Chemistry:Potassium metabisulfite
| |
| |

| |
| Names | |
|---|---|
| Other names
Potassium pyrosulfite
Dipotassium disulfite Potassium metabisulphite Dipotassium disulphite | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
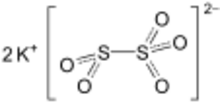
| K2O5S2 | |
| Molar mass | 222.31 g·mol−1 |
| Appearance | White crystalline powder |
| Odor | Pungent (sulfur dioxide) |
| Density | 2.34 g/cm3 (solid) |
| Melting point | 190 °C (374 °F; 463 K) decomposes |
| 450 g/L (20 °C) | |
| Solubility | Insoluble in ethanol |
| Hazards | |
| Main hazards | Irritant, asthma risk |
| Safety data sheet | ICSC 1175 |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| H315, H318, H335 | |
| P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P310, P312, P321, P332+313, P362, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions
|
Potassium bisulfite Potassium sulfite |
Other cations
|
Sodium metabisulfite |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
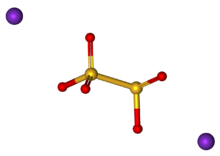
Potassium metabisulfite, K2S2O5, also known as potassium pyrosulfite, is a white crystalline powder with a pungent odour. It is mainly used as an antioxidant or chemical sterilant.[1] As a disulfite, it is chemically very similar to sodium metabisulfite, with which it is sometimes used interchangeably. Potassium metabisulfite has a monoclinic crystal structure.
Preparation and reactions
Potassium metabisulfite can be prepared by treating a solution of potassium hydroxide with sulfur dioxide.[2]
- 2 SO2 + 2 KOH → K2S2O5 + H2O
It decomposes at 190 °C, yielding potassium sulfite and sulfur dioxide:
- K2S2O5 → K2SO3 + SO2
Uses
It is used as a food additive, also known as E224.[3] It is restricted in use and may cause allergic reactions in some sensitive persons.[4]
Wine
Potassium metabisulfite is a common wine or must additive, in which it forms sulfur dioxide (SO2). Sulfur dioxide is a disinfectant. It also acts as a potent antioxidant, protecting both the color and delicate flavors of wine.
A high dose would be 3 grams of potassium metabisulfite per six-gallon bucket of must (yielding roughly 75 ppm of SO2) prior to fermentation; then 6 grams per six-gallon bucket (150 ppm of SO2) at bottling. Some countries regulate the SO2 content of wines.[5]
Winemaking equipment is sanitized by spraying with a 1% SO2 (2 tsp potassium metabisulfite per L) solution.
Beer
Potassium metabisulfite is sometimes used in the brewing industry to inhibit the growth of wild bacteria and fungi. This step is called 'stabilizing'. It is also used to neutralize monochloramine from tap water. It is used both by homebrewers and commercial brewers alike. It is not used as much for brewing beer, because the wort is almost always boiled, which kills most microorganisms.
Other uses
- Potassium metabisulfite is sometimes added to lemon juice as a preservative.
- Potassium metabisulfite is used in the textile industry for dyeing and cotton printing.
- Potassium metabisulfite is sometimes used to precipitate gold from solution in aqua regia (as an alternative to sodium sulfite).
- It is a component of certain photographic developers and solutions used in photographic fixing.[6]
- It is used as a bleaching agent in the production of coconut cream.
- It is used in some pickles as a preservative.
- It is used in tint etching iron-based metal samples for microstructural analysis. [7]
- It is used in Aam papad as a preservative.
See also
References
- ↑ Barberá, José Jiménez; Metzger, Adolf; Wolf, Manfred (2000). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a25_477.
- ↑ Johnstone, H. F. (1946). "Sulfites and Pyrosulfites of the Alkali Metals". Inorganic Syntheses. Inorganic Syntheses. 2. pp. 162–167. doi:10.1002/9780470132333.ch49. ISBN 9780470132333.
- ↑ List of E-number food additives
- ↑ Metcalfe, Dean D.; Simon, Ronald A. (2003). Food allergy: adverse reactions to food and food additives. Malden, MA: Wiley-Blackwell. pp. 324–339. ISBN 978-0-632-04601-0.
- ↑ "The Truth About Sulfites in Wine & the Myths of Red Wine Headaches". https://www.thekitchn.com/the-truth-about-sulfites-in-wine-myths-of-red-wine-headaches-100878.
- ↑ "Potassium Metabisulfite". http://encyclopedia2.thefreedictionary.com/potassium+metabisulfite.
- ↑ "Color Metallography". 2011-05-04. https://vacaero.com/information-resources/metallography-with-george-vander-voort/991-color-metallography.html.
 |


