Chemistry:Potassium sorbate
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Potassium (2E,4E)-hexa-2,4-dienoate | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C6H7KO2 | |
| Molar mass | 150.218 g·mol−1 |
| Appearance | White crystals |
| Odor | Yes |
| Density | 1.363 g/cm3 |
| Melting point | 270 °C (518 °F; 543 K) decomposes |
| 58.5 g/100 mL (100 °C) | |
| Solubility in other solvents |
|
| Hazards | |
| Safety data sheet | Fisher Scientific |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| HH319Script error: No such module "Preview warning".Category:GHS errors | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
4920 mg/kg (oral, rat)[3] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
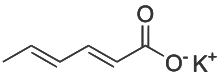
Potassium sorbate is the potassium salt of sorbic acid, structural formula CH3CH=CH−CH=CH−CO2K. It is a white salt that is very soluble in water (58.2% at 20 °C). It is primarily used as a food preservative (E number 202).[4] Potassium sorbate is effective in a variety of applications including food, wine, and personal care products. While sorbic acid occurs naturally in rowan and hippophae berries, virtually all of the world's supply of sorbic acid, from which potassium sorbate is derived, is manufactured synthetically.
Production
Potassium sorbate is produced industrially by neutralizing sorbic acid with potassium hydroxide. The precursor sorbic acid is produced in a two-step process via the condensation of crotonaldehyde and ketene.[5][6][7]
Uses
Potassium sorbate is used to inhibit molds and yeasts in many foods, such as cheese, wine, yogurt, dried meats, apple cider, dried fruits, soft drinks and fruit drinks, and baked goods.[8] It can also be found in the ingredients list of many dried fruit products. In addition, herbal dietary supplement products generally contain potassium sorbate, which acts to prevent mold and microbes and to increase shelf life. It is used in quantities at which no adverse health effects are known, over short periods of time.[9] Labeling of this preservative on ingredient statements reads as "potassium sorbate" or "E202".
Also, it is used in many personal care products to inhibit the development of microorganisms to increase shelf stability. Some manufacturers use this preservative as a replacement for parabens.
Also known as "wine stabilizer", potassium sorbate produces sorbic acid when added to wine. It serves two purposes:
- When active fermentation has ceased and the wine is racked for the final time after clearing, potassium sorbate renders any surviving yeast incapable of multiplying. Yeast living at that moment can continue fermenting any residual sugar into CO2 and alcohol, but when they die, no new yeast will be present to cause future fermentation. When a wine is sweetened before bottling, potassium sorbate is used to prevent refermentation when used in conjunction with potassium metabisulfite. It is primarily used with sweet wines, sparkling wines, and some hard ciders, but may be added to table wines, which may not maintain their clarity after fining.
- It also inhibits bacteria, especially Clostridium botulinum.[10] Tube feeding of potassium sorbate reduces the amount of pathogenic bacteria in the stomach.[11]
Some molds (notably some Trichoderma and Penicillium strains) and yeasts are able to detoxify sorbates by decarboxylation, producing piperylene (1,3-pentadiene). The pentadiene manifests as a typical odor of kerosene or petroleum.[12]
Toxicology
In pure form, potassium sorbate is a skin, eye, and respiratory irritant.[13][14] Concentrations up to 0.5% are not significant skin irritants.[15]
As a food additive, potassium sorbate is used as a preservative in concentrations of 0.025%–0.1%,[16] which in a 100 g serving yields an intake of 25–100 mg. In the United States, no more than 0.1% is allowed in fruit butters, jellies, preserves, and related products.[17] Up to 0.4% has been studied in low-salt, naturally-fermented pickles, and when combined with calcium chloride, 0.2% made "good quality pickles."[18] Potassium sorbate has about 74% of sorbic acid's anti-microbial activity.[16] When calculated as sorbic acid, 0.3% is allowed in "cold pack cheese food."[19] The upper pH limit for effectiveness is 6.5.[16]
The maximum acceptable daily intake for human consumption is 25 mg/kg, or 1.75 g daily for an average adult (70 kg).[9][20] Under some conditions, particularly at high concentrations or when combined with nitrites, potassium sorbate has shown genotoxic activity in vitro.[20]
Three studies conducted in the 1970s did not find it to have any carcinogenic effects in rats.[21][22][23] However, a 2009-2023 cohort study of 105,260 participants in France found a 14% increase in overall cancer risk and 26% increase in breast cancer risk associated with high consumption of potassium sorbate.[24]
See also
References
- ↑ Merck Index, 11th Edition, 7661.
- ↑ |SIAL&N5=SEARCH_CONCAT_PNO|BRAND_KEY&F=SPEC Potassium sorbate at Sigma-Aldrich.
- ↑ Sax's Dangerous Properties of Industrial Materials. Wiley. 2004-10-15. p. 3043. doi:10.1002/0471701343. ISBN 978-0-471-47662-7.
- ↑ Nordic Food Additive Database Nordic Working Group on Food Toxicology and Risk Assessment.
- ↑ Erich Lück, Martin Jager and Nico Raczek "Sorbic Acid" in Ullmann's Encyclopedia of Industrial Chemistry, 2011, Wiley-VCH, Weinheim. doi:10.1002/14356007.a24_507.pub2
- ↑ patent process for commercial potassium sorbate
- ↑ "Agricultural Marketing Service". http://www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELPRDC5067082.
- ↑ Erich Lück, Martin Jager and Nico Raczek "Sorbic Acid" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2000.doi:10.1002/14356007.a24_507
- ↑ 9.0 9.1 "036. Sorbate, potassium (FAO Nutrition Meetings Report Series 40abc)". Inchem.org. http://www.inchem.org/documents/jecfa/jecmono/40abcj15.htm.
- ↑ "Effects of various concentrations of sodium nitrite and potassium sorbate on Clostridium botulinum toxin production in commercially prepared bacon". Journal of Food Science 45 (5): 1285–1292. September 1980. doi:10.1111/j.1365-2621.1980.tb06539.x.
- ↑ Tulamait, Aiman; Laghi, Franco; Mikrut, Kathleen et al. (2005). "Potassium sorbate reduces gastric colonization in patients receiving mechanical ventilization". J. Crit. Care 20 (3): 281–287. doi:10.1016/j.jcrc.2005.03.002. PMID 16253799.
- ↑ The Soft Drinks Companion – A technical handbook for the beverage industry, Chapter 10.
- ↑ "Potassium Sorbate". Chem One, Ltd.. 2010-06-14. http://www.chemone.com/default/msds/Potassium%20sorbate.pdf.
- ↑ "Sorbic Acid - National Library of Medicine HSDB Database". National Institute of Health. https://www.nlm.nih.gov/toxnet/index.html.
- ↑ Elder, R. E. (1988). "Final report on the safety assessment of sorbic acid and potassium sorbate.". J Am Coll Toxicol 7 (6): 837–880. doi:10.3109/10915818809078711. https://pdfs.semanticscholar.org/bbbe/1de833dfb7ebaf3959eaace0762e9b9ffb9b.pdf. Retrieved May 26, 2018.
- ↑ 16.0 16.1 16.2 "Potassium sorbate - Agricultural Marketing Service - USDA". U.S. government. https://www.ams.usda.gov/sites/default/files/media/P%20Sor%20technical%20advisory%20panel%20report.pdf. "Potassium sorbate is effective against yeasts, molds, and select bacteria, and is widely used at 0.025 to 0.10 % levels in cheeses, dips, yogurt, sour cream, bread, cakes, pies and fillings, baking mixes, doughs, icings, fudges, toppings, beverages, margarine, salads, fermented and acidified vegetables, olives, fruit products, dressings, smoked and salted fish, confections and mayonnaise."
- ↑ "21 CFR 150 - Fruit butters, jellies, preserves, and related products". Government Printing Office. April 1, 2011. https://www.gpo.gov/fdsys/pkg/CFR-2011-title21-vol2/pdf/CFR-2011-title21-vol2-part150.pdf.
- ↑ Guillou, A.A.; Floros, J.D.; Cousin, M.A. (1992). "Calcium Chloride and Potassium Sorbate Reduce Sodium Chloride used during Natural Cucumber Fermentation and Storage". Journal of Food Science 57 (6): 1364–1368. doi:10.1111/j.1365-2621.1992.tb06859.x. ISSN 0022-1147.
- ↑ "21 CFR 133 - Cheeses and related cheese products". Government Printing Office. April 1, 2011. https://www.gpo.gov/fdsys/pkg/CFR-2011-title21-vol2/pdf/CFR-2011-title21-vol2-part133.pdf.
- ↑ 20.0 20.1 Carocho, Márcio; Barreiro, Maria Filomena; Morales, Patricia; Ferreira, Isabel C. F. R. (2014). "Adding Molecules to Food, Pros and Cons: A Review on Synthetic and Natural Food Additives". Comprehensive Reviews in Food Science and Food Safety 13 (4): 377–399. doi:10.1111/1541-4337.12065. PMID 33412697.
- ↑ Hendy, R. J.; Hardy, J.; Gaunt, I. F.; Kiss, I. S.; Butterworth, K. R. (1976). "Long-term toxicity studies of sorbic acid in mice". Food and Cosmetics Toxicology 14 (5): 381–386. doi:10.1016/S0015-6264(76)80173-3. PMID 1010505.
- ↑ Gaunt, I. F.; Butterworth, K. R.; Hardy, J.; Gangolli, S. D. (1975). "Long-term toxicity of sorbic acid in the rat". Food and Cosmetics Toxicology 13 (1): 31–45. doi:10.1016/0015-6264(75)90080-2. PMID 1123201.
- ↑ Mason, P. L.; Gaunt, I. F.; Hardy, J.; Kiss, I. S.; Butterworth, K. R.; Gangolli, S. D. (1976). "Long-term toxicity of parasorbic acid in rats". Food and Cosmetics Toxicology 14 (5): 387–394. doi:10.1016/S0015-6264(76)80174-5. PMID 1010506.
- ↑ Hasenböhler, Anaïs; Javaux, Guillaume; de la Garanderie, Marie Payen de; de Edelenyi, Fabien Szabo; Yvroud-Hoyos, Paola; Agaësse, Cédric; De Sa, Alexandre; Huybrechts, Inge et al. (2026). "Intake of food additive preservatives and incidence of cancer: results from the NutriNet-Santé prospective cohort". BMJ 392 (8479). doi:10.1136/bmj-2025-084917.
- ↑ Saltmarsh, Mike (2015-03-15). "Recent trends in the use of food additives in the United Kingdom". Journal of the Science of Food and Agriculture 95 (4): 649–652. doi:10.1002/jsfa.6715. ISSN 1097-0010. PMID 24789520. Bibcode: 2015JSFA...95..649S. "... the preservative used in the study, sodium benzoate, has been replaced by potassium sorbate in the majority of soft drinks.".
 |

