Chemistry:Benzotriazole
Benzotriazole (BTA) is a heterocyclic compound with the chemical formula C
6H
4N
3H. It can be viewed as the fusion of a benzene and triazole rings. It is a white solid, although impure samples can appear tan. It is used as a corrosion inhibitor for copper and silver.[1][2]
Structure and synthesis
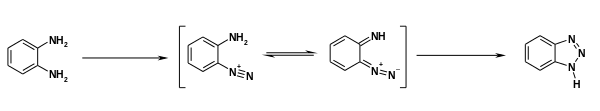
Benzotriazole features two fused rings. It can in principle exist as tautomers, but X-ray crystallography establishes the structure with hydrogen at the 1 position. The N=N and HN-N distances are 1.306 and 1.340 Å.[3]
Benzotriazole can be prepared by the reaction of o-phenylenediamine with a mixture sodium nitrite and acetic acid (a portion of this reaction mechanism is shown below):[4][5][6]
Reactions
Acid-base behavior
BTA is a weak Bronsted acid with a pKa = 8.2.[7] It is a weak Brønsted base, as indicated by the low pKa < 0 of its conjugate acid, [HBTA]+.[8]
It is also a Lewis base, binding Lewis acids at the C-N=N center. A variety coordination complexes are known such as the tetrahedral 2:1 derivative with zinc chloride, ZnCl2(BTA)2.[9] In some complexes, BTA binds metals as its conjugate base forming polymers and oligomers.[10] It binds to copper surfaces, serving as a corrosion inhibitor.[11]
N-alkylation
Deprotonation of BTA followed by treament with alkyl halides gives a mixture of 1- and 2-alkyl derivatives.[12] Aromatic aldehydes (ArCHO) in the presence of ethanol gives benzotriazole-based N,O-acetals:
- ArCHO + BtH + EtOH → ArCH(OEt)(Bt) + H
2O
These acetals are susceptible to deprotonation, giving access to acylsilanes[13] and acylboranes.
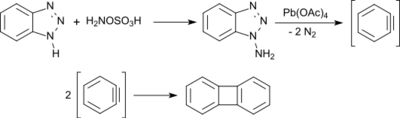
N-amination of BTA with hydroxylamine-O-sulfonic acid gives 1-aminobenzotriazole. Oxidation of this amine with lead(IV) acetate affords benzyne, which rapidly dimerises to biphenylene.[14]
Applications
Benzotriazole has been used as a restrainer (or anti-fogging agent) in photographic emulsions or developing solutions, and as a reagent for the analytical determination of silver. More importantly, it has been extensively used as a corrosion inhibitor in the atmosphere and underwater. BTA can be used as antifreezes, heating and cooling systems, hydraulic fluids, and vapor-phase inhibitors as well.[citation needed]
Corrosion inhibition
Benzotriazole is an corrosion inhibitor for non-ferrous metals like copper and silver.[2] It is known that a passive layer, consisting of a complex between copper and benzotriazole, is formed when copper is immersed in a solution containing benzotriazole. The passive layer is insoluble in aqueous and many organic solutions. There is a positive correlation between the thickness of the passive layer and the efficiency of preventing corrosion.[15] BTA is used in heritage conservation, notably for the treatment of bronze disease, and in dishwashing detergents for silver protection.[2]
Environmental relevance
Benzotriazole is fairly water-soluble, is not readily degraded, and has a limited sorption tendency. It is only partly removed in wastewater treatment plants and a substantial fraction reaches surface water such as rivers and lakes.[16] It is of low toxicity and a low health hazard to humans although exhibiting some antiestrogenic properties.[17] Benzotriazole (and tolyltriazole) is a common "polar organic persistent pollutant", often detected at >0.1 μg/L.[18] One source of this pollution is their use as anti-icing/deicing agents in airports.[19] Another source is dishwasher detergents, and some manufacturers are moving away from Benzotriazole to address pollution concerns.[2]
Benzotriazole derivatives
Many modifications of benzotriazole have been reported.[12][20][21][22] Alizapride is a commercial drug containing a benzotriazole ring system.
References
- ↑ "1,2,3-BENZOTRIAZOLE | CAMEO Chemicals | NOAA". https://cameochemicals.noaa.gov/chemical/19869.
- ↑ 2.0 2.1 2.2 2.3 "1H-Benzotriazole Round Table". German Centre for Micropollutants. https://www.umweltbundesamt.de/en/1h-benzotriazole-round-table.
- ↑ Krawczyk, Sławomir; Gdaniec, Maria (2005). "Polymorph β of 1 H -benzotriazole". Acta Crystallographica Section E: Structure Reports Online 61 (9): o2967–o2969. doi:10.1107/S1600536805025523.
- ↑ Robert A. Smiley "Phenylene- and Toluenediamines" in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a19_405
- ↑ Damschroder, R. E.; Peterson, W. D. (1940). "1,2,3-Benzotriazole". Organic Syntheses 20: 16. doi:10.15227/orgsyn.020.0016.
- ↑ Pereira, Claudio M. P.; Stefani, Helio A.; Guzen, Karla P.; Orfao, Aline T. G. (2007-07-31). "Improved Synthesis of Benzotriazoles and 1-Acylbenzotriazoles by Ultrasound Irradiation.". ChemInform 38 (31). doi:10.1002/chin.200731104. ISSN 0931-7597. https://onlinelibrary.wiley.com/doi/10.1002/chin.200731104.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedBenzotriazole: A novel synthetic auxiliary - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedAdventures with Benzotriazole - ↑ Søtofte, Inger; Nielsen, Kurt; Trysberg, Lennart; Uggla, Rolf; Nielsen, P. H. (1981). "Benzotriazole Complexes. II. The Crystal Structures of Benzotriazolium Tetrachlorocobaltate(II), Bis(benzotriazole)dichlorozinc(II) and Polymeric Tetrakis(benzotriazolato)dizinc(II)". Acta Chemica Scandinavica 35a: 739–745. doi:10.3891/acta.chem.scand.35a-0739.
- ↑ Wang, Yan; Hu, Man-Cheng; Zhai, Quan-Guo; Li, Shu-Ni; Jiang, Yu-Cheng; Ji, Wen-Juan (2009). "Synthesis, structure and luminescent properties of an Organic–Inorganic hybrid solid based on unprecedented flower-basket-shaped [] clusters with 1,2-bis(benzotriazole)ethane ligands". Inorganic Chemistry Communications 12 (4): 281–285. doi:10.1016/j.inoche.2009.01.003.
- ↑ Sease, Catherine (May 1978). "Benzotriazole: A Review for Conservators". Studies in Conservation. 2 23 (2): 76–85. doi:10.2307/1505798.
- ↑ 12.0 12.1 Katritzky, Alan R.; Rachwal, Stanislaw (2010). "Synthesis of Heterocycles Mediated by Benzotriazole. 1. Monocyclic Systems". Chemical Reviews 110 (3): 1564–1610. doi:10.1021/cr900204u. PMID 19799386.
- ↑ Katritzky, Alan R.; Wang, Zuoquan; Lang, Hengyuan (1996). "Novel and Convenient Synthesis of Aroyl-, Heteroaroyl-, Alkenoyl-, and Alkynoylsilanes". Organometallics 15 (2): 486–490. doi:10.1021/om950712b.
- ↑ Campbell, C.D.; Rees, C.W. (1969). "Reactive intermediates. Part I. Synthesis and oxidation of 1- and 2-aminobenzotriazole". J. Chem. Soc. C 1969 (5): 742–747. doi:10.1039/J39690000742.
- ↑ Finšgar, M.; Milošev I. (11 March 2010). "Inhibition of copper corrosion by 1,2,3-benzotriazole: A review". Corrosion Science 52 (9): 2737–2749. doi:10.1016/j.corsci.2010.05.002. Bibcode: 2010Corro..52.2737F.
- ↑ Giger, W; Schaffner, C; Kohler, HP (2006). "Benzotriazole and tolyltriazole as aquatic contaminants. 1. Input and occurrence in rivers and lakes". Environmental Science & Technology 40 (23): 7186–92. doi:10.1021/es061565j. PMID 17180965.
- ↑ Farré, Marinel la; Pérez, Sandra; Kantiani, Lina; Barceló, Damià (2008). "Fate and toxicity of emerging pollutants, their metabolites and transformation products in the aquatic environment". TrAC Trends in Analytical Chemistry 27 (11): 991–1007. doi:10.1016/j.trac.2008.09.010. ISSN 0165-9936.
- ↑ Loos, Robert; Locoro, Giovanni; Comero, Sara; Contini, Serafino; Schwesig, David; Werres, Friedrich; Balsaa, Peter; Gans, Oliver et al. (2010). "Pan-European survey on the occurrence of selected polar organic persistent pollutants in ground water". Water Research 44 (14): 4115–4126. doi:10.1016/j.watres.2010.05.032. PMID 20554303. Bibcode: 2010WatRe..44.4115L.
- ↑ Castro, Sigifredo; Davis, Lawrence C.; Erickson, Larry E. (2005). "Natural, cost-effective, and sustainable alternatives for treatment of aircraft deicing fluid waste". Environmental Progress 24 (1): 26–33. doi:10.1002/ep.10059. Bibcode: 2005EnvPr..24...26C.
- ↑ Katritzky, Alan R.; Rachwal, Stanislaw (2011). "Synthesis of Heterocycles Mediated by Benzotriazole. 2. Bicyclic Systems". Chemical Reviews 111 (11): 7063–7120. doi:10.1021/cr200031r. PMID 21894899.
- ↑ Akter, Monalisa; Rupa, Kavuri; Anbarasan, Pazhamalai (2022). "1,2,3-Triazole and Its Analogues: New Surrogates for Diazo Compounds". Chemical Reviews 122 (15): 13108–13205. doi:10.1021/acs.chemrev.1c00991. PMID 35852917.
- ↑ Katritzky, Alan R.; Lan, Xiangfu; Yang, Jason Z.; Denisko, Olga V. (1998). "Properties and Synthetic Utility of N -Substituted Benzotriazoles". Chemical Reviews 98 (2): 409–548. doi:10.1021/cr941170v. PMID 11848906.
 |