Chemistry:Triethyl phosphate
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Triethyl phosphate | |
| Other names
Phosphoric acid triethyl ester
Phosphoric ester (archaic) Flame retardant TEP[2] Tris(ethyl) phosphate Triethoxyphosphine oxide Ethyl phosphate (neutral) | |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | TEP |
| ChEBI | |
| ChemSpider | |
| DrugBank | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| (CH 3CH 2) 3PO 4 | |
| Molar mass | 182.156 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.072 g/cm3 |
| Melting point | −56.5 °C (−69.7 °F; 216.7 K) |
| Boiling point | 215 °C (419 °F; 488 K) |
| Miscible | |
| −125.3·10−6 cm3/mol | |
| Hazards | |
| Safety data sheet | [3] |
| NFPA 704 (fire diamond) | |
| Flash point | 107 °C (225 °F; 380 K) |
| Related compounds | |
Related compounds
|
Trimethyl phosphate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
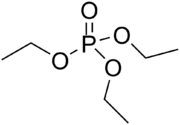
Triethyl phosphate is an organic chemical compound with the formula (CH
3CH
2)
3PO
4 or Et
3PO
4, where Et is ethyl. It is a colorless liquid. It is the triethyl ester of phosphoric acid and can be called "phosphoric acid, triethyl ester".
Its primary uses are as an industrial catalyst (in acetic anhydride synthesis), a polymer resin modifier, and a plasticizer (e.g. for unsaturated polyesters). In smaller scale it is used as a solvent for e.g. cellulose acetate, flame retardant, an intermediate for pesticides and other chemicals, stabilizer for peroxides, a strength agent for rubber and plastic including vinyl polymers and unsaturated polyesters, etc.[4]
History
It was studied for the first time by French chemist Jean Louis Lassaigne in the early 19th century.
See also
- Franz Anton Voegeli
References
- ↑ "Zhangjiagang Shunchang Chemical Co., Ltd". Triethylphosphate. Archived from the original on December 17, 2004. https://web.archive.org/web/20041217051620/http://www.shunchangchem.com/template/produe90.htm. Retrieved June 13, 2009.
- ↑ https://pubchem.ncbi.nlm.nih.gov/compound/Triethyl-Phosphate
- ↑ https://assets.thermofisher.com/DirectWebViewer/private/document.aspx?prd=ALFAA40001~~PDF~~MTR~~AGHS~~EN~~2024-03-27%2004:53:15~~Triethyl%20phosphate~~
- ↑ Triethylphosphate, International Programme on Chemical Safety
 |

