Biology:Lipofuscin
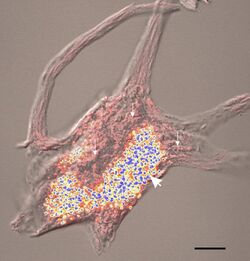
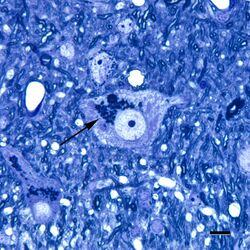
Lipofuscin is the name given to fine yellow-brown pigment granules composed of lipid-containing residues of lysosomal digestion.[1][2] It is considered to be one of the aging or "wear-and-tear" pigments, found in the liver, kidney, heart muscle, retina, adrenals, nerve cells, and ganglion cells.[3]
Formation and turnover
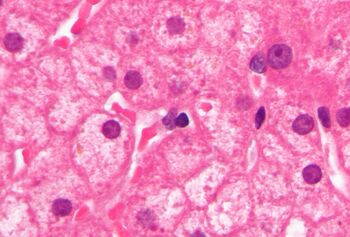
Lipofuscin appears to be the product of the oxidation of unsaturated fatty acids and may be symptomatic of membrane damage, or damage to mitochondria and lysosomes. Aside from a large lipid content, lipofuscin is known to contain sugars and metals, including mercury, aluminium, iron, copper and zinc.[4] Lipofuscin is also accepted as consisting of oxidized proteins (30–70%) as well as lipids (20–50%).[5] It is a type of lipochrome[6] and is specifically arranged around the nucleus.
The accumulation of lipofuscin-like material may be the result of an imbalance between formation and disposal mechanisms. Such accumulation can be induced in rats by administering a protease inhibitor (leupeptin); after a period of three months, the levels of the lipofuscin-like material return to normal, indicating the action of a significant disposal mechanism.[7] However, this result is controversial, as it is questionable if the leupeptin-induced material is true lipofuscin.[8][9] There exists evidence that "true lipofuscin" is not degradable in vitro;[10][11][12] whether this holds in vivo over longer time periods is not clear.
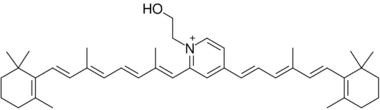
The ABCR -/- knockout mouse has delayed dark adaptation but normal final rod threshold relative to controls.[13] Bleaching the retina with strong light leads to formation of toxic cationic bis-pyridinium salt, N-retinylidene-N-retinyl-ethanolamine (A2E), which causes dry and wet age-related macular degeneration.[14] From this experiment, it was concluded that ABCR has a significant role in preventing formation of A2E in extracellular photoreceptor surfaces during bleach recovery.
Relation to diseases
Lipofuscin accumulation in the eye, is a major risk factor implicated in macular degeneration, a degenerative disease,[15] and Stargardt disease, an inherited juvenile form of macular degeneration.
In the peripheral nervous system, abnormal accumulation of lipofuscin known as lipofuscinosis[1] is associated with a family of neurodegenerative disorders – neuronal ceroid lipofuscinoses, the most common of these is Batten disease.
Also, pathological accumulation of lipofuscin is implicated in Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis, certain lysosomal diseases, acromegaly, denervation atrophy, lipid myopathy, chronic obstructive pulmonary disease,[16] and centronuclear myopathy. Accumulation of lipofuscin in the colon is the cause of the condition melanosis coli.
On the other hand, myocardial lipofuscin accumulation more directly reflects chronological ageing rather than human cardiac pathology.[17]
Possible therapies
Calorie restriction,[4] vitamin E,[4] and increased glutathione appear to reduce or halt the production of lipofuscin.
The nootropic drug piracetam appears to significantly reduce accumulation of lipofuscin in the brain tissue of rats.[18]
Other possible treatments:
- Centrophenoxine[19]
- Acetyl-L-carnitine[20]
- Ginkgo biloba[21]
- Dimethylethanolamine[22]
- Curcumin[23]
Wet macular degeneration can be treated using selective photothermolysis where a pulsed unfocused laser predominantly heats and kills lipofuscin-rich cells, leaving untouched healthy cells to multiply and fill in the gaps.[citation needed] The technique is also used as a skin treatment to remove tattoos, liverspots, and in general make skin appear younger. This ability to selectively target lipofuscin has opened up research opportunities in the field of anti-aging medicine.
Soraprazan (remofuscin) has been found to remove lipofuscin from retinal pigment epithelial cells in animals.[24] This opens up a new therapy option for the treatment of dry age-related macular degeneration and Stargardt disease, for which there is currently no treatment. The drug has now been granted orphan drug designation for the treatment of Stargardt disease by the European Medicines Agency.[25]
Other uses
Lipofuscin quantification is used for age determination in various crustaceans such as lobsters and spiny lobsters.[26][27] Since these animals lack bony parts, they cannot be aged in the same way as bony fish, in which annual increments in the ear-bones or otoliths are commonly used. Age determination of fish and shellfish is a fundamental step in generating basic biological data such as growth curves, and is needed for many stock assessment methods. Several studies have indicated that quantifying the amount of lipofuscin present in the eye-stalks of various crustaceans can give an index of their age. This method has not yet been widely applied in fisheries management mainly due to problems in relating lipofuscin levels in wild-caught animals with accumulation curves derived from aquarium-reared animals.
See also
References
- ↑ 1.0 1.1 Alberts, Daniel Albert (2012). Dorland's illustrated medical dictionary. (32nd ed.). Philadelphia, PA: Saunders/Elsevier. p. 1062. ISBN 978-1-4160-6257-8.
- ↑ "Medical Definition of LIPOFUSCIN" (in en). https://www.merriam-webster.com/medical/lipofuscin.
- ↑ Young B, Lowe JS, Stevens A, Heath JW. Wheater's Functional Histology: A Text and Atlas. 6th ed. Elsevier
- ↑ 4.0 4.1 4.2 Chris Gaugler, "Lipofuscin ", Stanislaus Journal of Biochemical Reviews May 1997
- ↑ Double, KL; Dedov, VN; Fedorow, H; Kettle, E; Halliday, GM; Garner, B; Brunk, UT (June 2008). "The comparative biology of neuromelanin and lipofuscin in the human brain.". Cellular and Molecular Life Sciences 65 (11): 1669–82. doi:10.1007/s00018-008-7581-9. PMID 18278576.
- ↑ "lipochrome", The Free Dictionary, https://medical-dictionary.thefreedictionary.com/lipochrome, retrieved 2021-02-18
- ↑ Katz, ML; Rice, LM; Gao, CL (1999). "Reversible accumulation of lipofuscin-like inclusions in the retinal pigment epithelium". Investigative Ophthalmology & Visual Science 40 (1): 175–181. PMID 9888441. http://www.iovs.org/cgi/content/abstract/40/1/175.
- ↑ Terman, Alexei; Brunk, Ulf T. (1999). "Is Lipofuscin Eliminated from Cells?". Investigative Ophthalmology and Visual Science 40 (10): 2463–2464. PMID 10476822. http://www.iovs.org/cgi/content/full/40/10/2463.
- ↑ Davies, Sallyanne; Ellis, Steven (1999). "Lipofuscin Turnover". Investigative Ophthalmology and Visual Science 40 (8): 1887–1888. PMID 10393067. http://www.iovs.org/cgi/content/full/40/8/1887.
- ↑ Terman, A, Brunk, UT (1998). "On the degradability and exocytosis of ceroid/lipofuscin in cultured rat cardiac myocytes". Mech Ageing Dev 100 (2): 145–156. doi:10.1016/S0047-6374(97)00129-2. PMID 9541135.
- ↑ Terman, A; Brunk, UT (1998). "Ceroid/lipofuscin formation in cultured human fibroblasts: the role of oxidative stress and lysosomal proteolysis". Mech Ageing Dev 104 (3): 277–291. doi:10.1016/s0047-6374(98)00073-6. PMID 9818731.
- ↑ Elleder, M; Drahota, Z; Lisá, V; Mares, V; Mandys, V; Müller, J; Palmer, DN (1995). "Tissue culture loading test with storage granules from animal models of neuronal ceroid-lipofuscinosis (Batten disease): testing their lysosomal degradability by normal and Batten cells". Am J Med Genet 57 (2): 213–221. doi:10.1002/ajmg.1320570220. PMID 7668332.
- ↑ "Insights into the function of Rim protein in photoreceptors and etiology of Stargardt's disease from the phenotype in abcr knockout mice". Cell 98 (1): 13–23. July 1999. doi:10.1016/S0092-8674(00)80602-9. PMID 10412977.
- ↑ "Retinopathy in mice induced by disrupted all-trans-retinal clearance". The Journal of Biological Chemistry 283 (39): 26684–93. September 2008. doi:10.1074/jbc.M804505200. PMID 18658157.
- ↑ John Lacey, "Harvard Medical signs agreement with Merck to develop potential therapy for macular degeneration", 23-May-2006
- ↑ Joakim Allaire; François Maltais; Pierre LeBlanc; Pierre-Michel Simard; François Whittom; Jean-François Doyon; Clermont Simard; Jean Jobin (2002). "Lipofuscin accumulation in the vastus lateralis muscle in patients with chronic obstructive pulmonary disease". Muscle and Nerve 25 (3): 383–389. doi:10.1002/mus.10039. PMID 11870715.
- ↑ Kakimoto, Yu; Okada, Chisa; Kawabe, Noboru; Sasaki, Ayumi; Tsukamoto, Hideo; Nagao, Ryoko; Osawa, Motoki (2019). "Myocardial lipofuscin accumulation in ageing and sudden cardiac death". Scientific Reports 9 (1): 3304. doi:10.1038/s41598-019-40250-0. ISSN 2045-2322. PMID 30824797. Bibcode: 2019NatSR...9.3304K.
- ↑ Paula-Barbosa, M. (1991). "The effects of Piracetam on lipofuscin of the rat cerebellar and hippocampa; neurons after long-term alcohol treatment and withdrawal". Alcoholism: Clinical and Experimental Research 15 (5): 834–838. doi:10.1111/j.1530-0277.1991.tb00610.x. PMID 1755517.
- ↑ Roy, D; Pathak, DN; Singh, R (1983). "Effect of centrophenoxine on the antioxidative enzymes in various regions of the aging rat brain.". Exp Gerontol 18 (3): 185–97. doi:10.1016/0531-5565(83)90031-1. PMID 6416880.
- ↑ Amenta F, Ferrante F, et al., Reduced lipofuscin accumulation in senescent rat brain by long-term acetyl-L-carnitine treatment. Arch Gerontol Geriatr. 1989 Sep-Oct;9(2):147-53.
- ↑ Huang, SZ; Luo, YJ; Wang, L; Cai, KY (Jan 2005). "Effect of ginkgo biloba extract on livers in aged rats.". World J Gastroenterol 11 (1): 132–5. doi:10.3748/wjg.v11.i1.132. PMID 15609412.
- ↑ Stenbäck, Frej; Weisburger, J. H.; Williams, G. M. (1988-02-01). "Effect of lifetime, administration of dimethylaminoethanol on longevity, aging changes, and cryptogenic neoplasms in C3H mice" (in en). Mechanisms of Ageing and Development 42 (2): 129–138. doi:10.1016/0047-6374(88)90068-1. ISSN 0047-6374. PMID 3361965. https://dx.doi.org/10.1016/0047-6374%2888%2990068-1.
- ↑ Shen, Li-Rong; Parnell, Laurence D.; Ordovas, Jose M.; Lai, Chao-Qiang (January 2013). "Curcumin and aging". BioFactors 39 (1): 133–140. doi:10.1002/biof.1086. ISSN 1872-8081. PMID 23325575. https://pubmed.ncbi.nlm.nih.gov/23325575/.
- ↑ Julien, S; Schraermeyer, U (Oct 2012). "Lipofuscin can be removed from the retinal pigment epithelium of monkeys". Neurobiol Aging 33 (10): 2390–7. doi:10.1016/j.neurobiolaging.2011.12.009. PMID 22244091.
- ↑ "EU/3/13/1208". 17 September 2018. https://www.ema.europa.eu/en/medicines/human/orphan-designations/eu3131208.
- ↑ Ingebrigt Uglem, Mark Belchier; Terje Svåsand (2005). "Age determination of European lobsters (Homarus gammarus L.) by histological quantification of lipofuscin". Journal of Crustacean Biology 25 (1): 95–99. doi:10.1651/c-2448.
- ↑ Kerry E. Maxwell; Thomas R. Matthews; Matt R. J. Sheehy; Rodney D. Bertelsen; Charles D. Derby (2007). "Neurolipofuscin is a measure of age in Panulirus argus, the Caribbean spiny lobster, in Florida". The Biological Bulletin 213 (1): 55–66. doi:10.2307/25066618. PMID 17679720. http://www.biolbull.org/content/213/1/55.full.
20. Young B, Lowe JS, Stevens A, Heath JW. Wheater's Functional Histology: A Text and Atlas. 6th ed. Elsevier
External links for general reviews
- "Lipofuscin". Int J Biochem Cell Biol 36 (8): 1400–4. 2004. doi:10.1016/j.biocel.2003.08.009. PMID 15147719.
- Histology at neuro.wustl.edu
- Histology image: 20301loa – Histology Learning System at Boston University
- Destroying Lipofuscin and Destroying Cancer, FightAging.org
- Unfocused Pulsed Lasers Selectively Destroy Lipofuscin, AcceleratingFuture.com
- Lipofuscin
 |
