Chemistry:Edible packaging
Edible packaging refers to packaging which is edible and biodegradable.
Edible food packaging
Several manufacturers are developing or producing food packaging that is edible. One example is made based on the seaweed, Eucheuma cottonii.[1][2]
Traditional water containers
About 50 billion single-use plastic water bottles made of polyethylene terephthalate (PET) are produced in the United States each year, and most are discarded.[3] According to the National Association for PET Container Resources, the recycling rate for PET has held steady at 31% since 2013.[4]
Polyesters like PET can be broken down through hydrolytic degradation: the ester linkage is cut by a water molecule. The reaction proceeds differently in acidic or alkaline conditions, but works best at temperatures between 200 - 300 °C. Under environmental conditions the process is undetectably slow.[5]
PET is considered to be essentially non-biodegradable, with plastic bottles estimated to take as long as 450 years to decompose.[6] Because of this, other packaging materials are being sought.
Calcium alginate gel
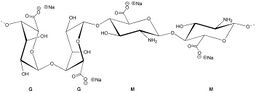
Alginates are the natural product of brown algae and have been used extensively in wound dressing, drug delivery and tissue engineering, as well as food applications.[7][8][9] Sodium alginate is an unbranched copolymer of 1,4-linked-β-d-mannuronate (M) and α-l-guluronate (G) sugars.
Sodium alginate (NaAlg) coagulates when exposed to calcium chloride (CaCl2) and forms calcium alginate (CaAlg2) and sodium chloride (NaCl), according to the following reaction:
2NaAlg + CaCl2 → CaAlg2 + 2NaCl
Safety and biodegradability
The biocompatibility of alginate gels has been studied extensively and their safety for consumption is well established.[10][11] As natural polysaccharides resistant to breakdown by human digestive enzymes, alginates are classified as dietary fiber. Although undigested if eaten, the Ooho capsule will gradually decompose as the calcium diffuses out of the gel matrix in the reverse of the reaction above.[12]
CaAlg2 + 2NaCl → 2NaAlg + CaCl2
Because it is a single-strand polymer, alginate can be depolymerized (broken into smaller units) by a variety of chemical reactions. Both acid and alkaline mechanisms can break down the linkages between the mannuronate (M) and guluronate (G) monomers. Free radical oxidation is another way the alginate can be degraded in the environment. Many bacterial species produce an enzyme (alginate lyase) which can break the molecule down into single sugar components, which can act as an energy source for the organism.[13]
See also
- Biodegradation
- Calcium alginate
- Creative Commons license
- Low plastic water bottle
- Molecular gastronomy
- Spherification
- Water bottle
- Starch-based foam peanuts
References
- ↑ In race for sustainable alternative to plastic, Indonesia bets on seaweed
- ↑ Evoware, the algae based packaging that disappears
- ↑ "Why Tap Water Is Better.". 2010-03-13. http://news.nationalgeographic.com/news/2010/03/100310/why-tap-water-is-better/.
- ↑ Moore, Rick (2015-10-13). "2014 U.S. PET container recycling rate holds at 31%". http://www.napcor.com/pdf/RRate2014_PR-FINAL.pdf.
- ↑ Kint*, Darwin (1999). "A review on the potential biodegradability of poly(ethylene terephthalate)". Polymer International 48 (5): 346–352. doi:10.1002/(SICI)1097-0126(199905)48:5<346::AID-PI156>3.0.CO;2-N.
- ↑ "Garbage Decomposition Time | Waste Segregation Guide". http://www.getwaste.info/garbage-decomposition-time/.
- ↑ Chiu, Chih-Tung; Lee, Jui-Sheng; Chu, Chi-Shung; Chang, Yi-Pin; Wang, Yng-Jiin (2008-02-12). "Development of two alginate-based wound dressings". Journal of Materials Science: Materials in Medicine 19 (6): 2503–2513. doi:10.1007/s10856-008-3389-2. ISSN 0957-4530. PMID 18266085.
- ↑ Tønnesen, Hanne Hjorth; Karlsen, Jan (2002-01-01). "Alginate in Drug Delivery Systems". Drug Development and Industrial Pharmacy 28 (6): 621–630. doi:10.1081/DDC-120003853. ISSN 0363-9045. PMID 12149954.
- ↑ Alsberg, E.; Anderson, K. W.; Albeiruti, A.; Franceschi, R. T.; Mooney, D. J. (2001-11-01). "Cell-interactive Alginate Hydrogels for Bone Tissue Engineering". Journal of Dental Research 80 (11): 2025–2029. doi:10.1177/00220345010800111501. ISSN 0022-0345. PMID 11759015.
- ↑ Lee, Kuen Yong; Mooney, David J. (2012-01-01). "Alginate: Properties and biomedical applications". Progress in Polymer Science 37 (1): 106–126. doi:10.1016/j.progpolymsci.2011.06.003. PMID 22125349.
- ↑ "CALCIUM ALGINATE - National Library of Medicine HSDB Database". http://toxnet.nlm.nih.gov/cgi-bin/sis/search/a?dbs+hsdb:@term+@DOCNO+1911.
- ↑ Bouhadir, Kamal H.; Lee, Kuen Yong; Alsberg, Eben; Damm, Kelly L.; Anderson, Kenneth W.; Mooney, David J. (2001-01-01). "Degradation of Partially Oxidized Alginate and Its Potential Application for Tissue Engineering". Biotechnology Progress 17 (5): 945–950. doi:10.1021/bp010070p. ISSN 1520-6033. PMID 11587588.
- ↑ Steinbüchel, Alexander (2005). Polysaccharides and Polyamides in the Food Industry. Wiley-Blackwell. pp. 222. ISBN 978-3-527-31345-7.