Chemistry:1,2-Cyclopentanedione
From HandWiki
| |
| Names | |
|---|---|
| Preferred IUPAC name
Cyclopentane-1,2-dione | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C5H6O2 | |
| Molar mass | 98.101 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.371 g/cm3 |
| Melting point | 56 °C (133 °F; 329 K) |
| Boiling point | 87–88 °C (189–190 °F; 360–361 K) (15 mm Hg) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
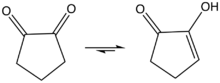
1,2-Cyclopentanedione is the organic compound with the formula (CH2)3(CO)2. It is one of two isomeric cyclopentanediones, the other being 1,3-cyclopentanedione. It was first prepared by base-induced condensation of di ethylglutarate with diethyloxalate, followed by hydrolysis of the resulting diketodiester followed by decarboxylation.[1] The enol is predicted to be about 1-3 kcal/mol more stable than the diketo form.[2] The enol structure has been confirmed by X-ray crystallography.[3]
Structurally related to 1,2-cyclopentanedione is 2-hydroxy-3-methyl-2-cyclopenten-1-one is a flavor additive, also called cyclotene.
References
- ↑ Hesse, Gerhard; Bücking, Eva (1949). "Die Synthese der Reduktinsäure (Synthesis of Reductic Acid)". Justus Liebigs Annalen der Chemie 563: 31–37. doi:10.1002/jlac.19495630105.
- ↑ Jana, Kalyanashis; Ganguly, Bishwajit (2018). "DFT Study to Explore the Importance of Ring Size and Effect of Solvents on the Keto–Enol Tautomerization Process of α- and β-Cyclodiones". ACS Omega 3 (7): 8429–8439. doi:10.1021/acsomega.8b01008. PMID 31458971.
- ↑ Gerrard, Juliet A.; Meade, Susie J.; Morris, Jonathan C.; Steel, Peter J. (2000). "Two crystalline modifications of 2-hydroxycyclopent-2-enone". Acta Crystallographica Section C: Crystal Structure Communications 56 (10): 1251–1253. doi:10.1107/S0108270100009550. PMID 11025315.
 |
