Chemistry:Ammonium dichromate

| |
| |
| |
| Names | |
|---|---|
| IUPAC name
Ammonium dichromate
| |
| Other names
Ammonium bichromate
Ammonium pyrochromate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1439 |
| |
| |
| Properties | |
| (NH 4) 2Cr 2O 7 | |
| Molar mass | 252.063 g·mol−1 |
| Appearance | Orange-red crystals |
| Odor | odorless[1] |
| Density | 2.150 g/cm3 (20 °C (68 °F; 293 K))[1] |
| Melting point | 170 °C (338 °F; 443 K)[1] decomposes |
| |
| Solubility in ethanol | soluble |
| Structure | |
| monoclinic | |
| Hazards[1] | |
| GHS pictograms |     
|
| GHS Signal word | Danger |
| H272, H301, H312, H314, H317, H330, H334, H340, H350, H360, H372, H410 | |
| P201, P202, P210, P220, P221, P260, P264, P270, P271, P272, P273, P280, P284, P301+310+330Script error: No such module "Preview warning".Category:GHS errors, P301+330+331, P303+361+353, P304+340+310, P305+351+338+310, P308+313, P333+313, P342+311, P363, P370+378, P391, P403+233 | |
| NFPA 704 (fire diamond) | |
| 218 °C (424 °F; 491 K)[2] | |
Threshold limit value (TLV)
|
0.0002 mg/m3 (as Cr(VI)) (TWA), 0.0005 mg/m3 (as Cr(VI)) (STEL), 1 mg/10m3 (as CrO 3) (C) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
|
LC50 (median concentration)
|
0.2 mg/L (200 mg/m3) - 4h (Rat, inhalation, dust / mist)[1] |
| NIOSH (US health exposure limits):Template:Chembox NIOSH (set)/formatPocketGuideLink | |
PEL (Permissible)
|
0.005 mg/m3 (as Cr) |
REL (Recommended)
|
8 hours, 0.0002 mg/m3 (as Cr) |
IDLH (Immediate danger)
|
15 mg/m3 (as Cr(VI)) |
| Related compounds | |
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
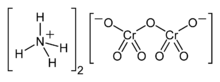
Ammonium dichromate is an inorganic compound with the formula (NH
4)
2Cr
2O
7. In this compound, as in all chromates and dichromates, chromium is in a +6 oxidation state, commonly known as hexavalent chromium. It is a salt consisting of ammonium ions and dichromate ions.
Ammonium dichromate is used in demonstrations of tabletop "volcanoes".[3] However, this demonstration has become unpopular due to concerns about the compound's carcinogenic nature. It has been used in pyrotechnics and in the early days of photography. It is also used in holography.
Properties
At standard temperature and pressure, the compound exists as orange, acidic crystals soluble in water and ethanol. It is formed by the action of chromic acid on ammonium hydroxide with subsequent crystallisation.[4]
The (NH
4)
2Cr
2O
7 crystal (C2/c, z = 4) contains a single type of ammonium ion, at sites of symmetry C1(2,3). Each NH+
4 centre is surrounded irregularly by eight oxygen atoms at N–O distances ranging from ca. 2.83 to 3.17 Å, typical of hydrogen bonds.[5]
Uses
Pyrotechnics
It has been used in pyrotechnics as part of a conversion coating method for magnesium powder. It may also be used when a dichromate compatible with ammonium perchlorate (NH
4ClO
4) is needed; potassium dichromate (K
2Cr
2O
7) is commonly used, but reacts with ammonium perchlorate over time to form potassium perchlorate and ammonium dichromate. This mixture occupies 5% more volume than the original K
2Cr
2O
7 + NH
4ClO
4 mixture and leads to eventual cracking of stars and consolidated compositions.[6]
It was used in a composition in the early 20th century in a combination firework called a serpent's nest to produce the green grass (or nest) from which a separately made pharaoh's serpent or black snake composition emerged.[7]
Holography
In holography, it has been used since the 1960s to sensitize dichromated gelatins or other materials on which to expose a hologram. This method has the advantage of being low cost, using relatively common materials, and only requiring isopropyl alcohol for development, but the gelatins created with these methods are sensitive to humidty. Potassium dichromate is also used for this purpose.[8][9][10]
Other
It was used in the early days of photography as well as in lithography, as a source of pure nitrogen in the laboratory, and as a catalyst.[11] It is also used as a mordant for dyeing pigments, in manufacturing of alizarin, chrome alum, leather tanning and oil purification.[4]{{page needed|date=June 2025} Photosensitive films containing PVA, ammonium dichromate, and a phosphor are spin-coated as aqueous slurries in the production of the phosphor raster of television screens and other devices. The ammonium dichromate acts as the photoactive site.[12]
Reactions
Tabletop volcanoes and thermal decomposition
File:12. Реакција на диспропорционирање.webm

The volcano demonstration involves igniting a pile of the salt, which initiates the following exothermic conversion:[14]
- (NH
4)
2Cr
2O
7(s) → Cr
2O
3(s) + N
2(g) + 4 H
2O(g) - (ΔH = −429.1±3 kcal/mol)
Like ammonium nitrate, it is thermodynamically unstable.[15][16] Its decomposition reaction produces voluminous dark green powdered chromium(III) oxide. Not all of the ammonium dichromate decomposes in this reaction. When the green powder is added to water a yellow/orange solution is obtained from left over ammonium dichromate.[citation needed]
Observations obtained using relatively high magnification microscopy during a kinetic study of the thermal decomposition of ammonium dichromate provided evidence that salt breakdown proceeds with the intervention of an intermediate liquid phase rather than a solid phase. The characteristic darkening of (NH
4)
2Cr
2O
7 crystals as a consequence of the onset of decomposition can be ascribed to the dissociative loss of ammonia accompanied by progressive anion condensation to Cr
3O2−
10, Cr
4O2−
13, etc., ultimately yielding CrO
3. The CrO
3 has been identified as a possible molten intermediate participating in (NH
4)
2Cr
2O
7 decomposition.[17]
Oxidation reactions
Ammonium dichromate is a strong oxidising agent and reacts, often violently, with many reducing agents. In general, the stronger the reducing agent, the more violent the reaction.[15]
It has also been used to promote the oxidation of alcohols and thiols. Ammonium dichromate, in the presence of Mg(HSO
4)
2 and wet SiO
2 can act as a very efficient reagent for the oxidative coupling of thiols under solvent free conditions. The reactions produce reasonably good yields under relatively mild conditions.[18] The compound is also used in the oxidation of aliphatic alcohols to their corresponding aldehydes and ketones in ZrCl
4/wet SiO
2 in solvent free conditions, again with relatively high yields.[19]
Safety
Ammonium dichromate is highly acutely toxic. It ranges from having a strong irritant effect on skin to causing severe chemical burns and skin corrosion. Inhalation of dust may be fatal. Ingestion results in death from multi-organ failure if not treated quickly.[20][21] Ascorbic acid may be a useful early treatment.[22]
Like many hexavalent chromium compounds it is a proven carcinogen, mutagen, and reproductive toxin.[1]
Incidents
In sealed containers, ammonium dichromate is likely to explode if heated.[15] In 1986, two workers were killed and 14 others injured at Diamond Shamrock Chemicals in Ashtabula, Ohio, when 2,000 lb (910 kg) of ammonium dichromate exploded as it was being dried in a heater.[23]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Sigma-Aldrich Co., Ammonium dichromate. Retrieved on 5 December 2025.
- ↑ 2.0 2.1 "SDS - Ammonium dichromate". ThermoFisher Scientific. 28 December 2021. https://www.fishersci.com/store/msds?partNumber=AC446921000&productDescription=AMMONIUM+DICHROMATE+ACS+100GR&vendorId=VN00032119&countryCode=US&language=en.
- ↑ "Ammonium Dichromate Volcano". Division of Chemical Education, Inc., American Chemical Society. 6 March 2012. https://www.chemedx.org/video/ammonium-dichromate-volcano.
- ↑ 4.0 4.1 Lewis, Richard J. (2007). Hawley's condensed chemical dictionary (15th ed.). Hoboken (N.J.): Wiley-Interscience. ISBN 978-0-471-76865-4.
- ↑ Keresztury, G.; Knop, O. (1982). "Infrared spectra of the ammonium ion in crystals. Part XII. Low-temperature transitions in ammonium dichromate, (NH4)2Cr2O7". Can. J. Chem. 60 (15): 1972–1976. doi:10.1139/v82-277.
- ↑ Shimizu, Takeo (February 1994). "Stabilizing Firework Compositions - (I) Minimum Solubility Law to Foresee the Degeneration, (II) A New Chemical Method of Magnesium Coating". Selected Pyrotechnic Publications of Dr. Takeo Shimizu, Part 1 (1985 to 1994), from the International Pyrotechnic Seminars. Whitewater, CO: Journal of Pyrotechnics. pp. 68–74. https://www.jpyro.co.uk/wp-content/uploads/Sh1_FULL_prn8.pdf#page=68. Retrieved 5 December 2025.
- ↑ Izzo, Attilio (1950) (in Italian). Pirotecnia E Fuochi Artificialli - Manuale pratico e ricettario per chi fabbrica fuochi artificiali e chi vende materie prime per usi pirotecnici. Milan, Italy: Ulricho Hoeppi. pp. 173–4, 230. https://www.mori.bz.it/Artifici%20ed%20Esplosivi/Izzo%20-%20pirotecnia.pdf. "Nido di serpenti (serpenti erba). - Sono giuochi fatti come al solito a forma di cono, avente la base di circa cm 4, contenenti una composizione che quando brucia produce come una zona erbosa da cui emergono i serpenti di faraone. Quando il cono è pieno di composizione simulante il verde della campagna, si aggiunge nel centro (alla base) la compo sizione polverizzata di solfocianuro di mercurio."
- ↑ Olivares-Pérez, A.; Ibarra-Torres, J.C.; Ortiz-Gutiérrez, M.; Pérez-Cortés, M.; Fuentes-Tapia, I. (December 2005). "Rosin (colophony) holograms sensitized with ammonium dichromate". Optical Materials 27 (12): 1825–1831. doi:10.1016/j.optmat.2004.07.018.
- ↑ Ponce de León, Yenisey; Montaño Flores, Beatriz; Ortiz-Gutiérrez, Mauricio; Ibarra Torres, Juan Carlos; Pérez Cortés, Mario (September 2021). "Real-time characterization of gelatin doped with potassium dichromate as a photosensitive material". Optik 242 (167310). doi:10.1016/j.ijleo.2021.167310. Bibcode: 2021Optik.24267310P.
- ↑ Calixto, Sergio; Alfaro-Gomez, Mariana (17 April 2025). "Dichromated Gelatin in Optics". Gels 11 (4): 298. doi:10.3390/gels11040298. PMID 40277734.
- ↑ Patnaik, Pradyot (2003). Handbook of inorganic chemicals. New York: McGraw-Hill. ISBN 0-07-049439-8.
- ↑ Havard, J. M.; Shim, S. Y.; Fr; eacute; chet, J. M. (1999). "Design of Photoresists with Reduced Environmental Impact. 1. Water-Soluble Resists Based on Photo-Cross-Linking of Poly(vinyl alcohol)". Chem. Mater. 11 (3): 719–725. doi:10.1021/cm980603y.
- ↑ Planned and performed by Marina Stojanovska, Miha Bukleski and Vladimir Petruševski, Department of Chemistry, FNSM, Ss. Cyril and Methodius University, Skopje, Macedonia.
- ↑ Neugebauer, C. A.; Margrave, J. L. (1957). "The Heat of Formation of Ammonium Dichromate". J. Phys. Chem. 61 (10): 1429–1430. doi:10.1021/j150556a040. Bibcode: 1957JPhCh..61.1429N.
- ↑ 15.0 15.1 15.2 Young, Jay A. (1 November 2005). "Ammonium Dichromate". Journal of Chemical Education 82 (11): 1617. doi:10.1021/ed082p1617. Bibcode: 2005JChEd..82.1617Y.
- ↑ G. A. P. Dalgaard; A. C. Hazell; R. G. Hazell (1974). "The Crystal Structure of Ammonium Dichromate, (NH4)2Cr2O7". Acta Chemica Scandinavica A28: 541–545. doi:10.3891/acta.chem.scand.28a-0541.
- ↑ Galwey, Andrew K.; Pöppl, László; Rajam, Sundara (1983). "A Melt Mechanism for the Thermal Decomposition of Ammonium Dichromate". J. Chem. Soc., Faraday Trans. 1 79 (9): 2143–2151. doi:10.1039/f19837902143.
- ↑ Shirini, F. (2003). "Solvent free oxidation of thiols by (NH4)2Cr2O7 in the presence of Mg(HSO4)2 and wet SiO2". Journal of Chemical Research 2003: 28–29. doi:10.3184/030823403103172823.
- ↑ Shirini, F. (2001). "ZrCl4/wet SiO2 promoted oxidation of alcohols by (NH4)2Cr2O7 in solution and solvent free condition". J. Chem. Research (S) 2001 (11): 467–477. doi:10.3184/030823401103168541.
- ↑ Sunilkumar, Menon; Ajith, Thekkuttuparambil; Parvathy, Vadakut (November 2014). "Acute ammonium dichromate poisoning in a 2 year-old child". Indian Journal of Critical Care Medicine 18 (11): 757–758. doi:10.4103/0972-5229.144024. PMID 25425845.
- ↑ Radhakrishnan, H; Gopi, M; Arumugam, A (2014). "Ammonium dichromate poisoning: A rare cause of acute kidney injury". Indian Journal of Nephrology 24 (6): 380–381. doi:10.4103/0971-4065.133781. PMID 25484533.
- ↑ Meert, Kathleen L; Ellis, Jolene; Aronow, Regine; Perrin, Eugene (October 1994). "Acute Ammonium Dichromate Poisoning". Annals of Emergency Medicine 24 (4): 748–750. doi:10.1016/S0196-0644(94)70288-8. PMID 8092606.
- ↑ Diamond, S. (19 January 1986). "Chemical in Blast Was Being Heated". The New York Times: p. 22. https://www.nytimes.com/1986/01/19/us/chemical-in-blast-was-being-heated.html.
External links
Template:Chromates and dichromates
 |

