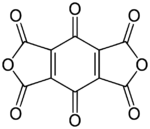
Chemistry:Benzoquinonetetracarboxylic dianhydride
From HandWiki
| |||
|
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1H,3H-Benzo[1,2-c:4,5-c′]difuran-1,3,4,5,7,8-hexone | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| C10O8 | |||
| Molar mass | 248.102 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
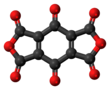
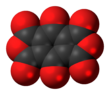
Benzoquinonetetracarboxylic dianhydride is an organic compound with formula C10O8 (an oxide of carbon) which can be seen as the result of removing two molecules of water H2O from benzoquinonetetracarboxylic acid.
It is a red solid, stable in dry air up to 140 °C and insoluble in ether, carbon tetrachloride, dichloromethane, and carbon disulfide. It reacts with acetone, ethyl acetate, tetrahydrofuran, ethanol, and water. It dissolves in methylated derivatives of benzene to give solutions ranging from orange to violet. When the molecule is exposed to moist air, it quickly turns blue.
The compound was synthesized in 1963 by P. R. Hammond, who claimed it was "one of the strongest π-electron acceptors so far described."[1]
See also
- Ethylenetetracarboxylic dianhydride
- Tetrahydroxy-1,4-benzoquinone biscarbonate
- Tetrahydroxy-1,4-benzoquinone bisoxalate
References
- ↑ Hammond, P. R. (1963). "1,4-Benzoquinone Tetracarboxylic Acid Dianhydride, C1008:A Strong Acceptor". Science 142 (3591): 502. doi:10.1126/science.142.3591.502. PMID 17748167. Bibcode: 1963Sci...142..502H.
 |