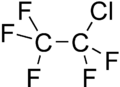
Chemistry:Chloropentafluoroethane
From HandWiki
|
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1-Chloro-1,1,2,2,2-pentafluoroethane | |||
| Other names
Freon 115, CFC-115, R-115, Fluorocarbon-115, Genetron 115, Halocarbon 115, Monochloropentafluoroethane
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEMBL | |||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1020 | ||
| |||
| |||
| Properties | |||
| C2ClF5 | |||
| Molar mass | 154.466 g/mol | ||
| Appearance | Colorless gas | ||
| Odor | Ethereal | ||
| Melting point | −99 °C (−146 °F; 174 K) | ||
| Boiling point | −39.1 °C (−38.4 °F; 234.1 K) | ||
| 59 mg/L | |||
| Vapor pressure | 7.9 atm (21°C)[1] | ||
| Hazards | |||
| Main hazards | In high concentrations may cause asphyxiation.[2] | ||
| GHS pictograms |  
| ||
| GHS Signal word | Warning | ||
| H280, H420 | |||
| P410+403, P502 | |||
| Flash point | 70.4 °C (158.7 °F; 343.5 K) | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none[1] | ||
REL (Recommended)
|
TWA 1000 ppm (6320 mg/m3)[1] | ||
IDLH (Immediate danger)
|
N.D.[1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
Chloropentafluoroethane is a chlorofluorocarbon (CFC) once used as a refrigerant and also known as R-115 and CFC-115. Its production and consumption has been banned since 1 January 1996 under the Montreal Protocol because of its high ozone depletion potential and very long lifetime when released into the environment.[3] CFC-115 is also a potent greenhouse gas.
Atmospheric properties
The atmospheric abundance of CFC-115 rose from 8.4 parts per trillion (ppt) in year 2010 to 8.7 ppt in 2020 based on analysis of air samples gathered from sites around the world.[4]
| Property | Value |
|---|---|
| Ozone depletion potential (ODP) | 0.44[5] (CCl3F = 1) |
| Global warming potential (GWP: 100-year) | 5,860[6] - 7,670[7] (CO2 = 1) |
| Atmospheric lifetime | 1,020[5] - 1,700[6] years |
See also
References
- ↑ 1.0 1.1 1.2 1.3 NIOSH Pocket Guide to Chemical Hazards. "#0131". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0131.html.
- ↑ http://encyclopedia.airliquide.com/sds/en/030_AL_EN.pdf[yes|permanent dead link|dead link}}]
- ↑ Ozone Depleting Substances List (Montreal Protocol)
- ↑ "AGAGE Data and Figures". Massachusetts Institute of Technology. https://agage.mit.edu/data/agage-data. Retrieved 2021-02-11.
- ↑ 5.0 5.1 John S. Daniel; Guus J.M. Velders; A.R. Douglass; P.M.D. Forster; D.A. Hauglustaine; I.S.A. Isaksen; L.J.M. Kuijpers; A. McCulloch et al. (2006). "Chapter 8. Halocarbon Scenarios, Ozone Depletion Potentials, and Global Warming Potentials". Scientific Assessment of Ozone Depletion: 2006. Geneva, Switzerland: World Meteorological Organization. http://www.esrl.noaa.gov/csd/assessments/ozone/2006/chapters/chapter8.pdf. Retrieved 9 October 2016.
- ↑ 6.0 6.1 "Chapter 8". AR5 Climate Change 2013: The Physical Science Basis. p. 731. https://www.ipcc.ch/report/ar5/wg1/.
- ↑ "Refrigerants - Environmental Properties". http://www.engineeringtoolbox.com/Refrigerants-Environment-Properties-d_1220.html. Retrieved 2016-09-12.
 |