Chemistry:Nicotinonitrile
| |
| Names | |
|---|---|
| Preferred IUPAC name
Pyridine-3-carbonitrile | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C6H4N2 | |
| Molar mass | 104.112 g·mol−1 |
| Density | 1.1590 |
| Melting point | 51 °C (124 °F; 324 K) |
| Boiling point | 206.9 °C (404.4 °F; 480.0 K) |
| Vapor pressure | 0.296 mm Hg |
| Structure[1] | |
| Monoclinic | |
| P21/c (No. 14) | |
a = 3.808 Å, b = 13.120 Å, c = 10.591 Å α = 90°, β = 97.97°, γ = 90°
| |
Lattice volume (V)
|
524 |
Formula units (Z)
|
4 |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302, H315, H319, H335 | |
| P261, P264, P270, P271, P280, P301+312, P302+352, P304+340, P305+351+338, P312, P321, P330, P332+313, P337+313, P362, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
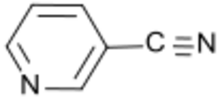
Nicotinonitrile or 3-cyanopyridine is an organic compound with the formula NCC5H4N. The molecule consists of a pyridine ring with a nitrile group attached to the 3-position. A colorless solid, it is produced by ammoxidation of 3-methylpyridine:[2]
- H3CC5H4N + NH3 + 1.5 O2 → NCC5H4N + 3 H2O
Nicotinonitrile is a precursor to the vitamin niacin.[3]
Nitrilase-catalyzed hydrolysis of 3-cyanopyridine by means of immobilized Rhodococcus rhodochrous J1[2] strains leads in quantitative yield to nicotinamide (vitamin B3).[4][5][6] The enzyme allows for a more selective synthesis as further hydrolysis of the amide to nicotinic acid is avoided.[7]
Oxidation of 3-cyanopyridine with hydrogen peroxide gives 3-cyanopyridine N-oxide, which hydrolyzes to nicotinic acid N-oxide, a precursor to pharmaceuticals.
References
- ↑ Kubiak, R.; Janczak, J.; Śledź, M. (June 2002). "Crystal structures of 2- and 3-cyanopyridine". Journal of Molecular Structure 610 (1–3): 59–64. doi:10.1016/S0022-2860(02)00012-1. Bibcode: 2002JMoSt.610...59K.
- ↑ 2.0 2.1 Abe, Nobuyuki; Ichimura, Hisao; Kataoka, Toshiaki; Morishita, Sinji; Shimizu, Shinkichi; Shoji, Takayuki; Watanabe, Nanao (2007). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a22_399.
- ↑ Eggersdorfer, Manfred; Adam, Geo; John, Michael; Hähnlein, Wolfgang; Labler, Ludvik; Baldenius, Kai-U.; von dem Bussche-Hünnefeld, Linda; Hilgemann, Eckhard et al. (2000) (in en). Vitamins. Wiley-VCH Verlag GmbH & Co. KGaA. pp. 148–150. doi:10.1002/14356007.a27_443. ISBN 978-3527306732.
- ↑ Nagasawa, Toru; Mathew, Caluwadewa Deepal; Mauger, Jacques; Yamada, Hideaki (1988). "Nitrile Hydratase-Catalyzed Production of Nicotinamide from 3-Cyanopyridine in Rhodococcus rhodochrous J1". Appl. Environ. Microbiol. 54 (7): 1766–1769. doi:10.1128/AEM.54.7.1766-1769.1988. PMID 16347686. Bibcode: 1988ApEnM..54.1766N.
- ↑ Hilterhaus, L.; Liese, A. (2007). "Building Blocks". in Ulber, Roland; Sell, Dieter. White Biotechnology. Advances in Biochemical Engineering / Biotechnology. 105. Springer Science & Business Media. pp. 133–173. doi:10.1007/10_033. ISBN 9783540456957. https://books.google.com/books?id=_tXoG93OWHgC&pg=PA141.
- ↑ Schmidberger, J. W.; Hepworth, L. J.; Green, A. P.; Flitsch, S. L. (2015). "Enzymatic Synthesis of Amides". in Faber, Kurt; Fessner, Wolf-Dieter; Turner, Nicholas J.. Biocatalysis in Organic Synthesis 1. Science of Synthesis. Georg Thieme Verlag. pp. 329–372. ISBN 9783131766113. https://books.google.com/books?id=8h_wBgAAQBAJ&pg=PA362.
- ↑ Petersen, Michael; Kiener, Andreas (1999). "Biocatalysis". Green Chem. 1 (2): 99–106. doi:10.1039/A809538H.
 |

