.[40]
Dendrimers
Dendrimers are polymeric macromolecules with a branched network similar to a tree structure. Generally, they are spherical and homogeneous. Surface charge and molecule chemistry can play crucial role with drug interaction and release. Poly(amidoamine) (PAMAM) dendrimers are the most commonly used system. A study investigated potential application of dendrimer-based formulation of haloperidol. Intranasal administration showed improved targeting, and solubility as well as high concentrations in the brain.[41] Drugs can be loaded in dendrimers through formulation and nanoconstruct.[42]
Importance of physiochemical properties
For drug delivery systems to bypass the blood brain barrier, modifications of physiochemical properties can enhance safety and efficacy. Size, surface charge, and lipophilicity play a major role in substance bypassing the blood brain barrier. Smaller, positively charged, or more lipophilic molecules enhance efficacy of nose-to-brain delivery. Decrease in delivery system size increases permeation. As the membrane is negatively charged, a particle with positive surface charge interacts electrostatically which enhances bioadhesion. Carriers with more lipophilicity exert better mucoadhesion and residence time. Drug system pH, solubility, and hydrogen bonding potential are other physiochemical properties which should be evaluated.
References
- ↑ Erdő, Franciska; Bors, Luca Anna; Farkas, Dániel; Bajza, Ágnes; Gizurarson, Sveinbjörn (October 2018). "Evaluation of intranasal delivery route of drug administration for brain targeting" (in en). Brain Research Bulletin 143: 155–170. doi:10.1016/j.brainresbull.2018.10.009. PMID 30449731. https://linkinghub.elsevier.com/retrieve/pii/S0361923018303678.
- ↑ Park, Haesun; Otte, Andrew; Park, Kinam (2022-02-01). "Evolution of drug delivery systems: From 1950 to 2020 and beyond" (in en). Journal of Controlled Release 342: 53–65. doi:10.1016/j.jconrel.2021.12.030. ISSN 0168-3659. PMID 34971694.
- ↑ Park, Kinam (2014-09-28). "Controlled drug delivery systems: Past forward and future back" (in en). Journal of Controlled Release. 30th Anniversary Special Issue 190: 3–8. doi:10.1016/j.jconrel.2014.03.054. ISSN 0168-3659. PMID 24794901.
- ↑ Crowe, Tyler P.; Hsu, Walter H. (March 2022). "Evaluation of Recent Intranasal Drug Delivery Systems to the Central Nervous System" (in en). Pharmaceutics 14 (3): 629. doi:10.3390/pharmaceutics14030629. ISSN 1999-4923. PMID 35336004.
- ↑ Veronesi, Michael C.; Alhamami, Mosa; Miedema, Shelby B.; Yun, Yeonhee; Ruiz-Cardozo, Miguel; Vannier, Michael W. (2020). "Imaging of intranasal drug delivery to the brain". American Journal of Nuclear Medicine and Molecular Imaging 10 (1): 1–31. ISSN 2160-8407. PMID 32211216.
- ↑ Emre, Ceren; Arroyo-García, Luis E.; Do, Khanh V.; Jun, Bokkyoo; Ohshima, Makiko; Alcalde, Silvia Gómez; Cothern, Megan L.; Maioli, Silvia et al. (2022-03-21). "Intranasal delivery of pro-resolving lipid mediators rescues memory and gamma oscillation impairment in AppNL-G-F/NL-G-F mice" (in en). Communications Biology 5 (1): 245. doi:10.1038/s42003-022-03169-3. ISSN 2399-3642. PMID 35314851.
- ↑ Cheng, Yu-Sung; Chen, Zih-ten; Liao, Tai-Yan; Lin, Chen; Shen, Howard C-H; Wang, Ya-Han; Chang, Chi-Wei; Liu, Ren-Shyan et al. (May 2017). "An intranasally delivered peptide drug ameliorates cognitive decline in Alzheimer transgenic mice" (in en). EMBO Molecular Medicine 9 (5): 703–715. doi:10.15252/emmm.201606666. ISSN 1757-4676. PMID 28356312.
- ↑ Al Harthi, Sitah; Alavi, Seyed Ebrahim; Radwan, Mahasen Ali; El Khatib, Mona Mohamed; AlSarra, Ibrahim Abdullah (2019-07-02). "Nasal delivery of donepezil HCl-loaded hydrogels for the treatment of Alzheimer's disease" (in en). Scientific Reports 9 (1): 9563. doi:10.1038/s41598-019-46032-y. ISSN 2045-2322. PMID 31266990. Bibcode: 2019NatSR...9.9563A.
- ↑ Su, Yu; Sun, Bixi; Gao, Xiaoshu; Dong, Xinyue; Fu, Lanbo; Zhang, Yingxin; Li, Zhulin; Wang, Yue et al. (2020). "Intranasal Delivery of Targeted Nanoparticles Loaded With miR-132 to Brain for the Treatment of Neurodegenerative Diseases". Frontiers in Pharmacology 11: 1165. doi:10.3389/fphar.2020.01165. ISSN 1663-9812. PMID 32848773.
- ↑ Fonseca, Leonor C.; Lopes, João A.; Vieira, João; Viegas, Cláudia; Oliveira, Cláudia S.; Hartmann, Rafael P.; Fonte, Pedro (2021-04-01). "Intranasal drug delivery for treatment of Alzheimer's disease" (in en). Drug Delivery and Translational Research 11 (2): 411–425. doi:10.1007/s13346-021-00940-7. ISSN 2190-3948. PMID 33638130. https://doi.org/10.1007/s13346-021-00940-7.
- ↑ Ullah, Irfan; Chung, Kunho; Bae, Sumin; Li, Yan; Kim, Chunggu; Choi, Boyoung; Nam, Hye Yeong; Kim, Sun Hwa et al. (2020-04-06). "Nose-to-Brain Delivery of Cancer-Targeting Paclitaxel-Loaded Nanoparticles Potentiates Antitumor Effects in Malignant Glioblastoma" (in en). Molecular Pharmaceutics 17 (4): 1193–1204. doi:10.1021/acs.molpharmaceut.9b01215. ISSN 1543-8384. PMID 31944768. https://pubs.acs.org/doi/10.1021/acs.molpharmaceut.9b01215.
- ↑ Chung, Kunho; Ullah, Irfan; Kim, Nahyeon; Lim, Jaeyeoung; Shin, Jungah; Lee, Sangah C.; Jeon, Sangmin; Kim, Sun Hwa et al. (2020-07-02). "Intranasal delivery of cancer-targeting doxorubicin-loaded PLGA nanoparticles arrests glioblastoma growth". Journal of Drug Targeting 28 (6): 617–626. doi:10.1080/1061186X.2019.1706095. ISSN 1061-186X. PMID 31852284. https://doi.org/10.1080/1061186X.2019.1706095.
- ↑ Ha, Junkyu; Kim, Minkyung; Lee, Youngki; Lee, Minhyung (2021-09-17). "Intranasal delivery of self-assembled nanoparticles of therapeutic peptides and antagomirs elicits anti-tumor effects in an intracranial glioblastoma model" (in en). Nanoscale 13 (35): 14745–14759. doi:10.1039/D1NR03455C. ISSN 2040-3372. PMID 34474460. https://pubs.rsc.org/en/content/articlelanding/2021/nr/d1nr03455c.
- ↑ Liu, Shanshan; Yang, Shili; Ho, Paul C. (January 2018). "Intranasal administration of carbamazepine-loaded carboxymethyl chitosan nanoparticles for drug delivery to the brain" (in en). Asian Journal of Pharmaceutical Sciences 13 (1): 72–81. doi:10.1016/j.ajps.2017.09.001. PMID 32104380.
- ↑ Wang, Julie Tzu-Wen; Rodrigo, Ana C.; Patterson, Anna K.; Hawkins, Kirsten; Aly, Mazen M. S.; Sun, Jia; Al Jamal, Khuloud T.; Smith, David K. (July 2021). "Enhanced Delivery of Neuroactive Drugs via Nasal Delivery with a Self-Healing Supramolecular Gel" (in en). Advanced Science 8 (14): 2101058. doi:10.1002/advs.202101058. ISSN 2198-3844. PMID 34029010.
- ↑ Tanaka, Akiko; Furubayashi, Tomoyuki; Arai, Mari; Inoue, Daisuke; Kimura, Shunsuke; Kiriyama, Akiko; Kusamori, Kosuke; Katsumi, Hidemasa et al. (2018-03-05). "Delivery of Oxytocin to the Brain for the Treatment of Autism Spectrum Disorder by Nasal Application" (in en). Molecular Pharmaceutics 15 (3): 1105–1111. doi:10.1021/acs.molpharmaceut.7b00991. ISSN 1543-8384. PMID 29338251. https://pubs.acs.org/doi/10.1021/acs.molpharmaceut.7b00991.
- ↑ Chang, Jui-Chih; Chao, Yi-Chun; Chang, Huei-Shin; Wu, Yu-Ling; Chang, Hui-Ju; Lin, Yong-Shiou; Cheng, Wen-Ling; Lin, Ta-Tsung et al. (2021-05-19). "Intranasal delivery of mitochondria for treatment of Parkinson's Disease model rats lesioned with 6-hydroxydopamine" (in en). Scientific Reports 11 (1): 10597. doi:10.1038/s41598-021-90094-w. ISSN 2045-2322. PMID 34011937. Bibcode: 2021NatSR..1110597C.
- ↑ Wang, Julie Tzu-Wen; Rodrigo, Ana C.; Patterson, Anna K.; Hawkins, Kirsten; Aly, Mazen M. S.; Sun, Jia; Al Jamal, Khuloud T.; Smith, David K. (July 2021). "Enhanced Delivery of Neuroactive Drugs via Nasal Delivery with a Self-Healing Supramolecular Gel" (in en). Advanced Science 8 (14): 2101058. doi:10.1002/advs.202101058. ISSN 2198-3844. PMID 34029010.
- ↑ Peng, Huan; Li, Yan; Ji, Weihong; Zhao, Ruichen; Lu, Zhiguo; Shen, Jie; Wu, Yanyue; Wang, Jianze et al. (2022-01-25). "Intranasal Administration of Self-Oriented Nanocarriers Based on Therapeutic Exosomes for Synergistic Treatment of Parkinson's Disease" (in en). ACS Nano 16 (1): 869–884. doi:10.1021/acsnano.1c08473. ISSN 1936-0851. PMID 34985280. https://pubs.acs.org/doi/10.1021/acsnano.1c08473.
- ↑ Bi, Chenchen; Wang, Aiping; Chu, Yongchao; Liu, Sha; Mu, Hongjie; Liu, Wanhui; Wu, Zimei; Sun, Kaoxiang et al. (2016-12-07). "Intranasal delivery of rotigotine to the brain with lactoferrin-modified PEG-PLGA nanoparticles for Parkinson's disease treatment" (in English). International Journal of Nanomedicine 11: 6547–6559. doi:10.2147/IJN.S120939. PMID 27994458.
- ↑ Marwari, Subhi; Poulsen, Anders; Shih, Norrapat; Lakshminarayanan, Rajamani; Kini, R. Manjunatha; Johannes, Charles William; Dymock, Brian William; Dawe, Gavin Stewart (October 2019). "Intranasal administration of a stapled relaxin-3 mimetic has anxiolytic- and antidepressant-like activity in rats" (in en). British Journal of Pharmacology 176 (20): 3899–3923. doi:10.1111/bph.14774. ISSN 0007-1188. PMID 31220339.
- ↑ Wang, Qiang-Song; Li, Kefeng; Gao, Li-Na; Zhang, Ye; Lin, Ke-Ming; Cui, Yuan-Lu (2020-05-19). "Intranasal delivery of berberine via in situ thermoresponsive hydrogels with non-invasive therapy exhibits better antidepressant-like effects" (in en). Biomaterials Science 8 (10): 2853–2865. doi:10.1039/C9BM02006C. ISSN 2047-4849. PMID 32270794. https://pubs.rsc.org/en/content/articlelanding/2020/bm/c9bm02006c.
- ↑ Mahmoud, Khaled Y.; Elhesaisy, Nahla A.; Rashed, Abdelrahman R.; Mikhael, Ebram S.; Fadl, Mahmoud I.; Elsadek, Mahmoud S.; Mohamed, Merna A.; Mostafa, Merna A. et al. (2023-01-10). "Exploring the potential of intranasally administered naturally occurring quercetin loaded into polymeric nanocapsules as a novel platform for the treatment of anxiety" (in en). Scientific Reports 13 (1): 510. doi:10.1038/s41598-023-27665-6. ISSN 2045-2322. PMID 36627363. Bibcode: 2023NatSR..13..510M.
- ↑ Serova, L. I.; Laukova, M.; Alaluf, L. G.; Pucillo, L.; Sabban, E. L. (2014-01-01). "Intranasal neuropeptide Y reverses anxiety and depressive-like behavior impaired by single prolonged stress PTSD model" (in en). European Neuropsychopharmacology 24 (1): 142–147. doi:10.1016/j.euroneuro.2013.11.007. ISSN 0924-977X. PMID 24326087. https://www.sciencedirect.com/science/article/pii/S0924977X13003386.
- ↑ Russell, Janice; Maguire, Sarah; Hunt, Glenn E.; Kesby, Alice; Suraev, Anastasia; Stuart, Jordyn; Booth, Jessica; McGregor, Iain S. (2018-01-01). "Intranasal oxytocin in the treatment of anorexia nervosa: Randomized controlled trial during re-feeding" (in en). Psychoneuroendocrinology 87: 83–92. doi:10.1016/j.psyneuen.2017.10.014. ISSN 0306-4530. PMID 29049935. https://www.sciencedirect.com/science/article/pii/S0306453017306340.
- ↑ Kashyap, Bhavani; Hanson, Leah R.; Frey II, William H. (2020-01-01). "Intranasal Insulin: a Treatment Strategy for Addiction" (in en). Neurotherapeutics 17 (1): 105–115. doi:10.1007/s13311-019-00822-4. ISSN 1878-7479. PMID 31898283. PMC 7007475. https://doi.org/10.1007/s13311-019-00822-4.
- ↑ Kasina, Vishal; Mownn, Robert J.; Bahal, Raman; Sartor, Gregory C. (July 2022). "Nanoparticle delivery systems for substance use disorder" (in en). Neuropsychopharmacology 47 (8): 1431–1439. doi:10.1038/s41386-022-01311-7. ISSN 1740-634X. PMID 35351961.
- ↑ Pang, Lulu; Zhu, Siqing; Ma, Jinqiu; Zhu, Lin; Liu, Yijing; Ou, Ge; Li, Ruiteng; Wang, Yaxin et al. (2021-07-01). "Intranasal temperature-sensitive hydrogels of cannabidiol inclusion complex for the treatment of post-traumatic stress disorder" (in en). Acta Pharmaceutica Sinica B 11 (7): 2031–2047. doi:10.1016/j.apsb.2021.01.014. ISSN 2211-3835. PMID 34386336.
- ↑ Ou, Ge; Li, Qian; Zhu, Lin; Zhang, Yuanyuan; Liu, Yijing; Li, Xin; Du, Lina; Jin, Yiguang (2022-03-01). "Intranasal hydrogel of armodafinil hydroxypropyl-β-cyclodextrin inclusion complex for the treatment of post-traumatic stress disorder" (in en). Saudi Pharmaceutical Journal 30 (3): 265–282. doi:10.1016/j.jsps.2022.01.009. ISSN 1319-0164. PMID 35498223.
- ↑ Gozes, Illana (2011-02-01). "Microtubules, schizophrenia and cognitive behavior: Preclinical development of davunetide (NAP) as a peptide-drug candidate" (in en). Peptides 32 (2): 428–431. doi:10.1016/j.peptides.2010.10.030. ISSN 0196-9781. PMID 21050875. https://www.sciencedirect.com/science/article/pii/S0196978110004833.
- ↑ Assadpour, Sara; Shiran, Mohammad Reza; Asadi, Peyman; Akhtari, Javad; Sahebkar, Amirhossein (2022-01-15). "Harnessing Intranasal Delivery Systems of Sumatriptan for the Treatment of Migraine" (in en). BioMed Research International 2022: e3692065. doi:10.1155/2022/3692065. ISSN 2314-6133. PMID 35075426.
- ↑ Lee, David; Minko, Tamara (December 2021). "Nanotherapeutics for Nose-to-Brain Drug Delivery: An Approach to Bypass the Blood Brain Barrier" (in en). Pharmaceutics 13 (12): 2049. doi:10.3390/pharmaceutics13122049. ISSN 1999-4923. PMID 34959331.
- ↑ Lee, David; Minko, Tamara (December 2021). "Nanotherapeutics for Nose-to-Brain Drug Delivery: An Approach to Bypass the Blood Brain Barrier" (in en). Pharmaceutics 13 (12): 2049. doi:10.3390/pharmaceutics13122049. ISSN 1999-4923. PMID 34959331.
- ↑ Dhaliwal, Harkiranpreet Kaur; Fan, Yingfang; Kim, Jonghan; Amiji, Mansoor M. (2020-06-01). "Intranasal Delivery and Transfection of mRNA Therapeutics in the Brain Using Cationic Liposomes" (in en). Molecular Pharmaceutics 17 (6): 1996–2005. doi:10.1021/acs.molpharmaceut.0c00170. ISSN 1543-8384. PMID 32365295. https://pubs.acs.org/doi/10.1021/acs.molpharmaceut.0c00170.
- ↑ Arora, Deepshi; Bhatt, Shailendra; Kumar, Manish; Verma, Ravinder; Taneja, Yugam; Kaushal, Nikita; Tiwari, Abhishek; Tiwari, Varsha et al. (2022). "QbD-based rivastigmine tartrate-loaded solid lipid nanoparticles for enhanced intranasal delivery to the brain for Alzheimer's therapeutics". Frontiers in Aging Neuroscience 14: 960246. doi:10.3389/fnagi.2022.960246. ISSN 1663-4365. PMID 36034142.
- ↑ Madane, Rohini G.; Mahajan, Hitendra S. (2016-05-03). "Curcumin-loaded nanostructured lipid carriers (NLCs) for nasal administration: design, characterization, and in vivo study". Drug Delivery 23 (4): 1326–1334. doi:10.3109/10717544.2014.975382. ISSN 1071-7544. PMID 25367836.
- ↑ Bayanati, Masoumeh; Khosroshahi, Abolfazl Ghafouri; Alvandi, Maryam; Mahboobian, Mohammad Mehdi (2021-03-13). "Fabrication of a Thermosensitive In Situ Gel Nanoemulsion for Nose to Brain Delivery of Temozolomide" (in en). Journal of Nanomaterials 2021: e1546798. doi:10.1155/2021/1546798. ISSN 1687-4110.
- ↑ Yousfan, Amal; Rubio, Noelia; Natouf, Abdul Hakim; Daher, Aamal; Al-Kafry, Nedal; Venner, Kerrie; Kafa, Houmam (2020-08-03). "Preparation and characterisation of PHT-loaded chitosan lecithin nanoparticles for intranasal drug delivery to the brain" (in en). RSC Advances 10 (48): 28992–29009. doi:10.1039/D0RA04890A. ISSN 2046-2069. PMID 35520085. Bibcode: 2020RSCAd..1028992Y.
- ↑ Shah, Pranav; Dubey, Priya; Vyas, Bhavin; Kaul, Ankur; Mishra, Anil Kumar; Chopra, Dimple; Patel, Priya (2021-01-01). "Lamotrigine loaded PLGA nanoparticles intended for direct nose to brain delivery in epilepsy: pharmacokinetic, pharmacodynamic and scintigraphy study". Artificial Cells, Nanomedicine, and Biotechnology 49 (1): 511–522. doi:10.1080/21691401.2021.1939709. ISSN 2169-1401. PMID 34151674.
- ↑ Zhuang, Xiaoying; Xiang, Xiaoyu; Grizzle, William; Sun, Dongmei; Zhang, Shuangqin; Axtell, Robert C.; Ju, Songwen; Mu, Jiangyao et al. (2011-10-01). "Treatment of Brain Inflammatory Diseases by Delivering Exosome Encapsulated Anti-inflammatory Drugs From the Nasal Region to the Brain" (in English). Molecular Therapy 19 (10): 1769–1779. doi:10.1038/mt.2011.164. ISSN 1525-0016. PMID 21915101.
- ↑ Katare, Yogesh K.; Daya, Ritesh P.; Sookram Gray, Christal; Luckham, Roger E.; Bhandari, Jayant; Chauhan, Abhay S.; Mishra, Ram K. (2015-09-08). "Brain Targeting of a Water Insoluble Antipsychotic Drug Haloperidol via the Intranasal Route Using PAMAM Dendrimer" (in en). Molecular Pharmaceutics 12 (9): 3380–3388. doi:10.1021/acs.molpharmaceut.5b00402. ISSN 1543-8384. PMID 26226403.
- ↑ Chauhan, Abhay Singh (April 2018). "Dendrimers for Drug Delivery" (in en). Molecules 23 (4): 938. doi:10.3390/molecules23040938. ISSN 1420-3049. PMID 29670005.
 |