Chemistry:Acenaphthene
Acenaphthene is a polycyclic aromatic hydrocarbon (PAH) consisting of naphthalene with an ethylene bridge connecting positions 1 and 8. It is a colourless solid. Coal tar consists of about 0.3% of this compound.[1]
Production and reactions
Acenaphthene was prepared for the first time in 1866[2] by Marcellin Berthelot by reacting hot naphthalene vapours with acetylene, and a year later he reproduced a similar reaction with ethylene as well as discovered acenaphthene in coal tar.[3] Later Berthelot and Bardy synthesized the compound by cyclization of α-ethylnaphthalene. Industrially, it is still obtained from coal tar together with its derivative acenaphthylene (and many other compounds).
Like other arenes, acenaphthene forms complexes with low valent metal centers. One example is (η6-acenaphthene)Mn(CO)3]+.[4]
Uses
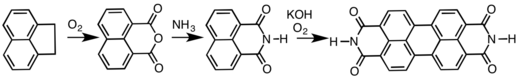
It is used on a large scale to prepare naphthalene dicarboxylic anhydride, which is a precursor to dyes and optical brighteners[1] (such as 1,4-bis(2-benzoxazolyl)naphthalene). Besides that, the anhydride is also the precursor to perylenetetracarboxylic dianhydride, precursor to several commercial pigments and dyes.[5][6]
References
- ↑ 1.0 1.1 Griesbaum, Karl; Behr, Arno; Biedenkapp, Dieter; Voges, Heinz-Werner; Garbe, Dorothea; Paetz, Christian; Collin, Gerd; Mayer, Dieter et al. (2000). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a13_227.
- ↑ (in fr) Annales de chimie et de physique. Chez Crochard. 1866. https://books.google.com/books?id=jytCAAAAcAAJ&pg=PA467.
- ↑ (in fr) Annales de chimie et de physique. Masson.. 1867. https://books.google.com/books?id=lLZJAQAAMAAJ&pg=PA226.
- ↑ S. B. Kim, S. Lotz, S. Sun, Y. K. Chung, R. D. Pike, D. A. Sweigart "Manganese Tricarbonyl Transfer (MTT) Agents" Inorganic Syntheses, 2010, Vol. 35, 109–128. doi:10.1002/9780470651568.ch6
- ↑ K. Hunger. W. Herbst "Pigments, Organic" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2012. doi:10.1002/14356007.a20_371
- ↑ Greene, M. "Perylene Pigments" in High Performance Pigments, 2009, Wiley-VCH, Weinheim. pp. 261-274.doi:10.1002/9783527626915.ch16
 |