Biology:Amyloid beta
| Amyloid beta peptide (beta-APP) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
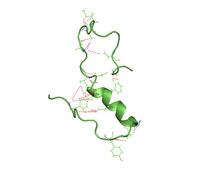 A partially folded structure of amyloid beta(1 40) in an aqueous environment (pdb 2lfm)[1] | |||||||||
| Identifiers | |||||||||
| Symbol | APP | ||||||||
| Pfam | PF03494 | ||||||||
| InterPro | IPR013803 | ||||||||
| SCOP2 | 2lfm / SCOPe / SUPFAM | ||||||||
| TCDB | 1.C.50 | ||||||||
| OPM superfamily | 304 | ||||||||
| OPM protein | 2y3k | ||||||||
| Membranome | 45 | ||||||||
| |||||||||
| amyloid beta (A4) precursor protein (peptidase nexin-II, Alzheimer disease) | |
|---|---|
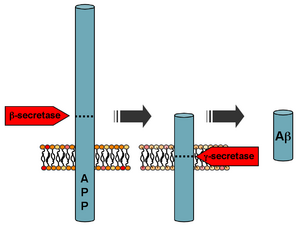 Processing of the amyloid precursor protein | |
| Identifiers | |
| Symbol | APP |
| Alt. symbols | AD1 |
| NCBI gene | 351 |
| HGNC | 620 |
| OMIM | 104760 |
| RefSeq | NM_000484 |
| UniProt | P05067 |
| Other data | |
| Locus | Chr. 21 q21.2 |
Amyloid beta (Aβ or Abeta) denotes peptides of 36–43 amino acids that are the main component of the amyloid plaques found in the brains of people with Alzheimer's disease.[2] The peptides derive from the amyloid-beta precursor protein (APP), which is cleaved by beta secretase and gamma secretase to yield Aβ in a cholesterol-dependent process and substrate presentation.[3] Aβ molecules can aggregate to form flexible soluble oligomers which may exist in several forms. It is now believed that certain misfolded oligomers (known as "seeds") can induce other Aβ molecules to also take the misfolded oligomeric form, leading to a chain reaction akin to a prion infection. The oligomers are toxic to nerve cells.[4] The other protein implicated in Alzheimer's disease, tau protein, also forms such prion-like misfolded oligomers, and there is some evidence that misfolded Aβ can induce tau to misfold.[5][6]
A study has suggested that APP and its amyloid potential is of ancient origins, dating as far back as early deuterostomes.[7]
Normal function
The normal function of Aβ is not well understood.[8] Though some animal studies have shown that the absence of Aβ does not lead to any obvious loss of physiological function,[9][10] several potential activities have been discovered for Aβ, including activation of kinase enzymes,[11][12] protection against oxidative stress,[13][14] regulation of cholesterol transport,[15][16] functioning as a transcription factor,[17][18] and anti-microbial activity (potentially associated with Aβ's pro-inflammatory activity).[19][20][21]
The glymphatic system clears metabolic waste from the mammalian brain, and in particular amyloid beta.[22] A number of proteases have been implicated by both genetic and biochemical studies as being responsible for the recognition and degradation of amyloid beta; these include insulin degrading enzyme[23] and presequence protease.[24] The rate of removal is significantly increased during sleep.[25] However, the significance of the glymphatic system in Aβ clearance in Alzheimer's disease is unknown.[26]
Disease associations
Aβ is the main component of amyloid plaques, extracellular deposits found in the brains of people with Alzheimer's disease.[27] Aβ can also form the deposits that line cerebral blood vessels in cerebral amyloid angiopathy. The plaques are composed of a tangle of Aβ oligomers [28] and regularly ordered aggregates called amyloid fibrils,[29] a protein fold shared by other peptides such as the prions associated with protein misfolding disease, also known as proteinopathy.
Alzheimer's disease
Research suggests that soluble oligomeric forms of the amyloid beta may be causative agents in the development of Alzheimer's disease.[30] It is generally believed that Aβ oligomers are the most toxic.[31] Several genetic, cell biology, biochemical and animal studies using experimental models support the concept that Aβ plays a central role in the development of Alzheimer's disease pathology.[32][33]
Brain Aβ is elevated in people with sporadic Alzheimer's disease. Aβ is the main constituent of brain parenchymal and vascular amyloid; it contributes to cerebrovascular lesions and is neurotoxic.[32][33][34] It is unresolved how Aβ accumulates in the central nervous system and subsequently initiates the disease of cells. Significant efforts have been focused on the mechanisms responsible for Aβ production, including the proteolytic enzymes gamma- and β-secretases which generate Aβ from its precursor protein, APP (amyloid precursor protein).[35][36] Aβ circulates in plasma, cerebrospinal fluid (CSF) and brain interstitial fluid (ISF) mainly as soluble Aβ40.[32][37] Amyloid plaques contain both Aβ40 and Aβ42,[38] while vascular amyloid is predominantly the shorter Aβ40. Several sequences of Aβ were found in both lesions.[39][40]
Increases in either total Aβ levels or the relative concentration of both Aβ40 and Aβ42 (where the former is more concentrated in cerebrovascular plaques and the latter in neuritic plaques)[41] have been implicated in the pathogenesis of both familial and sporadic Alzheimer's disease. Due to its more hydrophobic nature, the Aβ42 is the most amyloidogenic form of the peptide. However the central sequence KLVFFAE is known to form amyloid on its own, and probably forms the core of the fibril.[citation needed] One study further correlated Aβ42 levels in the brain not only with onset of Alzheimer's disease, but also reduced cerebrospinal fluid pressure, suggesting that a build-up or inability to clear Aβ42 fragments may play a role into the pathology.[42]
The "amyloid hypothesis" — that the plaques are responsible for the pathology of Alzheimer's disease — is accepted by the majority of researchers, but is not conclusively established. An alternative hypothesis is that amyloid oligomers rather than plaques are responsible for the disease.[31][43]
Cancer
While Aβ has been implicated in cancer development, prompting studies on a variety of cancers to elucidate the nature of its possible effects, results are largely inconclusive. Aβ levels have been assessed in relation to a number of cancers, including esophageal, colorectal, lung, and hepatic, in response to observed reductions in risk for developing Alzheimer's disease in survivors of these cancers. All cancers were shown to be associated positively with increased Aβ levels, particularly hepatic cancers.[44] This direction of association however has not yet been established. Studies focusing on human breast cancer cell lines have further demonstrated that these cancerous cells display an increased level of expression of amyloid precursor protein.[45]
Down syndrome
Adults with Down syndrome had accumulation of amyloid in association with evidence of Alzheimer's disease, including declines in cognitive functioning, memory, fine motor movements, executive functioning, and visuospatial skills.[46]
Formation
Aβ is formed after sequential cleavage of the amyloid precursor protein (APP), a transmembrane glycoprotein of undetermined function. APP can be cleaved by the proteolytic enzymes α-, β- and γ-secretase; Aβ protein is generated by successive action of the β and γ secretases. The γ secretase, which produces the C-terminal end of the Aβ peptide, cleaves within the transmembrane region of APP and can generate a number of isoforms of 30–51 amino acid residues in length.[47] The most common isoforms are Aβ40 and Aβ42; the longer form is typically produced by cleavage that occurs in the endoplasmic reticulum, while the shorter form is produced by cleavage in the trans-Golgi network.[48]
Genetics
Autosomal-dominant mutations in APP cause hereditary early-onset Alzheimer's disease (familial AD, fAD). This form of AD accounts for no more than 10% of all cases, and the vast majority of AD is not accompanied by such mutations.[49] However, familial Alzheimer's disease is likely to result from altered proteolytic processing. This is evidenced by the fact that many mutations that lead to fAD occur near γ-secretase cleavage sites on APP.[50] One of the most common mutations causing fAD, London Mutation, occurs at codon 717 of the APP gene,[51][52] and results in a valine to isoleucine amino acid substitution. Histochemical analysis of the APP V717I mutation has revealed extensive Aβ pathology throughout neuroaxis as well as widespread cerebral amyloid angiopathy (CAA).[53]
The gene for the amyloid precursor protein is located on chromosome 21, and accordingly people with Down syndrome have a very high incidence of Alzheimer's disease.[54]
Structure and toxicity
Amyloid beta is commonly thought to be intrinsically unstructured, meaning that in solution it does not acquire a unique tertiary fold but rather populates a set of structures. As such, it cannot be crystallized and most structural knowledge on amyloid beta comes from NMR and molecular dynamics. Early NMR-derived models of a 26-aminoacid polypeptide from amyloid beta (Aβ 10–35) show a collapsed coil structure devoid of significant secondary structure content.[55] However, the most recent (2012) NMR structure of (Aβ 1-40) has significant secondary and tertiary structure.[1] Replica exchange molecular dynamics studies suggested that amyloid beta can indeed populate multiple discrete structural states;[56] more recent studies identified a multiplicity of discrete conformational clusters by statistical analysis.[57] By NMR-guided simulations, amyloid beta 1-40 and amyloid beta 1-42 also seem to feature highly different conformational states,[58] with the C-terminus of amyloid beta 1-42 being more structured than that of the 1-40 fragment.
Low-temperature and low-salt conditions allowed to isolate pentameric disc-shaped oligomers devoid of beta structure.[59] In contrast, soluble oligomers prepared in the presence of detergents seem to feature substantial beta sheet content with mixed parallel and antiparallel character, different from fibrils;[60] computational studies suggest an antiparallel beta-turn-beta motif instead for membrane-embedded oligomers.[61]
Immunotherapy research
Immunotherapy may stimulate the host immune system to recognize and attack Aβ, or provide antibodies that either prevent plaque deposition or enhance clearance of plaques or Aβ oligomers. Oligomerization is a chemical process that converts individual molecules into a chain consisting of a finite number of molecules. Prevention of oligomerization of Aβ has been exemplified by active or passive Aβ immunization. In this process antibodies to Aβ are used to decrease cerebral plaque levels. This is accomplished by promoting microglial clearance and/or redistributing the peptide from the brain to systemic circulation. Antibodies that target Aβ and were tested in clinical trials included aducanumab, bapineuzumab, crenezumab, gantenerumab, lecanemab, and solanezumab.[62][63]
Measuring amyloid beta
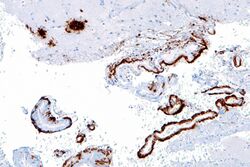
Imaging compounds, notably Pittsburgh compound B, (6-OH-BTA-1, a thioflavin), can selectively bind to amyloid beta in vitro and in vivo. This technique, combined with PET imaging, is used to image areas of plaque deposits in those with Alzheimer's.[64]
Post mortem or in tissue biopsies
Amyloid beta can be measured semiquantitatively with immunostaining, which also allows one to determine location. Amyloid beta may be primarily vascular, as in cerebral amyloid angiopathy, or in amyloid plaques in white matter.[65]
One sensitive method is ELISA which is an immunosorbent assay which utilizes a pair of antibodies that recognize amyloid beta.[66][67]
Atomic force microscopy, which can visualize nanoscale molecular surfaces, can be used to determine the aggregation state of amyloid beta in vitro.[68]
Vibrational microspectroscopy is a label-free method that measures the vibration of molecules in tissue samples.[69] Amyloid proteins like Aβ can be detected with this technique because of their high content of β-sheet structures.[70] Recently, the formation of Aβ fibrils was resolved in different plaque-types in Alzheimer's disease, indicating that plaques transit different stages in their development.[28]
Dual polarisation interferometry is an optical technique which can measure early stages of aggregation by measuring the molecular size and densities as the fibrils elongate.[71][72] These aggregate processes can also be studied on lipid bilayer constructs.[73]
See also
- TPM21
- Sylvain Lesné – Aβ*56
References
- ↑ 1.0 1.1 "A partially folded structure of amyloid-beta(1-40) in an aqueous environment". Biochemical and Biophysical Research Communications 411 (2): 312–6. July 2011. doi:10.1016/j.bbrc.2011.06.133. PMID 21726530.
- ↑ "The amyloid beta peptide: a chemist's perspective. Role in Alzheimer's and fibrillization". Chemical Reviews 112 (10): 5147–92. October 2012. doi:10.1021/cr3000994. PMID 22813427. http://centaur.reading.ac.uk/30230/2/AbetaRevisednew%20-IWH%20%281%29.pdf.
- ↑ "Regulation of beta-amyloid production in neurons by astrocyte-derived cholesterol". Proceedings of the National Academy of Sciences of the United States of America 118 (33): e2102191118. August 2021. doi:10.1073/pnas.2102191118. PMID 34385305. Bibcode: 2021PNAS..11802191W.
- ↑ "Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer's amyloid beta-peptide". Nature Reviews. Molecular Cell Biology 8 (2): 101–12. February 2007. doi:10.1038/nrm2101. PMID 17245412.
- ↑ "Alzheimer disease: a tale of two prions". Prion 7 (1): 14–9. Jan–Feb 2013. doi:10.4161/pri.22118. PMID 22965142.
- ↑ "Ubiquitous amyloids". Applied Biochemistry and Biotechnology 166 (7): 1626–43. April 2012. doi:10.1007/s12010-012-9549-3. PMID 22350870.
- ↑ "Origins of amyloid-β". BMC Genomics 14 (1): 290. April 2013. doi:10.1186/1471-2164-14-290. PMID 23627794.
- ↑ "Functional roles of amyloid-beta protein precursor and amyloid-beta peptides: evidence from experimental studies". Journal of Alzheimer's Disease 18 (2): 401–12. 2009. doi:10.3233/JAD-2009-1154. PMID 19584429.
- ↑ "Beta-amyloid exhibits antagonistic effects on alpha 7 nicotinic acetylcholine receptors in orchestrated manner". Journal of Medical Hypotheses and Ideas 8 (2): 48–52. 2014. doi:10.1016/j.jmhi.2014.01.001.
- ↑ "BACE1 (beta-secretase) knockout mice do not acquire compensatory gene expression changes or develop neural lesions over time". Neurobiology of Disease 14 (1): 81–8. October 2003. doi:10.1016/S0969-9961(03)00104-9. PMID 13678669.
- ↑ "Targeting the JNK MAPK cascade for inhibition: basic science and therapeutic potential". Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics 1697 (1–2): 89–101. March 2004. doi:10.1016/j.bbapap.2003.11.016. PMID 15023353.
- ↑ "Signaling effect of amyloid-beta(42) on the processing of AβPP". Experimental Neurology 221 (1): 18–25. January 2010. doi:10.1016/j.expneurol.2009.09.002. PMID 19747481.
- ↑ "A novel function of monomeric amyloid beta-protein serving as an antioxidant molecule against metal-induced oxidative damage". The Journal of Neuroscience 22 (12): 4833–41. June 2002. doi:10.1523/JNEUROSCI.22-12-04833.2002. PMID 12077180.
- ↑ "Abeta40, either soluble or aggregated, is a remarkably potent antioxidant in cell-free oxidative systems". Biochemistry 48 (20): 4354–70. May 2009. doi:10.1021/bi802361k. PMID 19320465.
- ↑ "Function of beta-amyloid in cholesterol transport: a lead to neurotoxicity". FASEB Journal 16 (12): 1677–9. October 2002. doi:10.1096/fj.02-0285fje. PMID 12206998.
- ↑ "Amyloid beta-protein stimulates trafficking of cholesterol and caveolin-1 from the plasma membrane to the Golgi complex in mouse primary astrocytes". Neuroscience 162 (2): 328–38. August 2009. doi:10.1016/j.neuroscience.2009.04.049. PMID 19401218.
- ↑ "The Alzheimer's amyloid β-peptide (Aβ) binds a specific DNA Aβ-interacting domain (AβID) in the APP, BACE1, and APOE promoters in a sequence-specific manner: characterizing a new regulatory motif". Gene 488 (1–2): 1–12. November 2011. doi:10.1016/j.gene.2011.06.004. PMID 21699964.
- ↑ "Functional activity of the novel Alzheimer's amyloid β-peptide interacting domain (AβID) in the APP and BACE1 promoter sequences and implications in activating apoptotic genes and in amyloidogenesis". Gene 488 (1–2): 13–22. November 2011. doi:10.1016/j.gene.2011.06.017. PMID 21708232.
- ↑ "Antimicrobial properties of amyloid peptides". Molecular Pharmaceutics 9 (4): 708–17. April 2012. doi:10.1021/mp200419b. PMID 22081976.
- ↑ "Antimicrobial peptides in the brain: neuropeptides and amyloid". Frontiers in Bioscience 4 (4): 1375–80. June 2012. doi:10.2741/S339. PMID 22652879.
- ↑ "Amyloid, tau, pathogen infection and antimicrobial protection in Alzheimer's disease -conformist, nonconformist, and realistic prospects for AD pathogenesis". Translational Neurodegeneration 7: 34. 2018. doi:10.1186/s40035-018-0139-3. PMID 30603085.
- ↑ "A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β". Science Translational Medicine 4 (147): 147ra111. August 2012. doi:10.1126/scitranslmed.3003748. PMID 22896675.
- ↑ "Structures of human insulin-degrading enzyme reveal a new substrate recognition mechanism". Nature 443 (7113): 870–4. October 2006. doi:10.1038/nature05143. PMID 17051221. Bibcode: 2006Natur.443..870S.
- ↑ "Molecular basis of substrate recognition and degradation by human presequence protease". Structure 22 (7): 996–1007. July 2014. doi:10.1016/j.str.2014.05.003. PMID 24931469.
- ↑ "Sleep drives metabolite clearance from the adult brain". Science 342 (6156): 373–7. October 2013. doi:10.1126/science.1241224. PMID 24136970. Bibcode: 2013Sci...342..373X.
- ↑ "Clearance systems in the brain-implications for Alzheimer disease". Nature Reviews. Neurology 11 (8): 457–70. August 2015. doi:10.1038/nrneurol.2015.119. PMID 26195256.
- ↑ "Amyloid-beta: a crucial factor in Alzheimer's disease". Medical Principles and Practice 24 (1): 1–10. 2014. doi:10.1159/000369101. PMID 25471398.
- ↑ 28.0 28.1 "Label-free vibrational imaging of different Aβ plaque types in Alzheimer's disease reveals sequential events in plaque development". Acta Neuropathologica Communications 8 (1): 222. December 2020. doi:10.1186/s40478-020-01091-5. PMID 33308303.
- ↑ "Assembly of β-Amyloid Aggregates at the Molecular Level". Chemtracts-Organic Chemistry 13 (1): 51–56. 2000.
- ↑ "Amyloid-beta protein dimers isolated directly from Alzheimer's brains impair synaptic plasticity and memory". Nature Medicine 14 (8): 837–42. August 2008. doi:10.1038/nm1782. PMID 18568035.
- ↑ 31.0 31.1 "The toxicity of amyloid β oligomers". International Journal of Molecular Sciences 13 (6): 7303–27. 2012. doi:10.3390/ijms13067303. PMID 22837695.
- ↑ 32.0 32.1 32.2 "Amyloidosis and Alzheimer's disease". Advanced Drug Delivery Reviews 54 (12): 1539–51. December 2002. doi:10.1016/S0169-409X(02)00149-7. PMID 12453671.
- ↑ 33.0 33.1 "Clearing the brain's amyloid cobwebs". Neuron 32 (2): 177–80. October 2001. doi:10.1016/S0896-6273(01)00475-5. PMID 11683988.
- ↑ "Genetic dissection of Alzheimer's disease and related dementias: amyloid and its relationship to tau". Nature Neuroscience 1 (5): 355–8. September 1998. doi:10.1038/1565. PMID 10196523.
- ↑ "Beta-secretase cleavage of Alzheimer's amyloid precursor protein by the transmembrane aspartic protease BACE". Science 286 (5440): 735–41. October 1999. doi:10.1126/science.286.5440.735. PMID 10531052. https://escholarship.org/uc/item/7s76h7jk.
- ↑ "Beta-secretase (BACE) as a drug target for Alzheimer's disease". Advanced Drug Delivery Reviews 54 (12): 1589–602. December 2002. doi:10.1016/S0169-409X(02)00157-6. PMID 12453676.
- ↑ Transport-clearance hypothesis for Alzheimer's disease and potential therapeutic implications. Landes Bioscience. 2003. pp. 114–122. https://www.ncbi.nlm.nih.gov/books/NBK5975/.
- ↑ "Amyloid plaque core protein in Alzheimer disease and Down syndrome". Proceedings of the National Academy of Sciences of the United States of America 82 (12): 4245–9. June 1985. doi:10.1073/pnas.82.12.4245. PMID 3159021. Bibcode: 1985PNAS...82.4245M.
- ↑ "The length of amyloid-beta in hereditary cerebral hemorrhage with amyloidosis, Dutch type. Implications for the role of amyloid-beta 1-42 in Alzheimer's disease". The Journal of Biological Chemistry 271 (50): 32185–91. December 1996. doi:10.1074/jbc.271.50.32185. PMID 8943274.
- ↑ "beta-Amyloid-(1-42) is a major component of cerebrovascular amyloid deposits: implications for the pathology of Alzheimer disease". Proceedings of the National Academy of Sciences of the United States of America 90 (22): 10836–40. November 1993. doi:10.1073/pnas.90.22.10836. PMID 8248178. Bibcode: 1993PNAS...9010836R.
- ↑ "Soluble amyloid beta peptide concentration as a predictor of synaptic change in Alzheimer's disease". The American Journal of Pathology 155 (3): 853–62. September 1999. doi:10.1016/S0002-9440(10)65184-X. PMID 10487842.
- ↑ "Levels of amyloid-beta-42 and CSF pressure are directly related in patients with Alzheimer's disease". Journal of Neural Transmission 124 (12): 1621–1625. December 2017. doi:10.1007/s00702-017-1786-8. PMID 28866757.
- ↑ "Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis". Science 300 (5618): 486–9. April 2003. doi:10.1126/science.1079469. PMID 12702875. Bibcode: 2003Sci...300..486K.
- ↑ "Plasma Amyloid-Beta Levels in Patients with Different Types of Cancer". Neurotoxicity Research 31 (2): 283–288. February 2017. doi:10.1007/s12640-016-9682-9. PMID 27913965.
- ↑ "Amyloid-β precursor protein promotes cell proliferation and motility of advanced breast cancer". BMC Cancer 14: 928. December 2014. doi:10.1186/1471-2407-14-928. PMID 25491510.
- ↑ "Cognitive decline and brain amyloid-β accumulation across 3 years in adults with Down syndrome". Neurobiology of Aging 58: 68–76. October 2017. doi:10.1016/j.neurobiolaging.2017.05.019. PMID 28715661.
- ↑ "Characterization of intermediate steps in amyloid beta (Aβ) production under near-native conditions". The Journal of Biological Chemistry 289 (3): 1540–50. January 2014. doi:10.1074/jbc.M113.498246. PMID 24225948.
- ↑ "Distinct sites of intracellular production for Alzheimer's disease A beta40/42 amyloid peptides". Nature Medicine 3 (9): 1016–20. September 1997. doi:10.1038/nm0997-1016. PMID 9288729.
- ↑ Alzheimer's Association (March 2008). "2008 Alzheimer's disease facts and figures". Alzheimer's & Dementia 4 (2): 110–33. doi:10.1016/j.jalz.2008.02.005. PMID 18631956.
- ↑ "Pathogenic APP mutations near the gamma-secretase cleavage site differentially affect Abeta secretion and APP C-terminal fragment stability". Human Molecular Genetics 10 (16): 1665–71. August 2001. doi:10.1093/hmg/10.16.1665. PMID 11487570.
- ↑ "Early-onset Alzheimer's disease caused by mutations at codon 717 of the beta-amyloid precursor protein gene". Nature 353 (6347): 844–6. October 1991. doi:10.1038/353844a0. PMID 1944558. Bibcode: 1991Natur.353..844C.
- ↑ "Familial Alzheimer's disease with the amyloid precursor protein position 717 mutation and sporadic Alzheimer's disease have the same cytoskeletal pathology". Neuroscience Letters 137 (2): 221–4. March 1992. doi:10.1016/0304-3940(92)90408-y. PMID 1584463.
- ↑ "Prominent amyloid plaque pathology and cerebral amyloid angiopathy in APP V717I (London) carrier - phenotypic variability in autosomal dominant Alzheimer's disease". Acta Neuropathologica Communications 8 (1): 31. March 2020. doi:10.1186/s40478-020-0891-3. PMID 32164763.
- ↑ "Alzheimer's disease and Down's syndrome: sharing of a unique cerebrovascular amyloid fibril protein". Biochemical and Biophysical Research Communications 122 (3): 1131–5. August 1984. doi:10.1016/0006-291X(84)91209-9. PMID 6236805.
- ↑ "The Alzheimer's peptide a beta adopts a collapsed coil structure in water". Journal of Structural Biology 130 (2–3): 130–41. June 2000. doi:10.1006/jsbi.2000.4288. PMID 10940221.
- ↑ "Amyloid beta-protein monomer folding: free-energy surfaces reveal alloform-specific differences". Journal of Molecular Biology 384 (2): 450–64. December 2008. doi:10.1016/j.jmb.2008.09.039. PMID 18835397.
- ↑ "Atomic-level characterization of the ensemble of the Aβ(1-42) monomer in water using unbiased molecular dynamics simulations and spectral algorithms". Journal of Molecular Biology 405 (2): 570–83. January 2011. doi:10.1016/j.jmb.2010.10.015. PMID 21056574.
- ↑ "The Alzheimer's peptides Abeta40 and 42 adopt distinct conformations in water: a combined MD / NMR study". Journal of Molecular Biology 368 (5): 1448–57. May 2007. doi:10.1016/j.jmb.2007.02.093. PMID 17397862.
- ↑ "Structural conversion of neurotoxic amyloid-beta(1-42) oligomers to fibrils". Nature Structural & Molecular Biology 17 (5): 561–7. May 2010. doi:10.1038/nsmb.1799. PMID 20383142.
- ↑ "Structural characterization of a soluble amyloid beta-peptide oligomer". Biochemistry 48 (9): 1870–7. March 2009. doi:10.1021/bi802046n. PMID 19216516.
- ↑ "Transmembrane structures for Alzheimer's Aβ(1-42) oligomers". Journal of the American Chemical Society 132 (38): 13300–12. September 2010. doi:10.1021/ja103725c. PMID 20822103.
- ↑ "Alzheimer's disease drug development pipeline: 2017". Alzheimer's & Dementia 3 (3): 367–384. September 2017. doi:10.1016/j.trci.2017.05.002. PMID 29067343.
- ↑ "Passive Aβ Immunotherapy: Current Achievements and Future Perspectives". Molecules 23 (5): 1068. May 2018. doi:10.3390/molecules23051068. PMID 29751505.
- ↑ "Imaging β-amyloid using [(18)F]flutemetamol positron emission tomography: from dosimetry to clinical diagnosis". European Journal of Nuclear Medicine and Molecular Imaging 43 (2): 362–373. February 2016. doi:10.1007/s00259-015-3208-1. PMID 26440450.
- ↑ "Quantitative Analysis of Amyloid Deposition in Alzheimer Disease Using PET and the Radiotracer 11C-AZD2184". Journal of Nuclear Medicine 55 (6): 932–8. June 2014. doi:10.2967/jnumed.113.133793. PMID 24732152.
- ↑ "Tissue Processing Prior to Analysis of Alzheimer's Disease Associated Proteins and Metabolites, Including Aβ". Amyloid Proteins. Methods in Molecular Biology. 849. 2012. pp. 493–506. doi:10.1007/978-1-61779-551-0_33. ISBN 978-1-61779-550-3.
- ↑ "Aβ Measurement by Enzyme-Linked Immunosorbent Assay". Amyloid Proteins. Methods in Molecular Biology. 849. 2012. pp. 507–27. doi:10.1007/978-1-61779-551-0_34. ISBN 978-1-61779-550-3.
- ↑ "In vitro characterization of conditions for amyloid-beta peptide oligomerization and fibrillogenesis". The Journal of Biological Chemistry 278 (13): 11612–22. March 2003. doi:10.1074/jbc.M210207200. PMID 12499373.
- ↑ Biomedical Vibrational Spectroscopy. Wiley. 2008. ISBN 978-0-470-22945-3.
- ↑ "Microspectroscopy (μFTIR) reveals co-localization of lipid oxidation and amyloid plaques in human Alzheimer disease brains". Analytical Chemistry 86 (24): 12047–54. December 2014. doi:10.1021/ac502667b. PMID 25415602.
- ↑ "Impairments of hippocampal synaptic plasticity induced by aggregated beta-amyloid (25-35) are dependent on stimulation-protocol and genetic background". Experimental Brain Research 179 (4): 621–30. June 2007. doi:10.1007/s00221-006-0819-6. PMID 17171334.
- ↑ "Monitoring the prevention of amyloid fibril formation by alpha-crystallin. Temperature dependence and the nature of the aggregating species". The FEBS Journal 274 (24): 6290–304. December 2007. doi:10.1111/j.1742-4658.2007.06144.x. PMID 18005258.
- ↑ "Insight into early events in the aggregation of the prion protein on lipid membranes". Biochimica et Biophysica Acta (BBA) - Biomembranes 1788 (10): 2245–51. October 2009. doi:10.1016/j.bbamem.2009.08.005. PMID 19703409.
External links
 |
