Chemistry:Greenhouse gas
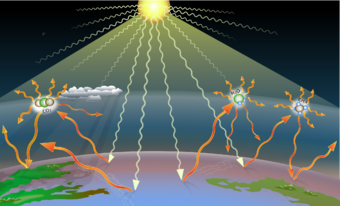
Greenhouse gases are the gases in the atmosphere that raise the surface temperature of planets such as the Earth. What distinguishes them from other gases is that they absorb the wavelengths of radiation that a planet emits, resulting in the greenhouse effect.[1] The Earth is warmed by sunlight, causing its surface to radiate heat, which is then mostly absorbed by greenhouse gases. Without greenhouse gases in the atmosphere, the average temperature of Earth's surface would be about −18 °C (0 °F),[2] rather than the present average of 15 °C (59 °F).[3][4]
The most abundant greenhouse gases in Earth's atmosphere, listed in decreasing order of average global mole fraction, are:[5][6] Water vapor (H2O), Carbon dioxide (CO2), Methane (CH4), Nitrous oxide (N2O), Ozone (O3), Chlorofluorocarbons (CFCs and HCFCs), Hydrofluorocarbons (HFCs), Perfluorocarbons (CF4, C2F6, etc.), SF6, and NF3. Yet, while water vapor is a potent greenhouse gas, humans are not directly adding to its concentrations,[7]. so it is not one of the primary drivers of climate change, but rather one of the feedbacks.[8] On the other hand, carbon dioxide is causing about three quarters of global warming and can take thousands of years to be fully absorbed by the carbon cycle.[9][10] Methane causes most of the remaining warming and lasts in the atmosphere for an average of 12 years.[11]
Human activities since the beginning of the Industrial Revolution (around 1750) have increased atmospheric methane concentrations by over 150% and carbon dioxide by over 50%,[12][13] up to a level not seen in over 3 million years.[14] The vast majority of carbon dioxide emissions by humans come from the combustion of fossil fuels, principally coal, petroleum (including oil) and natural gas. Additional contributions come from cement manufacturing, fertilizer production, and changes in land use like deforestation.[15]: 687 [16][17] Methane emissions originate from agriculture, fossil fuel production, waste, and other sources.[18]
According to Berkeley Earth, average global surface temperature has risen by more than 1.2 °C (2.2 °F) since the pre-industrial (1850–1899) period as a result of greenhouse gas emissions. If current emission rates continue then temperature rises will surpass 2.0 °C (3.6 °F) sometime between 2040 and 2070, which is the level the United Nations ' Intergovernmental Panel on Climate Change (IPCC) says is "dangerous".[19]
Properties
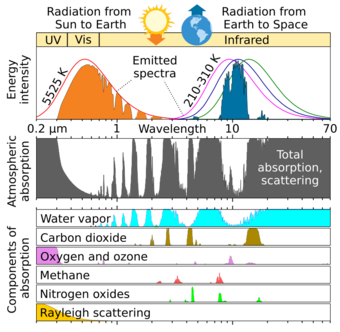
Greenhouse gases are infrared active, meaning that they absorb and emit infrared radiation in the same long wavelength range as what is emitted by the Earth's surface, clouds and atmosphere.[20]: 2233
99% of the Earth's dry atmosphere (excluding water vapor) is made up of nitrogen (N2) (78%) and oxygen (O2) (21%). Because their molecules contain two atoms of the same element, they have no asymmetry in the distribution of their electrical charges,[21] and so are almost totally unaffected by infrared thermal radiation,[22] with only an extremely minor effect from collision-induced absorption.[23][24][25] A further 0.9% of the atmosphere is made up by argon (Ar), which is monatomic, and so completely transparent to thermal radiation. On the other hand, carbon dioxide (0.04%), methane, nitrous oxide and even less abundant trace gases account for less than 0.1% of Earth's atmosphere, but because their molecules contain atoms of different elements, there is an asymmetry in electric charge distribution which allows molecular vibrations to interact with electromagnetic radiation. This makes them infrared active, and so their presence causes greenhouse effect.[21]
Radiative forcing

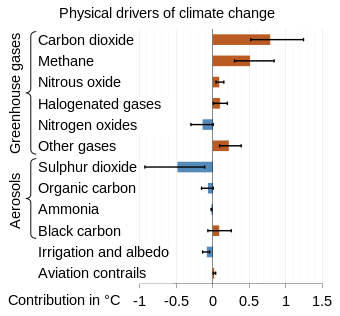
Earth absorbs some of the radiant energy received from the sun, reflects some of it as light and reflects or radiates the rest back to space as heat. A planet's surface temperature depends on this balance between incoming and outgoing energy. When Earth's energy balance is shifted, its surface becomes warmer or cooler, leading to a variety of changes in global climate.[26] Radiative forcing is a metric calculated in watts per square meter, which characterizes the impact of an external change in a factor that influences climate. It is calculated as the difference in top-of-atmosphere (TOA) energy balance immediately caused by such an external change A positive forcing, such as from increased concentrations of greenhouse gases, means more energy arriving than leaving at the top-of-atmosphere, which causes additional warming, while negative forcing, like from sulfates forming in the atmosphere from sulfur dioxide, leads to cooling.[20]: 2245 [27]
Within the lower atmosphere, greenhouse gases exchange thermal radiation with the surface and limit radiative heat flow away from it, which reduces the overall rate of upward radiative heat transfer.[28]: 139 [29] The increased concentration of greenhouse gases is also cooling the upper atmosphere, as it is much thinner than the lower layers, and any heat re-emitted from greenhouse gases is more likely to travel further to space than to interact with the fewer gas molecules in the upper layers. The upper atmosphere is also shrinking as the result.[30]
Global warming potential (GWP) and CO2 equivalents
Contributions of specific gases to the greenhouse effect

Overall greenhouse effect
This table shows the most important contributions to the overall greenhouse effect, without which the average temperature of Earth's surface would be about −18 °C (0 °F),[2] instead of around 15 °C (59 °F).[3] This table also specifies tropospheric ozone, because this gas has a cooling effect in the stratosphere, but a warming influence comparable to nitrous oxide and CFCs in the troposphere.[32]
| K&T (1997)[33] | Schmidt (2010)[34] | |||
|---|---|---|---|---|
| Contributor | Clear Sky | With Clouds | Clear Sky | With Clouds |
| Water vapor | 60 | 41 | 67 | 50 |
| Clouds | 31 | 25 | ||
| CO 2 |
26 | 18 | 24 | 19 |
| Tropospheric ozone (O3) | 8 | |||
| N 2O + CH 4 |
6 | |||
| Other | 9 | 9 | 7 | |
|
K&T (1997) used 353 ppm CO | ||||
Concentrations and other characteristics of greenhouse gases
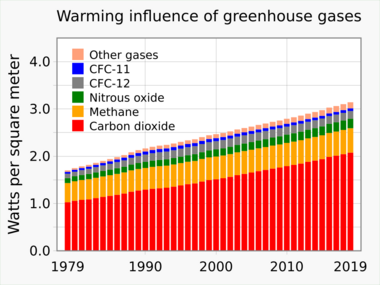
Anthropogenic changes to the natural greenhouse effect are sometimes referred to as the enhanced greenhouse effect.[20]: 2223 The contribution of each gas to the enhanced greenhouse effect is determined by the characteristics of that gas, its abundance, and any indirect effects it may cause. For example, the direct radiative effect of a mass of methane is about 84 times stronger than the same mass of carbon dioxide over a 20-year time frame.[38] Since the 1980s, greenhouse gas forcing contributions (relative to year 1750) are also estimated with high accuracy using IPCC-recommended expressions derived from radiative transfer models.[39]
The concentration of a greenhouse gas is typically measured in parts per million (ppm) or parts per billion (ppb) by volume. A CO
2 concentration of 420 ppm means that 420 out of every million air molecules is a CO
2 molecule. The first 30 ppm increase in CO
2 concentrations took place in about 200 years, from the start of the Industrial Revolution to 1958; however the next 90 ppm increase took place within 56 years, from 1958 to 2014.[13][40][41] Similarly, the average annual increase in the 1960s was only 37% of what it was in 2000 through 2007.[42]
Many observations are available online in a variety of atmospheric chemistry observational databases. The table below shows the most influential long-lived, well-mixed greenhouse gases, along with their tropospheric concentrations and direct radiative forcings, as identified by the Intergovernmental Panel on Climate Change (IPCC).[43] Abundances of these trace gases are regularly measured by atmospheric scientists from samples collected throughout the world.[44][45][46] It excludes water vapor because changes in its concentrations are calculated as a climate change feedback indirectly caused by changes in other greenhouse gases, as well as ozone, whose concentrations are only modified indirectly by various refrigerants that cause ozone depletion. Some short-lived gases (e.g. carbon monoxide, NOx) and aerosols (e.g. mineral dust or black carbon) are also excluded because of limited role and strong variation, alongside with minor refrigants and other halogenated gases, which have been mass-produced in smaller quantities than those in the table.[43]: 731–738 and Annex III of the 2021 IPCC WG1 Report[47]: 4–9
| Species | Lifetime
(years) [43]: 731 |
100-yr | Mole Fraction [ppt - except as noted]a + Radiative forcing [W m−2] [B] | Concentrations
up to year 2020 | ||||
|---|---|---|---|---|---|---|---|---|
| Baseline
Year 1750 |
TAR[50]
Year 1998 |
AR4[51]
Year 2005 |
AR5[43]: 678
Year 2011 |
AR6[47]: 4–9
Year 2019 | ||||
| CO2 [ppm] | [A] | 1 | 278 | 365 (1.46) | 379 (1.66) | 391 (1.82) | 410 (2.16) | 
|
| CH4 [ppb] | 12.4 | 28 | 700 | 1,745 (0.48) | 1,774 (0.48) | 1,801 (0.48) | 1866 (0.54) | 
|
| N2O [ppb] | 121 | 265 | 270 | 314 (0.15) | 319 (0.16) | 324 (0.17) | 332 (0.21) | Error creating thumbnail: Unable to save thumbnail to destination |
| CFC-11 | 45 | 4,660 | 0 | 268 (0.07) | 251 (0.063) | 238 (0.062) | 226 (0.066) | 
|
| CFC-12 | 100 | 10,200 | 0 | 533 (0.17) | 538 (0.17) | 528 (0.17) | 503 (0.18) | 
|
| CFC-13 | 640 | 13,900 | 0 | 4 (0.001) | - | 2.7 (0.0007) | 3.28 (0.0009) | cfc13 |
| CFC-113 | 85 | 6,490 | 0 | 84 (0.03) | 79 (0.024) | 74 (0.022) | 70 (0.021) | 
|
| CFC-114 | 190 | 7,710 | 0 | 15 (0.005) | - | - | 16 (0.005) | cfc114 |
| CFC-115 | 1,020 | 5,860 | 0 | 7 (0.001) | - | 8.37 (0.0017) | 8.67 (0.0021) | cfc115 |
| HCFC-22 | 11.9 | 5,280 | 0 | 132 (0.03) | 169 (0.033) | 213 (0.0447) | 247 (0.0528) | 
|
| HCFC-141b | 9.2 | 2,550 | 0 | 10 (0.001) | 18 (0.0025) | 21.4 (0.0034) | 24.4 (0.0039) | 
|
| HCFC-142b | 17.2 | 5,020 | 0 | 11 (0.002) | 15 (0.0031) | 21.2 (0.0040) | 22.3 (0.0043) | 
|
| CH3CCl3 | 5 | 160 | 0 | 69 (0.004) | 19 (0.0011) | 6.32 (0.0004) | 1.6 (0.0001) | 
|
| CCl4 | 26 | 1,730 | 0 | 102 (0.01) | 93 (0.012) | 85.8 (0.0146) | 78 (0.0129) | 
|
| HFC-23 | 222 | 12,400 | 0 | 14 (0.002) | 18 (0.0033) | 24 (0.0043) | 32.4 (0.0062) | 
|
| HFC-32 | 5.2 | 677 | 0 | - | - | 4.92 (0.0005) | 20 (0.0022) | 
|
| HFC-125 | 28.2 | 3,170 | 0 | - | 3.7 (0.0009) | 9.58 (0.0022) | 29.4 (0.0069) | 
|
| HFC-134a | 13.4 | 1,300 | 0 | 7.5 (0.001) | 35 (0.0055) | 62.7 (0.0100) | 107.6 (0.018) | 
|
| HFC-143a | 47.1 | 4,800 | 0 | - | - | 12.0 (0.0019) | 24 (0.0040) | 
|
| HFC-152a | 1.5 | 138 | 0 | 0.5 (0.0000) | 3.9 (0.0004) | 6.4 (0.0006) | 7.1 (0.0007) | Error creating thumbnail: Unable to save thumbnail to destination |
| CF4 (PFC-14) | 50,000 | 6,630 | 40 | 80 (0.003) | 74 (0.0034) | 79 (0.0040) | 85.5 (0.0051) | 
|
| C2F6 (PFC-116) | 10,000 | 11,100 | 0 | 3 (0.001) | 2.9 (0.0008) | 4.16 (0.0010) | 4.85 (0.0013) | 
|
| SF6 | 3,200 | 23,500 | 0 | 4.2 (0.002) | 5.6 (0.0029) | 7.28 (0.0041) | 9.95 (0.0056) | Error creating thumbnail: Unable to save thumbnail to destination |
| SO2F2 | 36 | 4,090 | 0 | - | - | 1.71 (0.0003) | 2.5 (0.0005) | 
|
| NF3 | 500 | 16,100 | 0 | - | - | 0.9 (0.0002) | 2.05 (0.0004) | 
|
a Mole fractions: μmol/mol = ppm = parts per million (106); nmol/mol = ppb = parts per billion (109); pmol/mol = ppt = parts per trillion (1012).
A The IPCC states that "no single atmospheric lifetime can be given" for CO2.[43]: 731 This is mostly due to the rapid growth and cumulative magnitude of the disturbances to Earth's carbon cycle by the geologic extraction and burning of fossil carbon.[52] As of year 2014, fossil CO2 emitted as a theoretical 10 to 100 GtC pulse on top of the existing atmospheric concentration was expected to be 50% removed by land vegetation and ocean sinks in less than about a century, as based on the projections of coupled models referenced in the AR5 assessment.[53] A substantial fraction (20-35%) was also projected to remain in the atmosphere for centuries to millennia, where fractional persistence increases with pulse size.[54][55]
B Values are relative to year 1750. AR6 reports the effective radiative forcing which includes effects of rapid adjustments in the atmosphere and at the surface.[56]
Factors affecting concentrations
Atmospheric concentrations are determined by the balance between sources (emissions of the gas from human activities and natural systems) and sinks (the removal of the gas from the atmosphere by conversion to a different chemical compound or absorption by bodies of water).[57]: 512
Airborne fraction
The proportion of an emission remaining in the atmosphere after a specified time is the "airborne fraction" (AF). The annual airborne fraction is the ratio of the atmospheric increase in a given year to that year's total emissions.
As of 2006 the annual airborne fraction for CO2 was about 0.45. The annual airborne fraction increased at a rate of 0.25 ± 0.21% per year over the period 1959–2006.[58]
Atmospheric lifetime
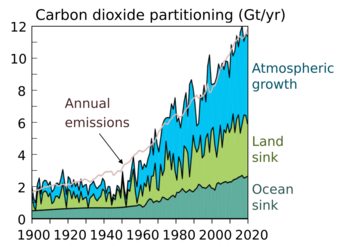
2 emissions have been absorbed by carbon sinks, including plant growth, soil uptake, and ocean uptake (2020 Global Carbon Budget).
Aside from water vapor, which has a residence time of about nine days,[59] major greenhouse gases are well mixed and take many years to leave the atmosphere.[60] Although it is not easy to know with precision how long it takes greenhouse gases to leave the atmosphere, there are estimates for the principal greenhouse gases. Jacob (1999)[61] defines the lifetime of an atmospheric species X in a one-box model as the average time that a molecule of X remains in the box. Mathematically can be defined as the ratio of the mass (in kg) of X in the box to its removal rate, which is the sum of the flow of X out of the box (), chemical loss of X (), and deposition of X () (all in kg/s):
- .[61]
If input of this gas into the box ceased, then after time , its concentration would decrease by about 63%.
The atmospheric lifetime of a species therefore measures the time required to restore equilibrium following a sudden increase or decrease in its concentration in the atmosphere. Individual atoms or molecules may be lost or deposited to sinks such as the soil, the oceans and other waters, or vegetation and other biological systems, reducing the excess to background concentrations. The average time taken to achieve this is the mean lifetime.
Carbon dioxide has a variable atmospheric lifetime, and cannot be specified precisely.[62][38][20]: 2237 Similar issues apply to other greenhouse gases, many of which have longer mean lifetimes than CO2, e.g. N2O has a mean atmospheric lifetime of 121 years.[38]
Water vapor
Water vapor concentrations fluctuate regionally, but human activity does not directly affect water vapor concentrations except at local scales, such as near irrigated fields. Indirectly, human activity that increases global temperatures will increase water vapor concentrations, because Clausius–Clapeyron relation establishes that more water vapor will be present per unit volume at elevated temperatures. This process known as water vapor feedback.[63] The atmospheric concentration of vapor is highly variable and depends largely on temperature, from less than 0.01% in extremely cold regions up to 3% by mass in saturated air at about 32 °C.[64]
Sources
Natural sources
Most greenhouse gases have both natural and human-caused sources. An exception are purely human-produced synthetic halocarbons which have no natural sources. During the pre-industrial Holocene, concentrations of existing gases were roughly constant, because the large natural sources and sinks roughly balanced. In the industrial era, human activities have added greenhouse gases to the atmosphere, mainly through the burning of fossil fuels and clearing of forests.[65][4]: 115
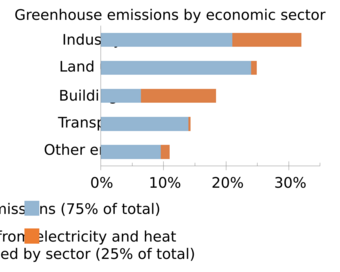
Greenhouse gas emissions from human activities
Monitoring
Greenhouse gas monitoring involves the direct measurement of greenhouse gas emissions and levels. There are several different methods of measuring carbon dioxide concentrations in the atmosphere, including infrared analyzing and manometry. Methane and nitrous oxide are measured by other instruments. Greenhouse gases are measured from space such as by the Orbiting Carbon Observatory and networks of ground stations such as the Integrated Carbon Observation System.[citation needed]
The Annual Greenhouse Gas Index (AGGI) is defined by atmospheric scientists at NOAA as the ratio of total direct radiative forcing due to long-lived and well-mixed greenhouse gases for any year for which adequate global measurements exist, to that present in year 1990.[37][66] These radiative forcing levels are relative to those present in year 1750 (i.e. prior to the start of the industrial era). 1990 is chosen because it is the baseline year for the Kyoto Protocol, and is the publication year of the first IPCC Scientific Assessment of Climate Change.
As such, NOAA states that the AGGI "measures the commitment that (global) society has already made to living in a changing climate. It is based on the highest quality atmospheric observations from sites around the world. Its uncertainty is very low."[67]
Data networks
Removal from the atmosphere
Natural processes
Carbon dioxide is removed from the atmosphere primarily through photosynthesis and enters the terrestrial and oceanic biospheres. Carbon dioxide also dissolves directly from the atmosphere into bodies of water (ocean, lakes, etc.), as well as dissolving in precipitation as raindrops fall through the atmosphere. When dissolved in water, carbon dioxide reacts with water molecules and forms carbonic acid, which contributes to ocean acidity. It can then be absorbed by rocks through weathering. It also can acidify other surfaces it touches or be washed into the ocean.[68]
Negative emissions
A number of technologies remove greenhouse gases emissions from the atmosphere. Most widely analyzed are those that remove carbon dioxide from the atmosphere, either to geologic formations such as bio-energy with carbon capture and storage and carbon dioxide air capture,[69] or to the soil as in the case with biochar.[69] Many long-term climate scenario models require large-scale human-made negative emissions to avoid serious climate change.[70] Negative emissions approaches are also being studied for atmospheric methane, called atmospheric methane removal.[71]
During geologic time scales
History of discovery

In the late 19th century, scientists experimentally discovered that N2 and O2 do not absorb infrared radiation (called, at that time, "dark radiation"), while water (both as true vapor and condensed in the form of microscopic droplets suspended in clouds) and CO
2 and other poly-atomic gaseous molecules do absorb infrared radiation.[73][74] In the early 20th century, researchers realized that greenhouse gases in the atmosphere made Earth's overall temperature higher than it would be without them. The term greenhouse was first applied to this phenomenon by Nils Gustaf Ekholm in 1901.[75][76]
During the late 20th century, a scientific consensus evolved that increasing concentrations of greenhouse gases in the atmosphere cause a substantial rise in global temperatures and changes to other parts of the climate system,[77] with consequences for the environment and for human health.
Other planets
Greenhouse gases exist in many atmospheres, creating greenhouse effects on Mars, Titan and particularly in the thick atmosphere of Venus.[78] While Venus has been described as the ultimate end state of runaway greenhouse effect, such a process would have virtually no chance of occurring from any increases in greenhouse gas concentrations caused by humans,[79] as the Sun's brightness is too low and it would likely need to increase by some tens of percents, which will take a few billion years.[80]
See also
- Carbon accounting
- Carbon budget
- Climate change feedback
- Greenhouse gas monitoring
- Greenhouse gas inventory
- List of refrigerants
References
- ↑ Matthews, J.B.R. et al. (2021-08-09). "Annex VII: Glossary". Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. IPCC / Cambridge University Press. pp. 2215–2256. doi:10.1017/9781009157896.022. ISBN 9781009157896. https://www.ipcc.ch/report/ar6/wg1/downloads/report/IPCC_AR6_WGI_AnnexVII.pdf.
- ↑ 2.0 2.1 "NASA GISS: Science Briefs: Greenhouse Gases: Refining the Role of Carbon Dioxide". http://www.giss.nasa.gov/research/briefs/ma_01/.
- ↑ 3.0 3.1 "Modern global climate change". Science 302 (5651): 1719–23. 2003. doi:10.1126/science.1090228. PMID 14657489. Bibcode: 2003Sci...302.1719K. https://zenodo.org/record/1230878. Retrieved 26 July 2019.
- ↑ 4.0 4.1 Le Treut, H., R. Somerville, U. Cubasch, Y. Ding, C. Mauritzen, A. Mokssit, T. Peterson and M. Prather, 2007: Chapter 1: Historical Overview of Climate Change. In: Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change [Solomon, S., D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
- ↑ "Atmospheric Concentration of Greenhouse Gases". U.S. Environmental Protection Agency. 2016-08-01. https://www.epa.gov/sites/default/files/2016-08/documents/print_ghg-concentrations-2016.pdf.
- ↑ "Inside the Earth's invisible blanket.". http://sequestration.org/science/greenhousegases.html.
- ↑ Gavin Schmidt (2010-10-01). "Taking the Measure of the Greenhouse Effect". NASA Goddard Institute for Space Studies - Science Briefs. https://www.giss.nasa.gov/research/briefs/2010_schmidt_05/.
- ↑ "NASA Science Mission Directorate article on the water cycle". Nasascience.nasa.gov. http://nasascience.nasa.gov/earth-science/oceanography/ocean-earth-system/ocean-water-cycle.
- ↑ "Global Greenhouse Gas Emissions Data". United States Environmental Protection Agency. 12 January 2016. https://www.epa.gov/ghgemissions/global-greenhouse-gas-emissions-data.
- ↑ "Climate Change Indicators: Greenhouse Gases". United States Environmental Protection Agency. 16 December 2015. https://www.epa.gov/climate-indicators/greenhouse-gases. "Carbon dioxide's lifetime cannot be represented with a single value because the gas is not destroyed over time, but instead moves among different parts of the ocean–atmosphere–land system. Some of the excess carbon dioxide is absorbed quickly (for example, by the ocean surface), but some will remain in the atmosphere for thousands of years, due in part to the very slow process by which carbon is transferred to ocean sediments."
- ↑ "Understanding methane emissions". International Energy Agency. https://www.iea.org/reports/global-methane-tracker-2023/understanding-methane-emissions.
- ↑ "Understanding methane emissions". International Energy Agency. https://www.iea.org/reports/global-methane-tracker-2023/understanding-methane-emissions. "The concentration of methane in the atmosphere is currently over two-and-a-half times greater than its pre-industrial levels"
- ↑ 13.0 13.1 "Carbon dioxide now more than 50% higher than pre-industrial levels" (in en). National Oceanic and Atmospheric Administration. 3 June 2022. https://www.noaa.gov/news-release/carbon-dioxide-now-more-than-50-higher-than-pre-industrial-levels.
- ↑ "Climate Change: Atmospheric Carbon Dioxide". https://www.climate.gov/news-features/understanding-climate/climate-change-atmospheric-carbon-dioxide.
- ↑ Canadell, J.G., P.M.S. Monteiro, M.H. Costa, L. Cotrim da Cunha, P.M. Cox, A.V. Eliseev, S. Henson, M. Ishii, S. Jaccard, C. Koven, A. Lohila, P.K. Patra, S. Piao, J. Rogelj, S. Syampungani, S. Zaehle, and K. Zickfeld, 2021: Chapter 5: Global Carbon and other Biogeochemical Cycles and Feedbacks. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [Masson-Delmotte, V., P. Zhai, A. Pirani, S.L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M.I. Gomis, M. Huang, K. Leitzell, E. Lonnoy, J.B.R. Matthews, T.K. Maycock, T. Waterfield, O. Yelekçi, R. Yu, and B. Zhou (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 673–816, doi:10.1017/9781009157896.007.
- ↑ "Global Greenhouse Gas Emissions Data". U.S. Environmental Protection Agency. 12 January 2016. https://www.epa.gov/ghgemissions/global-greenhouse-gas-emissions-data. "The burning of coal, natural gas, and oil for electricity and heat is the largest single source of global greenhouse gas emissions."
- ↑ "AR4 SYR Synthesis Report Summary for Policymakers – 2 Causes of change". ipcc.ch. https://www.ipcc.ch/publications_and_data/ar4/syr/en/spms2.html.
- ↑ "Global Methane Tracker 2023". International Energy Agency. https://www.iea.org/reports/global-methane-tracker-2023.
- ↑ "Analysis: When might the world exceed 1.5C and 2C of global warming?" (in en). 2020-12-04. https://www.carbonbrief.org/analysis-when-might-the-world-exceed-1-5c-and-2c-of-global-warming.
- ↑ 20.0 20.1 20.2 20.3 IPCC, 2021: Annex VII: Glossary [Matthews, J.B.R., V. Möller, R. van Diemen, J.S. Fuglestvedt, V. Masson-Delmotte, C. Méndez, S. Semenov, A. Reisinger (eds.)]. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [Masson-Delmotte, V., P. Zhai, A. Pirani, S.L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M.I. Gomis, M. Huang, K. Leitzell, E. Lonnoy, J.B.R. Matthews, T.K. Maycock, T. Waterfield, O. Yelekçi, R. Yu, and B. Zhou (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 2215–2256, doi:10.1017/9781009157896.022.
- ↑ 21.0 21.1 Archer, David (2011). Global Warming: Understanding the Forecast, Chapter 4: Greenhouse Gases (2 ed.). Wiley. ISBN 978-0470943410. http://forecast.uchicago.edu/chapter4.pdf. Retrieved 14 June 2023.
- ↑ Wei, Peng-Sheng; Hsieh, Yin-Chih; Chiu, Hsuan-Han; Yen, Da-Lun; Lee, Chieh; Tsai, Yi-Cheng; Ting, Te-Chuan (6 October 2018). "Absorption coefficient of carbon dioxide across atmospheric troposphere layer". Heliyon 4 (10): e00785. doi:10.1016/j.heliyon.2018.e00785. PMID 30302408. Bibcode: 2018Heliy...400785W.
- ↑ Höpfner, M.; Milz, M.; Buehler, S.; Orphall, J.; Stiller, G. (24 May 2012). "The natural greenhouse effect of atmospheric oxygen (O2) and nitrogen (N2)" (in en). Geophysical Research Letters 39 (L10706). doi:10.1029/2012GL051409. ISSN 1944-8007. Bibcode: 2012GeoRL..3910706H.
- ↑ "Which Gases Are Greenhouse Gases?". American Chemical Society. https://www.acs.org/content/acs/en/climatescience/greenhousegases/whichgases.html.
- ↑ Höpfner, M.; Milz, M.; Buehler, S.; Orphall, J.; Stiller, G. (24 May 2012). "The natural greenhouse effect of atmospheric oxygen (O2) and nitrogen (N2)" (in en). Geophysical Research Letters 39 (L10706). doi:10.1029/2012GL051409. ISSN 1944-8007. Bibcode: 2012GeoRL..3910706H.
- ↑ "Climate Change Indicators in the United States - Greenhouse Gases". U.S. Environmental Protection Agency (EPA). 2016. https://www.epa.gov/climate-indicators/greenhouse-gases..
- ↑ "Climate Change Indicators in the United States - Climate Forcing". U.S. Environmental Protection Agency (EPA). 2016. https://www.epa.gov/climate-indicators/climate-change-indicators-climate-forcing.[1]
- ↑ Wallace, J. M.; Hobbs, P. V. (2006). Atmospheric Science (2 ed.). Academic Press. ISBN 978-0-12-732951-2.
- ↑ Manabe, S.; Strickler, R. F. (1964). "Thermal Equilibrium of the Atmosphere with a Convective Adjustment". J. Atmos. Sci. 21 (4): 361–385. doi:10.1175/1520-0469(1964)021<0361:TEOTAW>2.0.CO;2. Bibcode: 1964JAtS...21..361M.
- ↑ Hatfield, Miles (30 June 2021). "NASA Satellites See Upper Atmosphere Cooling and Contracting Due to Climate Change". NASA. https://www.nasa.gov/general/nasa-satellites-see-upper-atmosphere-cooling-and-contracting-due-to-climate-change/.
- ↑ "NASA: Climate Forcings and Global Warming". January 14, 2009. http://earthobservatory.nasa.gov/Features/EnergyBalance/page7.php.
- ↑ "Atmospheric Concentration of Greenhouse Gases". U.S. Environmental Protection Agency. 2016-08-01. https://www.epa.gov/sites/default/files/2016-08/documents/print_ghg-concentrations-2016.pdf.
- ↑ Kiehl, J.T.; Kevin E. Trenberth (1997). "Earth's annual global mean energy budget". Bulletin of the American Meteorological Society 78 (2): 197–208. doi:10.1175/1520-0477(1997)078<0197:EAGMEB>2.0.CO;2. Bibcode: 1997BAMS...78..197K. http://www.geo.utexas.edu/courses/387h/PAPERS/kiehl.pdf.
- ↑ Schmidt, G.A.; R. Ruedy; R.L. Miller; A.A. Lacis (2010), "The attribution of the present-day total greenhouse effect", J. Geophys. Res. 115 (D20): pp. D20106, doi:10.1029/2010JD014287, Bibcode: 2010JGRD..11520106S, http://pubs.giss.nasa.gov/docs/2010/2010_Schmidt_etal_1.pdf, D20106. Web page
- ↑ "The NOAA Annual Greenhouse Gas Index (AGGI)". National Oceanic and Atmospheric Administration (NOAA). Spring 2023. https://gml.noaa.gov/aggi/aggi.html.
- ↑ "Annual Greenhouse Gas Index". U.S. Global Change Research Program. https://www.globalchange.gov/browse/indicators/annual-greenhouse-gas-index.
- ↑ 37.0 37.1 Butler J. and Montzka S. (2020). "The NOAA Annual Greenhouse Gas Index (AGGI)". NOAA Global Monitoring Laboratory/Earth System Research Laboratories. https://www.esrl.noaa.gov/gmd/aggi/aggi.html.
- ↑ 38.0 38.1 38.2 "Appendix 8.A". Intergovernmental Panel on Climate Change Fifth Assessment Report. p. 731. http://www.ipcc.ch/pdf/assessment-report/ar5/wg1/WG1AR5_Chapter08_FINAL.pdf. Retrieved 6 November 2017.
- ↑ Butler J. and Montzka S. (2020). "The NOAA Annual Greenhouse Gas Index (AGGI)". NOAA Global Monitoring Laboratory/Earth System Research Laboratories. https://www.esrl.noaa.gov/gmd/aggi/aggi.html.
- ↑ Charles J. Kibert (2016). "Background". Sustainable Construction: Green Building Design and Delivery. Wiley. ISBN 978-1119055327. https://books.google.com/books?id=qv3iCwAAQBAJ&pg=PA698.
- ↑ "Full Mauna Loa CO2 record". Earth System Research Laboratory. 2005. https://www.esrl.noaa.gov/gmd/ccgg/trends/full.html.
- ↑ Tans, Pieter (3 May 2008). "Annual CO2 mole fraction increase (ppm) for 1959–2007". National Oceanic and Atmospheric Administration Earth System Research Laboratory, Global Monitoring Division. ftp://ftp.cmdl.noaa.gov/ccg/co2/trends/co2_gr_mlo.txt. "additional details". http://www.esrl.noaa.gov/gmd/ccgg/trends/.; see also Masarie, K.A.; Tans, P.P. (1995). "Extension and integration of atmospheric carbon dioxide data into a globally consistent measurement record". J. Geophys. Res. 100 (D6): 11593–610. doi:10.1029/95JD00859. Bibcode: 1995JGR...10011593M. https://zenodo.org/record/1231364. Retrieved 26 July 2019.
- ↑ 43.0 43.1 43.2 43.3 43.4 43.5 "Chapter 8". AR5 Climate Change 2013: The Physical Science Basis. https://www.ipcc.ch/report/ar5/wg1/.
- ↑ "Global Monitoring Laboratory". NOAA Earth System Research Laboratories. https://www.esrl.noaa.gov/gmd/.
- ↑ "World Data Centre for Greenhouse Gases". World Meteorological Organization Global Atmosphere Watch Programme and Japan Meteorological Agency. https://gaw.kishou.go.jp/.
- ↑ "Advanced Global Atmospheric Gas Experiment". Massachusetts Institute of Technology. https://agage.mit.edu/.
- ↑ 47.0 47.1 Dentener F. J.; B. Hall; C. Smith, eds. (2021-08-09), Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, https://www.ipcc.ch/report/ar6/wg1/#FullReport
- ↑ "Long-term global trends of atmospheric trace gases". NOAA Earth System Research Laboratories. https://www.esrl.noaa.gov/gmd/hats/data.html.
- ↑ "AGAGE Data and Figures". Massachusetts Institute of Technology. https://agage.mit.edu/data/agage-data.
- ↑ "Chapter 6". TAR Climate Change 2001: The Scientific Basis. p. 358. https://www.ipcc.ch/report/ar3/wg1/.
- ↑ "Chapter 2". AR4 Climate Change 2007: The Physical Science Basis. p. 141. http://www.ipcc.ch/ipccreports/ar4-wg1.htm.
- ↑ Friedlingstein, P., Jones, M., O'Sullivan, M., Andrew, R., Hauck, J., Peters, G., Peters, W., Pongratz, J., Sitch, S., Le Quéré, C. and 66 others (2019) "Global carbon budget 2019". Earth System Science Data, 11(4): 1783–1838. doi:10.5194/essd-11-1783-2019
- ↑ "Figure 8.SM.4". Intergovernmental Panel on Climate Change Fifth Assessment Report - Supplemental Material. p. 8SM-16. https://www.ipcc.ch/site/assets/uploads/2018/07/WGI_AR5.Chap_.8_SM.pdf.
- ↑ Archer, David (2009). "Atmospheric lifetime of fossil fuel carbon dioxide". Annual Review of Earth and Planetary Sciences 37 (1): 117–34. doi:10.1146/annurev.earth.031208.100206. Bibcode: 2009AREPS..37..117A. https://orbi.uliege.be/handle/2268/12933.
- ↑ Joos, F. et al. (2013). "Carbon dioxide and climate impulse response functions for the computation of greenhouse gas metrics: A multi-model analysis". Atmospheric Chemistry and Physics 13 (5): 2793–2825. doi:10.5194/acpd-12-19799-2012. https://www.atmos-chem-phys.net/13/2793/2013/.
- ↑ Hansen, J. et al. (2005). "Efficacy of Climate Forcings". Journal of Geophysical Research: Atmospheres 119 (D18104). doi:10.1029/2005JD005776. Bibcode: 2005JGRD..11018104H.
- ↑ Denman, K.L., G. Brasseur, A. Chidthaisong, P. Ciais, P.M. Cox, R.E. Dickinson, D. Hauglustaine, C. Heinze, E. Holland, D. Jacob, U. Lohmann, S Ramachandran, P.L. da Silva Dias, S.C. Wofsy and X. Zhang, 2007: Chapter 7: Couplings Between Changes in the Climate System and Biogeochemistry. In: Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change [Solomon, S., D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M.Tignor and H.L. Miller (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA.
- ↑ Canadell, J.G.; Le Quere, C.; Raupach, M.R.; Field, C.B.; Buitenhuis, E.T.; Ciais, P.; Conway, T.J.; Gillett, N.P. et al. (2007). "Contributions to accelerating atmospheric CO2 growth from economic activity, carbon intensity, and efficiency of natural sinks". Proc. Natl. Acad. Sci. USA 104 (47): 18866–70. doi:10.1073/pnas.0702737104. PMID 17962418. Bibcode: 2007PNAS..10418866C.
- ↑ "AGU Water Vapor in the Climate System". Eso.org. 27 April 1995. http://www.eso.org/gen-fac/pubs/astclim/espas/pwv/mockler.html.
- ↑ Betts (2001). "6.3 Well-mixed Greenhouse Gases". Chapter 6 Radiative Forcing of Climate Change. Working Group I: The Scientific Basis IPCC Third Assessment Report – Climate Change 2001. UNEP/GRID-Arendal – Publications. http://www.grida.no/publications/other/ipcc%5Ftar/?src=/climate/ipcc_tar/wg1/218.htm. Retrieved 2010-10-16.
- ↑ 61.0 61.1 Jacob, Daniel (1999). Introduction to atmospheric chemistry. Princeton University Press. pp. 25–26. ISBN 978-0691001852. http://www-as.harvard.edu/people/faculty/djj/book/.
- ↑ "How long will global warming last?". RealClimate. 15 March 2005. http://www.realclimate.org/index.php/archives/2005/03/how-long-will-global-warming-last.
- ↑ Held, Isaac M.; Soden, Brian J. (November 2000). "Water vapor feedback and global warming" (in en). Annual Review of Energy and the Environment 25 (1): 441–475. doi:10.1146/annurev.energy.25.1.441. ISSN 1056-3466.
- ↑ Evans, Kimberly Masters (2005). "The greenhouse effect and climate change". The environment: a revolution in attitudes. Detroit: Thomson Gale. ISBN 978-0787690823. https://books.google.com/books?id=DdtzAAAACAAJ.
- ↑ "Chapter 3, IPCC Special Report on Emissions Scenarios, 2000". Intergovernmental Panel on Climate Change. 2000. https://ipcc.ch/pdf/special-reports/spm/sres-en.pdf.
- ↑ LuAnn Dahlman (14 August 2020). "Climate change: annual greenhouse gas index". NOAA Climate.gov science news & Information for a climate smart nation. https://www.climate.gov/news-features/understanding-climate/climate-change-annual-greenhouse-gas-index.
- ↑ "The NOAA Annual Greenhouse Gas Index (AGGI) - An Introduction". NOAA Global Monitoring Laboratory/Earth System Research Laboratories. https://www.esrl.noaa.gov/gmd/aggi/.
- ↑ "Many Planets, One Earth // Section 4: Carbon Cycling and Earth's Climate". Many Planets, One Earth 4. http://www.learner.org/courses/envsci/unit/text.php?unit=1&secNum=4. Retrieved 2012-06-24.
- ↑ 69.0 69.1 "Geoengineering the climate: science, governance and uncertainty". The Royal Society. 2009. http://royalsociety.org/displaypagedoc.asp?id=35151.
- ↑ Fisher, B.S., N. Nakicenovic, K. Alfsen, J. Corfee Morlot, F. de la Chesnaye, J.-Ch. Hourcade, K. Jiang, M. Kainuma, E. La Rovere, A. Matysek, A. Rana, K. Riahi, R. Richels, S. Rose, D. van Vuuren, R. Warren, 2007: Chapter 3: Issues related to mitigation in the long term context, In Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Inter-governmental Panel on Climate Change [B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer (eds)], Cambridge University Press, Cambridge,
- ↑ Jackson, Robert B.; Abernethy, Sam; Canadell, Josep G.; Cargnello, Matteo; Davis, Steven J.; Féron, Sarah; Fuss, Sabine; Heyer, Alexander J. et al. (2021-11-15). "Atmospheric methane removal: a research agenda" (in en). Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 379 (2210): 20200454. doi:10.1098/rsta.2020.0454. ISSN 1364-503X. PMID 34565221. Bibcode: 2021RSPTA.37900454J.
- ↑ "Coal Consumption Affecting Climate". Rodney and Otamatea Times, Waitemata and Kaipara Gazette (Warkworth, New Zealand): p. 7. 14 August 1912. https://paperspast.natlib.govt.nz/newspapers/ROTWKG19120814.2.56.5. Text was earlier published in Popular Mechanics, March 1912, p. 341.
- ↑ Arrhenius, Svante (1896). "On the influence of carbonic acid in the air upon the temperature of the ground". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 41 (251): 237–276. doi:10.1080/14786449608620846. http://www.rsc.org/images/Arrhenius1896_tcm18-173546.pdf. Retrieved 1 December 2020.
- ↑ Arrhenius, Svante (1897). "On the Influence of Carbonic Acid in the Air Upon the Temperature of the Ground". Publications of the Astronomical Society of the Pacific 9 (54): 14. doi:10.1086/121158. Bibcode: 1897PASP....9...14A.
- ↑ Easterbrook, Steve (18 August 2015). "Who first coined the term "Greenhouse Effect"?". http://www.easterbrook.ca/steve/2015/08/who-first-coined-the-term-greenhouse-effect/.
- ↑ Ekholm N (1901). "On The Variations Of The Climate Of The Geological And Historical Past And Their Causes". Quarterly Journal of the Royal Meteorological Society 27 (117): 1–62. doi:10.1002/qj.49702711702. Bibcode: 1901QJRMS..27....1E.
- ↑ Cook, J.; Nuccitelli, D.; Green, S.A.; Richardson, M.; Winkler, B.R.; Painting, R.; Way, R.; Jacobs, P. et al. (2013). "Quantifying the consensus on anthropogenic global warming in the scientific literature". Environmental Research Letters 8 (2): 024024. doi:10.1088/1748-9326/8/2/024024. Bibcode: 2013ERL.....8b4024C.
- ↑ Eddie Schwieterman. "Comparing the Greenhouse Effect on Earth, Mars, Venus, and Titan: Present Day and through Time". http://www.astro.washington.edu/users/eschwiet/essays/greenhouse_ASTR555.pdf.
- ↑ "Scoping of the IPCC 5th Assessment Report Cross Cutting Issues". Thirty-first Session of the IPCC Bali, 26–29 October 2009. https://www.ipcc.ch/site/assets/uploads/2018/03/inf3-6.pdf. Retrieved 24 March 2019.
- ↑ Hansen, James; Sato, Makiko; Russell, Gary; Kharecha, Pushker (2013). "Climate sensitivity, sea level and atmospheric carbon dioxide". Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 371 (2001): 20120294. doi:10.1098/rsta.2012.0294. PMID 24043864. Bibcode: 2013RSPTA.37120294H.
External links
- Carbon Dioxide Information Analysis Center (CDIAC), U.S. Department of Energy, https://cdiac.ess-dive.lbl.gov/, retrieved 2020-07-26
- Annual Greenhouse Gas Index (AGGI) from NOAA
- Atmospheric spectra of GHGs and other trace gases
 |
