Chemistry:2,2,6,6-Tetramethylpiperidine
| |||
|
| |||
| Names | |||
|---|---|---|---|
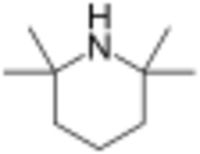
| Preferred IUPAC name
2,2,6,6-Tetramethylpiperidine | |||
| Other names
Norpempidine
Tetramethylpiperidine | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| Abbreviations | TMP | ||
| ChEMBL | |||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C9H19N | |||
| Molar mass | 141.254 g/mol | ||
| Appearance | Clear liquid | ||
| Density | 0.83 g/mL | ||
| Melting point | −59 °C (−74 °F; 214 K) | ||
| Boiling point | 152 °C (306 °F; 425 K) | ||
| Hazards | |||
| GHS pictograms |    
| ||
| GHS Signal word | Danger | ||
| H226, H301, H302, H314, H315, H319, H332, H335 | |||
| P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P280, P301+310, P301+312, P301+330+331, P302+352, P303+361+353, P304+312, P304+340, P305+351+338, P310, P312, P321, P330, P332+313 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
2,2,6,6-Tetramethylpiperidine, abbreviated TMP, HTMP, or TMPH, is an organic compound of the amine class. In appearance, it is a colorless liquid and has a "fishy", amine-like odor. This amine is used in chemistry as a hindered base (hindered amine). Although TMP finds limited use per se, its derivatives are a mainstay of hindered amine light stabilizers.
TMP is the starting material for an even stronger base, lithium tetramethylpiperidide and the radical species TEMPO. Another non-nucleophilic base is N,N-diisopropylethylamine. Its aqueous pKaH (conjugate acid dissociation constant, a measure of basicity) is 11.07 at 25 °C,[1] while its pKa (acid dissociation constant, a measure of acidity) is approximately 37.[2]
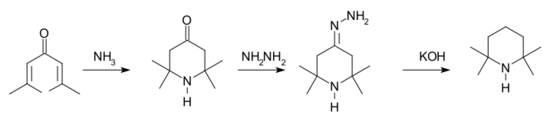
Preparation
Many routes for the synthesis of TMP have been reported. One method[3] starts with a conjugate addition reaction of ammonia to phorone. The intermediate triacetone amine is then reduced in a Wolff-Kishner reaction.
See also
- 2,6-Dimethylpiperidine
- Pempidine
- TEMPO ((2,2,6,6-Tetramethylpiperidin-1-yl)oxyl)
References
- ↑ CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data.. William M. Haynes, David R. Lide, Thomas J. Bruno (96th ed.). Boca Raton, Florida. 2015. ISBN 978-1-4822-6096-0. OCLC 910908643. https://www.worldcat.org/oclc/910908643.
- ↑ Reich, Hans (2022). "Bordwell pKa table". https://organicchemistrydata.org/hansreich/resources/pka/#pka_dmso_compilation.
- ↑ Detlef Kampmann; Georg Stuhlmüller; Roger Simon; Fabrice Cottet; Frédéric Leroux; Manfred Schlosser (2005). "A Large-Scale Low-Cost Access to the Lithium 2,2,6,6-Tetramethylpiperidide Precursor". Synthesis 2005 (6): 1028–1029. doi:10.1055/s-2004-834856.
External links
 |