Chemistry:White phosphorus
 White phosphorus sample with a chunk removed from the corner to expose un-oxidized material
| |
 Tetraphosphorus molecule
| |
| Names | |
|---|---|
| IUPAC names
White phosphorus
tetrahedro-Tetraphosphorus | |
| Systematic IUPAC name
1,2,3,4-Tetraphosphatricyclo[1.1.0.02,4]butane | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| 1856 | |
PubChem CID
|
|
| UN number | 1381 |
| |
| |
| Properties | |
| P4 | |
| Molar mass | 123.895 g·mol−1 |
| Density | 1.82 g/cm3 |
| Melting point | 44.1 °C; 111.4 °F; 317.3 K |
| Boiling point | 280 °C; 536 °F; 553 K |
| Hazards[1] | |
| GHS pictograms |    
|
| GHS Signal word | Danger |
| HH250Script error: No such module "Preview warning".Category:GHS errors, HH300+H330Script error: No such module "Preview warning".Category:GHS errors, HH314Script error: No such module "Preview warning".Category:GHS errors, HH400Script error: No such module "Preview warning".Category:GHS errors | |
| PP210Script error: No such module "Preview warning".Category:GHS errors, PP222Script error: No such module "Preview warning".Category:GHS errors, PP260Script error: No such module "Preview warning".Category:GHS errors, PP264Script error: No such module "Preview warning".Category:GHS errors, PP270Script error: No such module "Preview warning".Category:GHS errors, PP271Script error: No such module "Preview warning".Category:GHS errors, PP273Script error: No such module "Preview warning".Category:GHS errors, PP280Script error: No such module "Preview warning".Category:GHS errors, PP284Script error: No such module "Preview warning".Category:GHS errors, PP301+P310+P330Script error: No such module "Preview warning".Category:GHS errors, PP301+P330+P331Script error: No such module "Preview warning".Category:GHS errors, PP303+P361+P353Script error: No such module "Preview warning".Category:GHS errors, PP304+P340+P310Script error: No such module "Preview warning".Category:GHS errors, PP305+P351+P338+P310Script error: No such module "Preview warning".Category:GHS errors, PP335+P334Script error: No such module "Preview warning".Category:GHS errors, PP363Script error: No such module "Preview warning".Category:GHS errors, PP370+P378Script error: No such module "Preview warning".Category:GHS errors, PP391Script error: No such module "Preview warning".Category:GHS errors, PP403+P233Script error: No such module "Preview warning".Category:GHS errors, PP405Script error: No such module "Preview warning".Category:GHS errors, PP422Script error: No such module "Preview warning".Category:GHS errors, PP501Script error: No such module "Preview warning".Category:GHS errors | |
| NFPA 704 (fire diamond) | |
Threshold limit value (TLV)
|
0.1 mg/m3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
White phosphorus, yellow phosphorus, or simply tetraphosphorus (P4) is an allotrope of phosphorus. It is a translucent waxy solid that quickly yellows in light (due to its photochemical conversion into red phosphorus),[2] and impure white phosphorus is for this reason called yellow phosphorus. White phosphorus is the first allotrope of phosphorus that was discovered, isolated for the first time in 1669 by Henning Brand.[3]
When in an oxygen-containing atmosphere, it will exhibit a faint green glow in the absence of light. White phosphorus is also highly flammable and pyrophoric (self-igniting) upon contact with air. It is toxic, causing severe liver damage upon ingestion and phossy jaw from chronic ingestion or inhalation. The combustion of this form has a characteristic garlic odor, and samples are commonly coated with white "diphosphorus pentoxide", which consists of P
4O
10 tetrahedra with oxygen inserted between the phosphorus atoms and at their vertices. White phosphorus is only slightly soluble in water and can be stored under water. P
4 is soluble in benzene, oils, carbon disulfide, and disulfur dichloride.
Structure
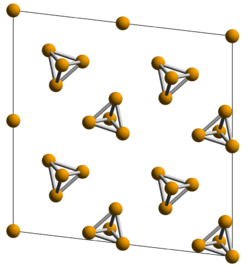
White phosphorus exists as molecules of four phosphorus atoms in a tetrahedral structure, with each phosphorus atom making three phosphorus—phosphorus single bonds for a total of six P-P bonds per tetrahedron. The tetrahedral arrangement results in ring strain and instability.[4] White phosphorus can take on one of two crystal allotropes that interechange reversibly above 195.2 K (−78.0 °C; −108.3 °F). The element's standard state is the body-centered cubic α form, which is metastable under standard conditions.[4] The β form is believed to have a hexagonal crystal structure.[5]
Molten and gaseous white phosphorus are also composed of these tetrahedra until 800 °C (1,500 °F; 1,100 K) when they start decomposing into P2 molecules.[6] The P4 molecule in the gas phase has a P-P bond length of rg = 2.1994(3) Å as was determined by gas electron diffraction.[7] The β form of white phosphorus contains three slightly different P4 molecules, i.e. 18 different P-P bond lengths — between 2.1768(5) and 2.1920(5) Å. The average P-P bond length is 2.183(5) Å.[6]
Chemical properties
Despite white phosphorus not being the most stable allotrope of phosphorus (see: black phosphorus), it is still used as the reference state for solid phosphorus and defined to have a standard enthalpy of formation of zero. This is because it is much easier to handle and purify for the purposes of collecting reference thermodynamic data.
In basic media, white phosphorus spontaneously disproportionates to phosphine and various phosphorus oxyacid salts.[8]
Many reactions of white phosphorus involve insertion into the P-P bonds, such as the reactions with oxygen, sulfur, phosphorus tribromide and the NO+ ion.
It ignites spontaneously in air at about 50 °C (122 °F), and at much lower temperatures if finely divided (due to melting-point depression). Phosphorus reacts with oxygen, usually forming two oxides depending on the amount of available oxygen: P
4O
6 (phosphorus trioxide) when reacted with a limited supply of oxygen, and P
4O
10 when reacted with excess oxygen. On rare occasions, P
4O
7, P
4O
8, and P
4O
9 are also formed, but in small amounts. This combustion gives phosphorus(V) oxide:
- P
4 + 5 O
2 → P
4O
10
Production and applications
The white allotrope can be produced using several methods. In the industrial process, phosphate rock is heated in an electric or fuel-fired furnace in the presence of carbon and silica.[9] Elemental phosphorus is then liberated as a vapour and can be collected under phosphoric acid. An idealized equation for this carbothermal reaction is shown for calcium phosphate (although phosphate rock contains substantial amounts of fluoroapatite, which would also form silicon tetrafluoride):
- 2 Ca
3(PO
4)
2 + 6 SiO
2 + 10 C → 6 CaSiO
3 + 10 CO + P
4
In this way, an estimated 750,000 tons were produced in 1988.[10]
Most (83% in 1988) white phosphorus is used as a precursor to phosphoric acid, half of which is used for food or medical products where purity is important. The other half is used for detergents.[needs update] Much of the remaining 17% is mainly used for the production of chlorinated compounds phosphorus trichloride, phosphorus oxychloride, and phosphorus pentachloride:[11]
- P
4 + 10Cl
2 → 4PCl
5
Other products derived from white phosphorus include phosphorus pentasulfide and various metal phosphides.[10]
White phosphorus is also used to make glyphosate, the active ingredient in Roundup herbicide and munitions. In the United States, Bayer is the sole producer of elemental phosphorus.[12]
Other polyhedrane analogues
Although white phosphorus forms the tetrahedron, the simplest possible Platonic solid, no other polyhedral phosphorus clusters are known.[13] White phosphorus converts to the thermodynamically more stable red allotrope, but that allotrope is not composed of isolated polyhedra.
A cubane-type cluster, in particular, is unlikely to form,[13] and the closest approach is the half-phosphorus compound P
4(CH)
4, produced from phosphaalkynes.[14] Other clusters are more thermodynamically favorable, and some have been partially formed as components of larger polyelemental compounds.[13]
Safety
White phosphorus is acutely toxic, with a lethal dose of 50-100 mg (1 mg/kg body weight). Its mode of action is not known but is thought to involve its reducing properties, possibly forming intermediate reducing compounds such as hypophosphite, phosphite, and phosphine. It damages the liver, kidneys, and other organs before eventually being metabolized to non-toxic phosphate. Chronic low-level exposure leads to tooth loss and phossy jaw which appears to be caused by the formation of amino bisphosphonates.[10][15][16]
White phosphorus is used as a weapon because it is pyrophoric. For the same reasons, it is dangerous to handle. Measures are taken to protect samples from air since it will react with oxygen at ambient temperatures, and even in small samples this can lead to self-heating and eventual combustion. There are anecdotal reports of problems for beachcombers who may collect washed-up samples while unaware of their true nature.[17][18]
References
- ↑ Sigma-Aldrich Co., Phosphorus, white.
- ↑ "White phosphorus" (in en). https://www.acs.org/molecule-of-the-week/archive/w/white-phosphorus.html.
- ↑ Weeks, Mary Elvira (1932). "The discovery of the elements. II. Elements known to the alchemists". Journal of Chemical Education 9 (1): 11. doi:10.1021/ed009p11. Bibcode: 1932JChEd...9...11W.
- ↑ 4.0 4.1 Housecroft, C. E.; Sharpe, A. G. (2004). Inorganic Chemistry (2nd ed.). Prentice Hall. p. 392. ISBN 978-0-13-039913-7.
- ↑ Durif, A.; Averbuch-Pouchot, M.T. (1996). Topics in phosphate chemistry. Singapore [u.a.]: World Scientific. p. 3. ISBN 978-981-02-2634-3.
- ↑ 6.0 6.1 Simon, Arndt; Borrmann, Horst; Horakh, Jörg (1997). "On the Polymorphism of White Phosphorus". Chemische Berichte 130 (9): 1235–1240. doi:10.1002/cber.19971300911.
- ↑ Cossairt, Brandi M.; Cummins, Christopher C.; Head, Ashley R.; Lichtenberger, Dennis L.; Berger, Raphael J. F.; Hayes, Stuart A.; Mitzel, Norbert W.; Wu, Gang (2010-06-01). "On the Molecular and Electronic Structures of AsP3 and P4". Journal of the American Chemical Society 132 (24): 8459–8465. doi:10.1021/ja102580d. ISSN 0002-7863. PMID 20515032. Bibcode: 2010JAChS.132.8459C.
- ↑ Engel, Robert; Cohen, JaimeLee Iolani (2004). Synthesis of Carbon-Phosphorus Bonds (2nd ed.). Boca Raton: CRC Press. §2.3. ISBN 0-8493-1617-0.
- ↑ Threlfall, R.E., (1951). 100 years of Phosphorus Making: 1851–1951. Oldbury: Albright and Wilson Ltd
- ↑ 10.0 10.1 10.2 Diskowski, Herbert; Hofmann, Thomas (2000). "Phosphorus". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a19_505. ISBN 978-3-527-30385-4.
- ↑ (in English) Chemistry Part I Class XII (Reprinted ed.). India: NCERT. January 2019. pp. 177. ISBN 978-81-7450-648-1. https://ncert.nic.in/ncerts/l/lech107.pdf.
- ↑ Tabuchi, Hiroko (8 March 2026). "A Trump Order Protected a Weedkiller. And Also a Weapon of War.". New York Times. https://www.nytimes.com/2026/03/08/climate/bayer-white-phosphate-glyphosate-roundup-trump-executive-order-munition.html.
- ↑ 13.0 13.1 13.2 Corbridge, D. E. C. (1995) "Phosphorus: An Outline of its Chemistry, Biochemistry, and Technology" 5th Edition Elsevier: Amsterdam. § 4.1.12. ISBN 0-444-89307-5.
- ↑ Streubel, Rainer (1995). "Phosphaalkyne Cyclooligomers: From Dimers to Hexamers—First Steps on the Way to Phosphorus–Carbon Cage Compounds". Angewandte Chemie International Edition in English 34 (4): 436–438. doi:10.1002/anie.199504361.
- ↑ Obscurants, National Research Council (US) Subcommittee on Military Smokes and (1999), "White Phosphorus Smoke" (in en), Toxicity of Military Smokes and Obscurants: Volume 2 (National Academies Press (US)), https://www.ncbi.nlm.nih.gov/books/NBK224560/, retrieved 2025-02-04
- ↑ Toxicological Profile for White Phosphorus (Report). U.S. Department of Health and Human Services. September 1997. https://www.atsdr.cdc.gov/toxprofiles/tp103.pdf. Retrieved 2025-02-05.
- ↑ Winter, George (1 October 2017). "A dangerous guide to beachcombing". https://www.chemistryworld.com/news/a-dangerous-guide-to-beachcombing/3008056.article.
- ↑ Staudenmaier, Rebecca (May 8, 2017). "Woman mistakes WWII-era munition for precious stone on German beach | DW | 05.08.2017". https://www.dw.com/en/woman-mistakes-wwii-era-munition-for-precious-stone-on-german-beach/a-39977702.
 |

