Chemistry:Human iron metabolism

Human iron metabolism is the set of chemical reactions that maintain human homeostasis of iron at the systemic and cellular level. Iron is both necessary to the body and potentially toxic. Controlling iron levels in the body is a critically important part of many aspects of human health and disease. Hematologists have been especially interested in systemic iron metabolism, because iron is essential for red blood cells, where most of the human body's iron is contained. Understanding iron metabolism is also important for understanding diseases of iron overload, such as hereditary hemochromatosis, and iron deficiency, such as iron-deficiency anemia.
Importance of iron regulation
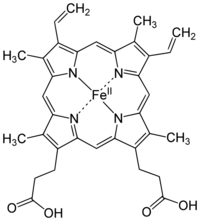
Iron is an essential bioelement for most forms of life, from bacteria to mammals. Its importance lies in its ability to mediate electron transfer. In the ferrous state (Fe2+), iron acts as an electron donor, while in the ferric state (Fe3+) it acts as an acceptor. Thus, iron plays a vital role in the catalysis of enzymatic reactions that involve electron transfer (reduction and oxidation, redox). Proteins can contain iron as part of different cofactors, such as iron–sulfur clusters (Fe-S) and heme groups, both of which are assembled in mitochondria.
Cellular respiration
Human cells require iron in order to obtain energy as ATP from a multi-step process known as cellular respiration, more specifically from oxidative phosphorylation at the mitochondrial cristae. Iron is present in the iron–sulfur cluster and heme groups of the electron transport chain proteins that generate a proton gradient that allows ATP synthase to synthesize ATP (chemiosmosis).
Heme groups are part of hemoglobin, a protein found in red blood cells that serves to transport oxygen from the lungs to other tissues. Heme groups are also present in myoglobin to store and diffuse oxygen in muscle cells.
Oxygen transport
The human body needs iron for oxygen transport. Oxygen (O2) is required for the functioning and survival of nearly all cell types. Oxygen is transported from the lungs to the rest of the body bound to the heme group of hemoglobin in red blood cells. In muscles cells, iron binds oxygen to myoglobin, which regulates its release.
Toxicity
Iron is also potentially toxic. Its ability to donate and accept electrons means that it can catalyze the conversion of hydrogen peroxide into free radicals. Free radicals can cause damage to a wide variety of cellular structures, and ultimately kill the cell.[1]
Iron bound to proteins or cofactors such as heme is safe. Also, there are virtually no truly free iron ions in the cell, since they readily form complexes with organic molecules. However, some of the intracellular iron is bound to low-affinity complexes, and is termed labile iron or "free" iron. Iron in such complexes can cause damage as described above.[2]
To prevent that kind of damage, all life forms that use iron bind the iron atoms to proteins. This binding allows cells to benefit from iron while also limiting its ability to do harm.[1][3] Typical intracellular labile iron concentrations in bacteria are 10-20 micromolar,[4] though they can be 10-fold higher in anaerobic environment,[5] where free radicals and reactive oxygen species are scarcer. In mammalian cells, intracellular labile iron concentrations are typically smaller than 1 micromolar, less than 5 percent of total cellular iron.[2]
Bacterial protection
In response to a systemic bacterial infection, the immune system initiates a process known as "iron withholding". If bacteria are to survive, then they must obtain iron from their environment. Disease-causing bacteria do this in many ways, including releasing iron-binding molecules called siderophores and then reabsorbing them to recover iron, or scavenging iron from hemoglobin and transferrin. The harder the bacteria have to work to get iron, the greater a metabolic price they must pay. That means that iron-deprived bacteria reproduce more slowly. So, control of iron levels appears to be an important defense against many bacterial infections. Certain bacteria species have developed strategies to circumvent that defense, TB causing bacteria can reside within macrophages, which present an iron rich environment and Borrelia burgdorferi uses manganese in place of iron. People with increased amounts of iron, as, for example, in hemochromatosis, are more susceptible to some bacterial infections.[6]
Although this mechanism is an elegant response to short-term bacterial infection, it can cause problems when it goes on so long that the body is deprived of needed iron for red cell production. Inflammatory cytokines stimulate the liver to produce the iron metabolism regulator protein hepcidin, that reduces available iron. If hepcidin levels increase because of non-bacterial sources of inflammation, like viral infection, cancer, auto-immune diseases or other chronic diseases, then the anemia of chronic disease may result. In this case, iron withholding actually impairs health by preventing the manufacture of enough hemoglobin-containing red blood cells.[3]
Body iron stores

Most well-nourished people in industrialized countries have 4 to 5 grams of iron in their bodies (~38 mg iron/kg body weight for women and ~50 mg iron/kg body for men).[7] Of this, about 2.5 g is contained in the hemoglobin needed to carry oxygen through the blood (around 0.5 mg of iron per mL of blood),[8] and most of the rest (approximately 2 grams in adult men, and somewhat less in women of childbearing age) is contained in ferritin complexes that are present in all cells, but most common in bone marrow, liver, and spleen. The liver stores of ferritin are the primary physiologic source of reserve iron in the body. The reserves of iron in industrialized countries tend to be lower in children and women of child-bearing age than in men and in the elderly. Women who must use their stores to compensate for iron lost through menstruation, pregnancy or lactation have lower non-hemoglobin body stores, which may consist of 500 mg, or even less.
Of the body's total iron content, about 400 mg is devoted to cellular proteins that use iron for important cellular processes like storing oxygen (myoglobin) or performing energy-producing redox reactions (cytochromes). A relatively small amount (3–4 mg) circulates through the plasma, bound to transferrin.[9] Because of its toxicity, free soluble iron is kept in low concentration in the body.
Iron deficiency first affects the storage of iron in the body, and depletion of these stores is thought to be relatively asymptomatic, although some vague and non-specific symptoms have been associated with it. Since iron is primarily required for hemoglobin, iron deficiency anemia is the primary clinical manifestation of iron deficiency. Iron-deficient people will suffer or die from organ damage well before their cells run out of the iron needed for intracellular processes like electron transport.
Macrophages of the reticuloendothelial system store iron as part of the process of breaking down and processing hemoglobin from engulfed red blood cells. Iron is also stored as a pigment called hemosiderin, which is an ill-defined deposit of protein and iron, created by macrophages where excess iron is present, either locally or systemically, e.g., among people with iron overload due to frequent blood cell destruction and the necessary transfusions their condition calls for. If systemic iron overload is corrected, over time the hemosiderin is slowly resorbed by the macrophages.
Mechanisms of iron regulation
Human iron homeostasis is regulated at two different levels. Systemic iron levels are balanced by the controlled absorption of dietary iron by enterocytes, the cells that line the interior of the intestines, and the uncontrolled loss of iron from epithelial sloughing, sweat, injuries and blood loss. In addition, systemic iron is continuously recycled. Cellular iron levels are controlled differently by different cell types due to the expression of particular iron regulatory and transport proteins.
Systemic iron regulation
Dietary iron uptake
The absorption of dietary iron is a variable and dynamic process. The amount of iron absorbed compared to the amount ingested is typically low, but may range from 5% to as much as 35% depending on circumstances and type of iron. The efficiency with which iron is absorbed varies depending on the source. Generally, the best-absorbed forms of iron come from animal products. Absorption of dietary iron in iron salt form (as in most supplements) varies somewhat according to the body's need for iron, and is usually between 10% and 20% of iron intake. Absorption of iron from animal products, and some plant products, is in the form of heme iron, and is more efficient, allowing absorption of from 15% to 35% of intake. Heme iron in animals is from blood and heme-containing proteins in meat and mitochondria, whereas in plants, heme iron is present in mitochondria in all cells that use oxygen for respiration.
Like most mineral nutrients, the majority of the iron absorbed from digested food or supplements is absorbed in the duodenum by enterocytes of the duodenal lining. These cells have special molecules that allow them to move iron into the body. To be absorbed, dietary iron can be absorbed as part of a protein such as heme protein or iron must be in its ferrous Fe2+ form. A ferric reductase enzyme on the enterocytes' brush border, duodenal cytochrome B (Dcytb), reduces ferric Fe3+ to Fe2+.[10] A protein called divalent metal transporter 1 (DMT1), which can transport several divalent metals across the plasma membrane, then transports iron across the enterocyte's cell membrane into the cell. If the iron is bound to heme, it is instead transported across the apical membrane by heme carrier protein 1 (HCP1).[11] Heme is then catabolized by microsomal heme oxygenase into biliverdin, releasing Fe2+.[12]
These intestinal lining cells can then either store the iron as ferritin, which is accomplished by Fe2+ binding to apoferritin (in which case the iron will leave the body when the cell dies and is sloughed off into feces), or the cell can release it into the body via the only known iron exporter in mammals, ferroportin. Hephaestin, a ferroxidase that can oxidize Fe2+ to Fe3+ and is found mainly in the small intestine, helps ferroportin transfer iron across the basolateral end of the intestine cells. Upon release into the bloodstream, Fe3+ binds transferrin and circulates to tissues. In contrast, ferroportin is post-translationally repressed by hepcidin, a 25-amino acid peptide hormone. The body regulates iron levels by regulating each of these steps. For instance, enterocytes synthesize more Dcytb, DMT1 and ferroportin in response to iron deficiency anemia.[13] Iron absorption from diet is enhanced in the presence of vitamin C and diminished by excess calcium, zinc, or manganese.[14]
The human body's rate of iron absorption appears to respond to a variety of interdependent factors, including total iron stores, the extent to which the bone marrow is producing new red blood cells, the concentration of hemoglobin in the blood, and the oxygen content of the blood. The body also absorbs less iron during times of inflammation, in order to deprive bacteria of iron. Recent discoveries demonstrate that hepcidin regulation of ferroportin is responsible for the syndrome of anemia of chronic disease.
Iron recycling and loss
Most of the iron in the body is hoarded and recycled by the reticuloendothelial system, which breaks down aged red blood cells. In contrast to iron uptake and recycling, there is no physiologic regulatory mechanism for excreting iron. People lose a small but steady amount by gastrointestinal blood loss, sweating and by shedding cells of the skin and the mucosal lining of the gastrointestinal tract. The total amount of loss for healthy people in the developed world amounts to an estimated average of 1 mg a day for men, and 1.5–2 mg a day for women with regular menstrual periods.[15] People with gastrointestinal parasitic infections, more commonly found in developing countries, often lose more.[1] Those who cannot regulate absorption well enough get disorders of iron overload. In these diseases, the toxicity of iron starts overwhelming the body's ability to bind and store it.[16]
Cellular iron regulation
Iron import
Most cell types take up iron primarily through receptor-mediated endocytosis via transferrin receptor 1 (TFR1), transferrin receptor 2 (TFR2) and GAPDH. TFR1 has a 30-fold higher affinity for transferrin-bound iron than TFR2 and thus is the main player in this process.[17][18] The higher order multifunctional glycolytic enzyme glyceraldehyde-3-phosphate dehydrogenase (GAPDH) also acts as a transferrin receptor.[19][20] Transferrin-bound ferric iron is recognized by these transferrin receptors, triggering a conformational change that causes endocytosis. Iron then enters the cytoplasm from the endosome via importer DMT1 after being reduced to its ferrous state by a STEAP family reductase.[21]
Alternatively, iron can enter the cell directly via plasma membrane divalent cation importers such as DMT1 and ZIP14 (Zrt-Irt-like protein 14).[22] Again, iron enters the cytoplasm in the ferrous state after being reduced in the extracellular space by a reductase such as STEAP2, STEAP3 (in red blood cells), Dcytb (in enterocytes) and SDR2.[21]
Iron import in some cancer cells
Iron can also enter cells via CD44 in complexes bound to hyaluronic acid during epithelial–mesenchymal transition (EMT). In this process, epithelial cells transform into mesenchymal cells with detachment from the basement membrane, to which they’re normally anchored, paving the way for the newly differentiated motile mesenchymal cells to begin migration away from the epithelial layer.[23][24]
While EMT plays a crucial role in physiological processes like implantation, where it enables the embryo to invade the endometrium to facilitate placental attachment, its dysregulation can also fuel the malignant spread of tumors empowering them to invade surrounding tissues and establish distant colonies (metastasis).[24]
Malignant cells often exhibit a heightened demand for iron, fueling their transition towards a more invasive mesenchymal state. This iron is necessary for the expression of mesenchymal genes, like those encoding transforming growth factor beta (TGF-β), crucial for EMT. Notably, iron’s unique ability to catalyze protein and DNA demethylation plays a vital role in this gene expression process.[23]
Conventional iron uptake pathways, such as those using the transferrin receptor 1 (TfR1), often prove insufficient to meet these elevated iron demands in cancer cells. As a result, various cytokines and growth factors trigger the upregulation of CD44, a surface molecule capable of internalizing iron bound to the hyaluronan complex. This alternative pathway, relying on CD44-mediated endocytosis, becomes the dominant iron uptake mechanism compared to the traditional TfR1-dependent route.[23][24]
The labile iron pool
In the cytoplasm, ferrous iron is found in a soluble, chelatable state which constitutes the labile iron pool (~0.001 mM).[25] In this pool, iron is thought to be bound to low-mass compounds such as peptides, carboxylates and phosphates, although some might be in a free, hydrated form (aqua ions).[25] Alternatively, iron ions might be bound to specialized proteins known as metallochaperones.[26] Specifically, poly-r(C)-binding proteins PCBP1 and PCBP2 appear to mediate transfer of free iron to ferritin (for storage) and non-heme iron enzymes (for use in catalysis).[22][27] The labile iron pool is potentially toxic due to iron's ability to generate reactive oxygen species. Iron from this pool can be taken up by mitochondria via mitoferrin to synthesize Fe-S clusters and heme groups.[21]
The storage iron pool
Iron can be stored in ferritin as ferric iron due to the ferroxidase activity of the ferritin heavy chain.[28] Dysfunctional ferritin may accumulate as hemosiderin, which can be problematic in cases of iron overload.[29] The ferritin storage iron pool is much larger than the labile iron pool, ranging in concentration from 0.7 mM to 3.6 mM.[25]
Iron export
Iron export occurs in a variety of cell types, including neurons, red blood cells, macrophages and enterocytes. The latter two are especially important since systemic iron levels depend upon them. There is only one known iron exporter, ferroportin.[30] It transports ferrous iron out of the cell, generally aided by ceruloplasmin and/or hephaestin (mostly in enterocytes), which oxidize iron to its ferric state so it can bind ferritin in the extracellular medium.[21] Hepcidin causes the internalization of ferroportin, decreasing iron export. Besides, hepcidin seems to downregulate both TFR1 and DMT1 through an unknown mechanism.[31] Another player assisting ferroportin in effecting cellular iron export is GAPDH.[32] A specific post translationally modified isoform of GAPDH is recruited to the surface of iron loaded cells where it recruits apo-transferrin in close proximity to ferroportin so as to rapidly chelate the iron extruded.[33]
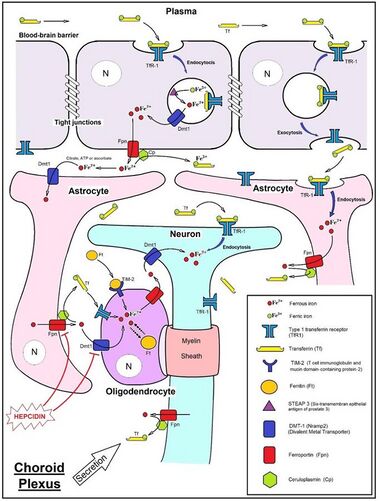
- The transcytosis pathway (illustrated in the upper right segment of the image), where the complex “Fe3+-transferrin-transferrin receptor 1 (TfR1)” undergoes endocytosis and exocytosis from the luminal pole to the cerebral extracellular matrix (ECM) and interstitial fluid.
- The facilitated transporter pathway, where endothelial cells internalize the complex “Fe3+-transferrin-transferrin receptor 1 (TfR1)” in endosome, reduce ferric Fe3+ ion to ferrous Fe2+ ion by STEAP3 enzyme and then Fe2+ ion crosses the endosomal membrane thanks to DMT1. Fe2+ is then exported to the extracellular matrix (ECM) and interstitial fluid, via ferroportin coupled with ceruloplasmin.
The expression of hepcidin, which only occurs in certain cell types such as hepatocytes, is tightly controlled at the transcriptional level and it represents the link between cellular and systemic iron homeostasis due to hepcidin's role as "gatekeeper" of iron release from enterocytes into the rest of the body.[21] Erythroblasts produce erythroferrone, a hormone which inhibits hepcidin and so increases the availability of iron needed for hemoglobin synthesis.[35]
Translational control of cellular iron
Although some control exists at the transcriptional level, the regulation of cellular iron levels is ultimately controlled at the translational level by iron-responsive element-binding proteins IRP1 and especially IRP2.[36] When iron levels are low, these proteins are able to bind to iron-responsive elements (IREs). IREs are stem loop structures in the untranslated regions (UTRs) of mRNA.[21]
Both ferritin and ferroportin contain an IRE in their 5' UTRs, so that under iron deficiency their translation is repressed by IRP2, preventing the unnecessary synthesis of storage protein and the detrimental export of iron. In contrast, TFR1 and some DMT1 variants contain 3' UTR IREs, which bind IRP2 under iron deficiency, stabilizing the mRNA, which guarantees the synthesis of iron importers.[21]
Pathology
Iron deficiency
Functional or actual iron deficiency can result from a variety of causes. These causes can be grouped into several categories:
- Increased demand for iron, which the diet cannot accommodate.
- Increased loss of iron (usually through loss of blood).
- Nutritional deficiency. This can result due to a lack of dietary iron or consumption of foods that inhibit iron absorption. Absorption inhibition has been observed caused by phytates in bran,[37] calcium from supplements or dairy products,[38] and tannins from tea,[39] although in all three of these studies the effect was small and the authors of the studies cited regarding bran and tea note that the effect will probably only have a noticeable impact when most iron is obtained from vegetable sources.
- Acid-reducing medications: Acid-reducing medications reduce the absorption of dietary iron. These medications are commonly used for gastritis, reflux disease, and ulcers. Proton pump inhibitors (PPIs), H2 antihistamines, and antacids will reduce iron metabolism.[40]
- Damage to the intestinal lining. Examples of causes of this kind of damage include surgery involving the duodenum or diseases like Crohn's or celiac sprue which severely reduce the surface area available for absorption. Helicobacter pylori infections also reduce the availability of iron.[41]
- Inflammation leading to hepcidin-induced restriction on iron release from enterocytes (see above).
- Is also a common occurrence in pregnant women, and in growing adolescents due to poor diets.
- Acute blood loss or acute liver cirrhosis creates a lack of transferrin therefore causing iron to be secreted from the body.
Iron overload
The body is able to substantially reduce the amount of iron it absorbs across the mucosa. It does not seem to be able to entirely shut down the iron transport process. Also, in situations where excess iron damages the intestinal lining itself (for instance, when children eat a large quantity of iron tablets produced for adult consumption), even more iron can enter the bloodstream and cause a potentially deadly syndrome of iron overload. Large amounts of free iron in the circulation will cause damage to critical cells in the liver, the heart and other metabolically active organs.
Iron toxicity results when the amount of circulating iron exceeds the amount of transferrin available to bind it, but the body is able to vigorously regulate its iron uptake. Thus, iron toxicity from ingestion is usually the result of extraordinary circumstances like iron tablet over-consumption[1][42] rather than variations in diet. The type of acute toxicity from iron ingestion causes severe mucosal damage in the gastrointestinal tract, among other problems.
Excess iron has been linked to higher rates of disease and mortality. For example, breast cancer patients with low ferroportin expression (leading to higher concentrations of intracellular iron) survive for a shorter period of time on average, while high ferroportin expression predicts 90% 10-year survival in breast cancer patients.[43] Similarly, genetic variations in iron transporter genes known to increase serum iron levels also reduce lifespan and the average number of years spent in good health.[44] It has been suggested that mutations that increase iron absorption, such as the ones responsible for hemochromatosis (see below), were selected for during Neolithic times as they provided a selective advantage against iron-deficiency anemia.[45] The increase in systemic iron levels becomes pathological in old age, which supports the notion that antagonistic pleiotropy or "hyperfunction" drives human aging.[44]
Chronic iron toxicity is usually the result of more chronic iron overload syndromes associated with genetic diseases, repeated transfusions or other causes. In such cases the iron stores of an adult may reach 50 grams (10 times normal total body iron) or more. The most common diseases of iron overload are hereditary hemochromatosis (HH), caused by mutations in the HFE gene, and the more severe disease juvenile hemochromatosis (JH), caused by mutations in either hemojuvelin (HJV)[46] or hepcidin (HAMP). The exact mechanisms of most of the various forms of adult hemochromatosis, which make up most of the genetic iron overload disorders, remain unsolved. So, while researchers have been able to identify genetic mutations causing several adult variants of hemochromatosis, they now must turn their attention to the normal function of these mutated genes.
See also
References
- ↑ 1.0 1.1 1.2 "Disorders of iron metabolism". The New England Journal of Medicine 342 (17): 1293–4. Apr 2000. doi:10.1056/NEJM200004273421716. PMID 10787338.
- ↑ 2.0 2.1 "The labile iron pool: characterization, measurement, and participation in cellular processes". Free Radical Biology and Medicine 33 (8): 1037–1046. 2002. doi:10.1016/s0891-5849(02)01006-7. PMID 12374615.
- ↑ 3.0 3.1 "Disorders of iron metabolism". The New England Journal of Medicine 341 (26): 1986–95. Dec 1999. doi:10.1056/NEJM199912233412607. PMID 10607817.
- ↑ "Intracellular free iron and its potential role in ultrahigh-pressure-induced inactivation of Escherichia coli". Applied and Environmental Microbiology 79 (2): 722–724. Jan 2013. doi:10.1128/aem.02202-12. PMID 23124235. Bibcode: 2013ApEnM..79..722Y.
- ↑ "Regulation of the intracellular free iron pool by Dpr provides oxygen tolerance to Streptococcus mutans.". Journal of Bacteriology 186 (18): 5997–6002. 2004. doi:10.1128/jb.186.18.5997-6002.2004. PMID 15342568.
- ↑ "Hepcidin, a key regulator of iron metabolism and mediator of anemia of inflammation". Blood 102 (3): 783–8. Aug 2003. doi:10.1182/blood-2003-03-0672. PMID 12663437.
- ↑ Gropper, Sareen S.; Smith, Jack L. (2013). Advanced Nutrition and Human Metabolism (6th ed.). Belmont, CA: Wadsworth. p. 481. ISBN 978-1133104056. https://books.google.com/books?id=3R0Yeu79jfQC&pg=PA481.
- ↑ Truswell, A. Stewart (2010-07-15) (in en). ABC of Nutrition. John Wiley & Sons. pp. 52. ISBN 9781444314229. https://books.google.com/books?id=01JfkuEaL4EC&pg=PA52.
- ↑ "Regulation of iron balance". UpToDate. 2011-11-07. http://www.uptodate.com/contents/regulation-of-iron-balance.
- ↑ "An iron-regulated ferric reductase associated with the absorption of dietary iron". Science 291 (5509): 1755–9. Mar 2001. doi:10.1126/science.1057206. PMID 11230685. Bibcode: 2001Sci...291.1755M.
- ↑ Rouault, Tracey A. (2005-09-09). "The Intestinal Heme Transporter Revealed" (in en). Cell 122 (5): 649–651. doi:10.1016/j.cell.2005.08.027. ISSN 0092-8674. PMID 16143096.
- ↑ Przybyszewska, Justyna; Żekanowska, Ewa (2014). "The role of hepcidin, ferroportin, HCP1, and DMT1 protein in iron absorption in the human digestive tract". Gastroenterology Review 4 (4): 208–213. doi:10.5114/pg.2014.45102. PMID 25276251.
- ↑ "Orchestration of iron homeostasis". The New England Journal of Medicine 352 (17): 1741–4. Apr 2005. doi:10.1056/NEJMp048363. PMID 15858181.
- ↑ Abbaspour, Nazanin (Feb 2014). "Review on iron and its importance for human health". J Res Med Sci 19 (2): 164–174. PMID 24778671.
- ↑ Janet, R Hunt (June 2009). "Body iron excretion by healthy men and women". The American Journal of Clinical Nutrition 89 (6): 1792–1798. doi:10.3945/ajcn.2009.27439. PMID 19386738.
- ↑ "Iron overload syndromes other than hereditary hemochromatosis". UpToDate. 2011-11-07. http://www.uptodate.com/contents/iron-overload-syndromes-other-than-hereditary-hemochromatosis.
- ↑ "Transferrin receptor 2-alpha supports cell growth both in iron-chelated cultured cells and in vivo". The Journal of Biological Chemistry 275 (22): 16618–25. June 2000. doi:10.1074/jbc.M908846199. PMID 10748106.
- ↑ "Comparison of the interactions of transferrin receptor and transferrin receptor 2 with transferrin and the hereditary hemochromatosis protein HFE". The Journal of Biological Chemistry 275 (49): 38135–8. December 2000. doi:10.1074/jbc.C000664200. PMID 11027676.
- ↑ "Characterization of glyceraldehyde-3-phosphate dehydrogenase as a novel transferrin receptor". The International Journal of Biochemistry & Cell Biology 44 (1): 189–99. Jan 2012. doi:10.1016/j.biocel.2011.10.016. PMID 22062951.
- ↑ "Secreted glyceraldehye-3-phosphate [sic] dehydrogenase is a multifunctional autocrine transferrin receptor for cellular iron acquisition". Biochimica et Biophysica Acta (BBA) - General Subjects 1830 (6): 3816–27. Jun 2013. doi:10.1016/j.bbagen.2013.03.019. PMID 23541988.
- ↑ 21.0 21.1 21.2 21.3 21.4 21.5 21.6 "Two to tango: regulation of Mammalian iron metabolism". Cell 142 (1): 24–38. Jul 2010. doi:10.1016/j.cell.2010.06.028. PMID 20603012.
- ↑ 22.0 22.1 Lane, D.J.R.; Merlot, A.M.; Huang, M.L.-H.; Bae, D.-H.; Jansson, P.J.; Sahni, S.; Kalinowski, D.S.; Richardson, D.R. (May 2015). "Cellular iron uptake, trafficking and metabolism: Key molecules and mechanisms and their roles in disease". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 1853 (5): 1130–1144. doi:10.1016/j.bbamcr.2015.01.021. PMID 25661197.
- ↑ 23.0 23.1 23.2 Müller, Sebastian; Sindikubwabo, Fabien; Cañeque, Tatiana; Lafon, Anne; Versini, Antoine; Lombard, Bérangère; Loew, Damarys; Wu, Ting-Di et al. (1 October 2020). "CD44 regulates epigenetic plasticity by mediating iron endocytosis". Nature chemistry 12 (10): 929–938. doi:10.1038/s41557-020-0513-5. ISSN 1755-4330. PMID 32747755. PMC 7612580. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7612580/.
- ↑ 24.0 24.1 24.2 Kalluri, Raghu; Weinberg, Robert A. (1 June 2009). "The basics of epithelial-mesenchymal transition". The Journal of Clinical Investigation 119 (6): 1420–1428. doi:10.1172/JCI39104. ISSN 0021-9738. PMID 19487818. PMC 2689101. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2689101/#:~:text=An%20epithelial%2Dmesenchymal%20transition%20(EMT,which%20includes%20enhanced%20migratory%20capacity%2C.
- ↑ 25.0 25.1 25.2 Yehuda, Shlomo; Mostofsky, David I., eds (2010). Iron Deficiency and Overload From Basic Biology to Clinical Medicine. Nutrition and Health. New York, NY: Humana Press. p. 230. doi:10.1007/978-1-59745-462-9. ISBN 9781934115220. https://www.springer.com/gp/book/9781934115220.
- ↑ "Transition metal speciation in the cell: insights from the chemistry of metal ion receptors". Science 300 (5621): 931–6. May 2003. doi:10.1126/science.1085049. PMID 12738850. Bibcode: 2003Sci...300..931F.
- ↑ Philpott, Caroline C.; Ryu, Moon-Suhn (22 July 2014). "Special delivery: distributing iron in the cytosol of mammalian cells". Frontiers in Pharmacology 5: 173. doi:10.3389/fphar.2014.00173. PMID 25101000.
- ↑ "Ferritin, iron homeostasis, and oxidative damage". Free Radical Biology & Medicine 33 (4): 457–63. Aug 2002. doi:10.1016/s0891-5849(02)00842-0. PMID 12160928.
- ↑ "Brain iron metabolism". Seminars in Pediatric Neurology 13 (3): 142–8. Sep 2006. doi:10.1016/j.spen.2006.08.002. PMID 17101452.
- ↑ "Cellular iron: ferroportin is the only way out". Cell Metabolism 1 (3): 155–7. Mar 2005. doi:10.1016/j.cmet.2005.02.005. PMID 16054057.
- ↑ "Hepcidin directly inhibits transferrin receptor 1 expression in astrocytes via a cyclic AMP-protein kinase A pathway". Glia 59 (6): 936–45. Jun 2011. doi:10.1002/glia.21166. PMID 21438013.
- ↑ Boradia, Vishant Mahendra; Raje, Manoj; Raje, Chaaya Iyengar (1 December 2014). "Protein moonlighting in iron metabolism: glyceraldehyde-3-phosphate dehydrogenase (GAPDH)". Biochemical Society Transactions 42 (6): 1796–1801. doi:10.1042/BST20140220. PMID 25399609.
- ↑ "Moonlighting cell-surface GAPDH recruits apotransferrin to effect iron egress from mammalian cells". Journal of Cell Science 127 (Pt 19): 4279–91. Oct 2014. doi:10.1242/jcs.154005. PMID 25074810.
- ↑ Berthou, Christian; Iliou, Jean Paul; Barba, Denis (5 December 2021). "Iron, neuro‐bioavailability and depression". EJHaem 3 (1): 263–275. doi:10.1002/jha2.321. ISSN 2688-6146. PMID 35846210. PMC 9175715. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9175715/.
- ↑ "Identification of erythroferrone as an erythroid regulator of iron metabolism". Nature Genetics 46 (7): 678–84. Jul 2014. doi:10.1038/ng.2996. PMID 24880340.
- ↑ "Systemic iron homeostasis and the iron-responsive element/iron-regulatory protein (IRE/IRP) regulatory network". Annual Review of Nutrition 28: 197–213. 2008. doi:10.1146/annurev.nutr.28.061807.155521. PMID 18489257.
- ↑ Hallberg L (1987). "Wheat fiber, phytates and iron absorption.". Scand J Gastroenterol Suppl 129: 73–9. doi:10.3109/00365528709095855. PMID 2820048.
- ↑ Lynch SR (2000). "The effect of calcium on iron absorption.". Nutr Res Rev 13 (2): 141–58. doi:10.1079/095442200108729043. PMID 19087437.
- ↑ "The effect of tea on iron absorption.". Gut 16 (3): 193–200. 1975. doi:10.1136/gut.16.3.193. PMID 1168162.
- ↑ "UpToDate". https://www.uptodate.com/contents/causes-and-diagnosis-of-iron-deficiency-and-iron-deficiency-anemia-in-adults.
- ↑ Annibale, Bruno; Capurso, Gabriele; Martino, Gina; Grossi, Cristina; Delle Fave, Gianfranco (December 2000). "Iron deficiency anaemia and Helicobacter pylori infection". International Journal of Antimicrobial Agents 16 (4): 515–519. doi:10.1016/s0924-8579(00)00288-0. PMID 11118871.
- ↑ Rudolph CD (2003). Rudolph's pediatrics. New York: McGraw-Hill, Medical Pub. Division. ISBN 978-0-07-112457-7.
- ↑ Pinnix, Zandra K.; Miller, Lance D.; Wang, Wei; D'Agostino, Ralph; Kute, Tim; Willingham, Mark C.; Hatcher, Heather; Tesfay, Lia et al. (2010-08-04). "Ferroportin and Iron Regulation in Breast Cancer Progression and Prognosis" (in en). Science Translational Medicine 2 (43): 43ra56. doi:10.1126/scitranslmed.3001127. ISSN 1946-6234. PMID 20686179.
- ↑ 44.0 44.1 Timmers, Paul R. H. J.; Wilson, James F.; Joshi, Peter K.; Deelen, Joris (Jul 2020). "Multivariate genomic scan implicates novel loci and haem metabolism in human ageing". Nature Communications 11 (3570): 3570. doi:10.1038/s41467-020-17312-3. PMID 32678081. Bibcode: 2020NatCo..11.3570T.
- ↑ Rametta, Raffaela; Meroni, Marica; Dongiovanni, Paola (15 May 2020). "From Environment to Genome and Back: A Lesson from HFE Mutations". International Journal of Molecular Sciences 21 (10): 3505. doi:10.3390/ijms21103505. PMID 32429125.
- ↑ "Molecular biology, genetics and biochemistry of the repulsive guidance molecule family". The Biochemical Journal 422 (3): 393–403. Sep 2009. doi:10.1042/BJ20090978. PMID 19698085.
Further reading
- Andrews, Simon; Norton, Ian; Salunkhe, Arvindkumar S.; Goodluck, Helen; Aly, Wafaa S.M.; Mourad-Agha, Hanna; Cornelis, Pierre (2013). "Chapter 7, Control of Iron Metabolism in Bacteria". in Banci, Lucia. Metallomics and the Cell. Metal Ions in Life Sciences. 12. Springer. pp. 203–39. doi:10.1007/978-94-007-5561-1_7. ISBN 978-94-007-5560-4. electronic-book ISBN:978-94-007-5561-1 ISSN 1559-0836 electronic-ISSN 1868-0402
- "Anemia of inflammation: the cytokine-hepcidin link". The Journal of Clinical Investigation 113 (9): 1251–3. May 2004. doi:10.1172/JCI21441. PMID 15124013.
- "Understanding iron homeostasis through genetic analysis of hemochromatosis and related disorders". Blood 106 (12): 3710–7. Dec 2005. doi:10.1182/blood-2005-05-1857. PMID 16030190.
- "Iron imports. I. Intestinal iron absorption and its regulation". American Journal of Physiology. Gastrointestinal and Liver Physiology 289 (4): G631–5. Oct 2005. doi:10.1152/ajpgi.00220.2005. PMID 16160078.
- "Iron". Nutrition (4th ed.). Sudbury, Massachusetts: Jones and Bartlett Publishers. 2011. pp. 510–514. ISBN 978-0-7637-7663-3. https://books.google.com/books?id=u1p_G8CJ-mwC&pg=PA510. Retrieved June 25, 2012. See esp. pp. 513-514.
- "Dietary diversification and modification of iron". Handbook of Nutrition and Pregnancy. Nutrition & Health. Totowa, New Jersey: Humana Press. 2008. pp. 350–351. doi:10.1007/978-1-59745-112-3. ISBN 978-1-59745-112-3. https://books.google.com/books?id=29EhDBLoPGEC&pg=PA350. Retrieved June 25, 2012.
- Panel on Micronutrients; Subcommittees on Upper Reference Levels of Nutrients and of Interpretation and Uses of Dietary Reference Intakes; the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes (2001). "Iron". Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. Washington, D.C: Food and Nutrition Board, Institute of Medicine. pp. 290–393. ISBN 978-0-309-07279-3. http://books.nap.edu/openbook.php?record_id=10026&page=290. Retrieved June 25, 2012.
- Reilly C (2004). "Iron". The Nutritional Trace Metals. Oxford, UK & Ames, Iowa: Blackwell Publishing. pp. 35–81. ISBN 978-1-4051-1040-2. https://books.google.com/books?id=825s4l-wS6AC. Retrieved June 25, 2012.
External links
- A comprehensive NIH factsheet on iron and nutrition
- Iron Disorders Institute: A nonprofit group concerned with iron disorders; site has helpful links and information on iron-related medical disorders.
- An interactive medical learning portal on iron metabolism
- Information about iron outside the body
 |