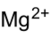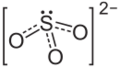Chemistry:Magnesium sulfite
From HandWiki
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Magnesium sulfite
| |||
| Other names
Magnesium sulphite
| |||
| Identifiers | |||
| |||
3D model (JSmol)
|
| ||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| UNII |
| ||
CompTox Dashboard (EPA)
|
| ||
| |||
| |||
| Properties | |||
| MgSO3 (anhydrous); MgSO3·6H2O | |||
| Molar mass | 104.368200 g/mol (anhydrous) 212.4599 g/mol (hexahydrate) | ||
| 5.2g/L at 298.2K (hexahydrate) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
Magnesium sulfite is the magnesium salt of sulfurous acid with the formula MgSO3. Its most common hydrated form has 6 water molecules making it a hexahydrate, MgSO3·6H2O. When heated above 40 °C (104 °F), it is dehydrated to magnesium sulfite trihydrate, or MgSO3·3H2O.[1] The anhydrous form is hygroscopic, meaning that it readily absorbs water from the air.
Solubility tables of MgSO3 hydrates PDF: [2]
See also
- Calcium sulfite
- Magnesium sulfate (Epsom salt)
References
- ↑ Nývlt, J., "Solubilities of Magnesium Sulfite," Journal of Thermal Analysis and Calorimetry, Volume 66, Number 2 / November, 2001
- ↑ [1], H.D.Lutz, Dept. of Chemistry, University of Slegen.
 |