Chemistry:Tetramethylethylene
From HandWiki
Template:Chembox DeltaHvap
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
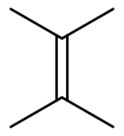
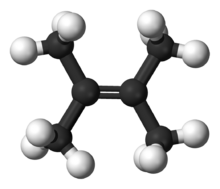
2,3-Dimethylbut-2-ene | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C6H12 | |
| Molar mass | 84.162 g·mol−1 |
| Appearance | colorless liquid |
| Density | 0.7075 g/cm3 (20 °C) |
| Melting point | −74.6 °C (−102.3 °F; 198.6 K) |
| Boiling point | 73.3 °C (163.9 °F; 346.4 K) |
| Critical point (T, P) | 521.0(9) K, 3.4(1) MPa[1] |
| insoluble | |
| Solubility | soluble in ethanol, ether, acetone, chloroform[1] |
| −65.9×10−6 cm3·mol−1[1] | |
Refractive index (nD)
|
1.4122 (20 °C)[1] |
| Thermochemistry[1] | |
Heat capacity (C)
|
174.7 J·mol−1·K−1 |
Std molar
entropy (S |
270.2 J·mol−1·K−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−101.4 kJ·mol−1 (liquid) −68.1 kJ·mol−1 (gas) |
Enthalpy of fusion (ΔfH⦵fus)
|
6.45 kJ·mol−1 (at melting point) |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| H225, H304 | |
| P210, P233, P240, P241, P242, P243, P280, P301+310, P303+361+353, P331, P370+378, P403+235, P405, P501 | |
| Flash point | −8 °C |
| 401 °C[1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Tetramethylethylene is a hydrocarbon with the formula Me2C=CMe2 (Me = methyl). A colorless liquid, it is the simplest tetrasubstituted alkene.
Synthesis
It can be prepared by base-catalyzed isomerization of 2,3-dimethyl-1-butene.[2] Another route involves direct dimerization of propylene.[3] It can also be produced by photolysis of tetramethylcyclobutane-1,3-dione.[4]
Reactions
Tetramethylethylene forms metal-alkene complexes with low-valent metals and reacts with diborane to give the monoalkyborane known as thexylborane.[5][6]
Oxidation gives pinacol.
It is a precursor to the herbicide fenpropathrin.[3]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data.. William M. Haynes, David R. Lide, Thomas J. Bruno (2016-2017, 97th ed.). Boca Raton, Florida. 2016. ISBN 978-1-4987-5428-6. OCLC 930681942. https://www.worldcat.org/oclc/930681942.
- ↑ Hattori, Hideshi (2001). "Solid base catalysts: Generation of basic sites and application to organic synthesis". Applied Catalysis A: General 222 (1–2): 247–259. doi:10.1016/S0926-860X(01)00839-0.
- ↑ 3.0 3.1 Olivier-Bourbigou, H.; Breuil, P. A. R.; Magna, L.; Michel, T.; Espada Pastor, M. Fernandez; Delcroix, D. (2020). "Nickel Catalyzed Olefin Oligomerization and Dimerization". Chemical Reviews 120 (15): 7919–7983. doi:10.1021/acs.chemrev.0c00076. PMID 32786672. https://hal-ifp.archives-ouvertes.fr/hal-02954532/file/Nickel%20Catalyzed%20Olefin.pdf.
- ↑ Nicholas J. Turro, Peter A. Leermakers, George F. Vesley (1967). "Cyclohexylidenecyclohexane". Organic Syntheses 47: 34. doi:10.15227/orgsyn.047.0034.
- ↑ Negishi, Ei-Ichi; Brown, Herbert C. (1974). "Thexylborane-A Highly Versatile Reagent for Organic Synthesis via Hydroboration". Synthesis 1974 (2): 77–89. doi:10.1055/s-1974-23248.
- ↑ Giordano, G.; Crabtree, R. H. (1990). "Di‐μ‐Chloro‐Bis(η 4 ‐1,5‐Cyclooctadiene)‐Dirhodium(I)". Di-μ-Chloro-Bis(η4-1,5-Cyclooctadiene)-Dirhodium(I). Inorganic Syntheses. 28. pp. 88–90. doi:10.1002/9780470132593.ch22. ISBN 9780471526193.
 |
