Chemistry:Triethylenetetramine
| |
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
N1,N1′-(Ethane-1,2-diyl)di(ethane-1,2-diamine) | |
Other names
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| 605448 | |
| ChEBI |
|
| ChEMBL |
|
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| 27008 | |
| KEGG | |
| MeSH | Trientine |
PubChem CID
|
|
| RTECS number |
|
| UNII |
|
| UN number | 2259 |
| |
| |
| Properties | |
| C6H18N4 | |
| Molar mass | 146.238 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Fishy, ammoniacal |
| Density | 982 mg mL−1 |
| Melting point | −34.6 °C; −30.4 °F; 238.5 K |
| Boiling point | 266.6 °C; 511.8 °F; 539.7 K |
| Miscible | |
| log P | 1.985 |
| Vapor pressure | <1 Pa (at 20 °C) |
Refractive index (nD)
|
1.496 |
| Thermochemistry | |
Heat capacity (C)
|
376 J K−1 mol−1 (at 60 °C) |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | DANGER |
| H312, H314, H317, H412 | |
| P273, P280, P305+351+338, P310 | |
| Flash point | 129 °C (264 °F; 402 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
|
| Related compounds | |
Related amines
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
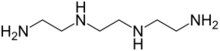
Triethylenetetramine (TETA and trien), also known as trientine (INN) when used medically, is an organic compound with the formula [CH2NHCH2CH2NH2]2. The pure freebase is a colorless oily liquid, but, like many amines, older samples assume a yellowish color due to impurities resulting from air-oxidation. It is soluble in polar solvents. The branched isomer tris(2-aminoethyl)amine and piperazine derivatives may also be present in commercial samples of TETA.[1] The hydrochloride salts are used medically as a treatment for copper toxicity.
Uses
Epoxy uses
The reactivity and uses of TETA are similar to those for the related polyamines ethylenediamine and diethylenetriamine. It is primarily used as a crosslinker ("hardener") in epoxy curing.[2][3] TETA, like other aliphatic amines, react quicker and at lower temperatures than aromatic amines due to less negative steric effects since the linear nature of the molecule provides it the ability to rotate and twist.
Medical uses
| Clinical data | |
|---|---|
| Trade names | Syprine, Cuprior, Cufence, others |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| DrugBank | |
| PDB ligand | |
The hydrochloride salt of TETA, referred to as trientine hydrochloride, is a chelating agent that is used to bind and remove copper in the body to treat Wilson's disease, particularly in those who are intolerant to penicillamine.[9] Some recommend trientine as first-line treatment, but experience with penicillamine is more extensive.[14]
Trientine hydrochloride (brand name Syprine) was approved for medical use in the United States in November 1985.[9]
Trientine tetrahydrochloride (brand name Cuprior) was approved for medical use in the European Union in September 2017.[12] It is indicated for the treatment of Wilson's disease in adults, adolescents and children five years of age or older who are intolerant to D-penicillamine therapy.[12]
Trientine dihydrochloride (brand name Cufence) was approved for medical use in the European Union in July 2019.[13] It is indicated for the treatment of Wilson's disease in adults, adolescents and children five years of age or older who are intolerant to D-penicillamine therapy.[13]
The most common side effects include nausea, especially when starting treatment, skin rash, duodenitis (inflammation of the duodenum, the part of the gut leading out of the stomach), and severe colitis (inflammation in the large bowel causing pain and diarrhea).[13]
Society and culture
Controversies
In the United States, Valeant Pharmaceuticals International raised the price of its Syprine brand of TETA from $625 to $21,267 for 100 pills over five years.[15] The New York Times said that this "egregious" price increase caused public outrage.[15] Teva Pharmaceuticals developed a generic, which patients and doctors expected to be cheaper, but when it was introduced in February 2018, Teva's price was $18,375 for 100 pills.[15] Aaron Kesselheim, who studies drug pricing at Harvard Medical School, said that drug companies price the product at what they think the market will bear.[15]
Production
TETA is prepared by heating ethylenediamine or ethanolamine/ammonia mixtures over an oxide catalyst. This process gives a variety of amines, especially ethylene amines which are separated by distillation and sublimation.[2][16]
Coordination chemistry
TETA is a tetradentate ligand in coordination chemistry, where it is referred to as trien.[17] Octahedral complexes of the type M(trien)L2 can adopt several diastereomeric structures.[18]
References
- ↑ "Ethyleneamines". Huntsman. 2007. http://www.huntsman.com/performance_products/Media%20Library/a_MC348531CFA3EA9A2E040EBCD2B6B7B06/Products_MC348531D0B9FA9A2E040EBCD2B6B7B06/Amines_MC348531D0BECA9A2E040EBCD2B6B7B06/Ethyleneamines_MC348531D0CD3A9A2E040EBCD2B6B7B06/files/ethyleneamines_brochure_huntsman_ethyleneamines.pdf.
- ↑ 2.0 2.1 Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. 2005. doi:10.1002/14356007.a02_001. ISBN 3527306730.
- ↑ "Triethylenetetramine" (in en). https://webbook.nist.gov/cgi/cbook.cgi?ID=C39421777&Mask=8.
- ↑ 4.0 4.1 "Trientine Waymade". 25 January 2021. https://www.tga.gov.au/apm-summary/trientine-waymade.
- ↑ 5.0 5.1 "AusPAR: Trientine dihydrochloride". 3 May 2021. https://www.tga.gov.au/auspar/auspar-trientine-dihydrochloride.
- ↑ "Summary Basis of Decision (SBD) for Mar-Trientine". 23 October 2014. https://hpr-rps.hres.ca/reg-content/summary-basis-decision-detailTwo.php?linkID=SBD00505&lang=en.
- ↑ "Cuprior 150 mg film-coated tablets - Summary of Product Characteristics (SmPC)". https://www.medicines.org.uk/emc/product/10100/smpc.
- ↑ "Trientine dihydrochloride Tillomed 250 mg capsules, hard - Summary of Product Characteristics (SmPC)". https://www.medicines.org.uk/emc/product/11626/smpc.
- ↑ 9.0 9.1 9.2 "Syprine- trientine hydrochloride capsule". 22 December 2016. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c34f77a7-996b-4470-b5df-d946a7fe5dbe.
- ↑ "Trientine hydrochloride capsule". 28 February 2020. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=a7cf5608-6446-4965-a855-5a0c0b7afddc.
- ↑ "Cuvrior- trientine tetrahydrochloride tablet, film coated". 20 May 2022. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=f73feeae-62ad-401e-b9f7-5cb269127750.
- ↑ 12.0 12.1 12.2 "Cuprior EPAR". 17 September 2018. https://www.ema.europa.eu/en/medicines/human/EPAR/cuprior. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ↑ 13.0 13.1 13.2 13.3 "Cufence EPAR". 24 May 2019. https://www.ema.europa.eu/en/medicines/human/EPAR/cufence. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ↑ "A practice guideline on Wilson disease" (pdf). Hepatology 37 (6): 1475–92. June 2003. doi:10.1053/jhep.2003.50252. PMID 12774027. http://www3.interscience.wiley.com/cgi-bin/fulltext/106595824/PDFSTART.[|permanent dead link|dead link}}]
- ↑ 15.0 15.1 15.2 15.3 Thomas, Katie (23 February 2018). "Patients Eagerly Awaited a Generic Drug. Then They Saw the Price.". https://www.nytimes.com/2018/02/23/health/valeant-drug-price-syprine.html.
- ↑ "Epoxide Resins". Plastics Materials (Seventh ed.). Oxford: Butterworth-Heinemann. 1999. pp. 744–777. doi:10.1016/B978-075064132-6/50067-X. ISBN 9780750641326. https://archive.org/details/plasticsmaterial00bryd.
- ↑ Stereochemistry of Coordination Compounds. Chichester: John Wiley. 1995. ISBN 047195599X. https://archive.org/details/stereochemistryo0000zele.
- ↑ "Three Isomers of the Trans -Diammine-[N,N′-bis(2-Aminoethyl)-1,2-Ethanediamine]-Cobalt(III) Complex Cation". Three Isomers of the trans-Diammine-[N,N′-bis(2-Aminoethyl)-1,2-Ethanediamine]-Cobalt(III) Complex Cation. Inorganic Syntheses. 23. 1985. pp. 79–82. doi:10.1002/9780470132548.ch16. ISBN 9780470132548.
External links
- "Triethylenetetramine". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/rn/112-24-3.
- "Trientine hydrochloride". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/trientine%20hydrochloride.
- "Trientine tetrahydrochloride". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/trientine%20tetrahydrochloride.
 |

