Chemistry:Neodymium(III) sulfide
36px 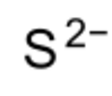
| |
| Names | |
|---|---|
| IUPAC names
Dineodymium trisulfide
| |
| Other names
Neodymium sulfide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| Properties | |
| Nd2S3 | |
| Molar mass | 384.66 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Neodymium(III) sulfide is a inorganic chemical compound with the formula Nd2S3[2] composed of a two neodymium atoms in the +3 oxidation state and three sulfur atoms in the +2 oxidation state. Like other rare earth sulfides, neodymium(III) sulfide is used as a high-performance inorganic pigment.[3]
Preparation
Neodymium(III) sulfide can directly be produced by reacting neodymium with sulfur:[4][5]
- 2Nd + 3S → Nd2S3
It can also be produced by sulfidizing neodymium oxide with H2S at 1450 °C:[4][6]
- Nd2O3 + 3 H2S → Nd2S3 + 3 H2O
Properties
Neodymium(III) sulfide is (as γ-form) a light green solid. The compound comes in three forms.[3] The α-form has an orthorhombic crystal structure, the β form has a tetragonal crystal structure, and the γ form has a cubic crystal structure. At 1650 °C in a vacuum, the γ compound decomposes to form neodymium monosulfide.[6]
Neodymium(III) sulfide has a high melting point and a lot of polymorphic forms which make it difficult to grow.[2] When heated, neodymium sulfide can lose sulfur atoms and can form a range of compositions between Nd2S3 and Nd3S4. Neodymium(III) sulfide is an electrical insulator.[4]
See also
References
- ↑ "Neodymium sulfide (Nd2S3)" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/Neodymium-sulfide-_Nd2S3.
- ↑ 2.0 2.1 Uspenskaya, S. I.; Eliseev, A. A.; Fedorov, A. A. (1975), Sheftal’, N. N.; Givargizov, E. I., eds., "Vapor Growth of Lanthanum and Neodymium Sulfide Crystals" (in en), РОСТ КРИСТАЛЛОВ/Rost Kristallov/Growth of Crystals (Boston, MA: Springer New York): pp. 257–260, doi:10.1007/978-1-4684-1689-3_55, ISBN 978-1-4684-1691-6, http://link.springer.com/10.1007/978-1-4684-1689-3_55, retrieved 2023-04-22
- ↑ 3.0 3.1 Faulkner, Edwin B.; Schwartz, Russell J. (2009-03-09) (in en). High Performance Pigments. John Wiley & Sons. ISBN 978-3-527-62692-2. https://books.google.com/books?id=ecD6AuaJyFwC&pg=PA28.
- ↑ 4.0 4.1 4.2 Cotton, Simon (2006). Lanthanide and Actinide Chemistry. John Wiley & Sons Ltd.
- ↑ A. W. Sleight and D. P. Kelly (1973) (in German), Rare-earth sesquisulfides, Ln2S3, Inorganic Syntheses, 14, McGraw-Hill Book Company, Inc., pp. 152–155
- ↑ 6.0 6.1 Meyer, G.; Morss, Lester R. (1990-12-31) (in en). Synthesis of Lanthanide and Actinide Compounds. Springer Science & Business Media. ISBN 978-0-7923-1018-1. https://books.google.com/books?id=bnS5elHL2w8C&pg=PA329.
 |

