Chemistry:Barium sulfide
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
| 13627 | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| BaS | |
| Molar mass | 169.39 g/mol |
| Appearance | white solid |
| Density | 4.25 g/cm3 [1] |
| Melting point | 2,235[2] °C (4,055 °F; 2,508 K) |
| Boiling point | decomposes |
| 2.88 g/100 mL (0 °C) 7.68 g/100 mL (20 °C) 60.3 g/100 mL (100 °C) (reacts) | |
| Solubility | insoluble in alcohol |
Refractive index (nD)
|
2.155 |
| Structure | |
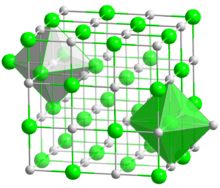
| Halite (cubic), cF8 | |
| Fm3m, No. 225 | |
| Octahedral (Ba2+); octahedral (S2−) | |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| H315, H319, H335, H400 | |
| P261, P264, P271, P273, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P391, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
226 mg/kg humans |
| Related compounds | |
Other anions
|
Barium oxide Barium selenide Barium telluride Barium polonide |
Other cations
|
Beryllium sulfide Magnesium sulfide Calcium sulfide Strontium sulfide Radium sulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Barium sulfide is the inorganic compound with the formula BaS. BaS is the barium compound produced on the largest scale.[3] It is an important precursor to other barium compounds including barium carbonate and the pigment lithopone, ZnS/BaSO4.[4] Like other chalcogenides of the alkaline earth metals, BaS is a short wavelength emitter for electronic displays.[5] It is colorless, although like many sulfides, it is commonly obtained in impure colored forms.
Discovery
BaS was prepared by the Italian alchemist Vincenzo Cascariolo (also known as Vincentius or Vincentinus Casciarolus or Casciorolus, 1571–1624) via the thermo-chemical reduction of BaSO4 (available as the mineral barite).[6] It is currently manufactured by an improved version of Cascariolo's process using coke in place of flour and charcoal. This kind of conversion is called a carbothermic reaction:
- BaSO
4 + 2C → BaS + 2CO
2
and also:
- BaSO
4 + 4C → BaS + 4CO
The basic method remains in use today. BaS dissolves in water. These aqueous solutions, when treated with sodium carbonate or carbon dioxide, give a white solid of barium carbonate, a source material for many commercial barium compounds.[7]
According to Harvey (1957),[8] in 1603, Vincenzo Cascariolo used barite, found at the bottom of Mount Paterno near Bologna, in one of his non-fruitful attempts to produce gold. After grinding and heating the mineral with charcoal under reducing conditions, he obtained a persistent luminescent material that soon came to be known as Lapis Boloniensis, or Bolognian stone.[9][10] The phosphorescence of the material obtained by Casciarolo made it a curiosity.[11][12][13]
Preparation
A modern procedure proceeds from barium carbonate:[14]
- BaCO
3 + H
2S → BaS + H
2O + CO
2
BaS crystallizes with the NaCl structure, featuring octahedral Ba2+ and S2− centres.
The observed melting point of barium sulfide is highly sensitive to impurities.[2]
Safety
BaS is quite poisonous, as are related sulfides, such as CaS, which evolve toxic hydrogen sulfide upon contact with water. Barium itself is also toxic.
References
- ↑ Lide, David R., ed (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. ISBN 0-8493-0487-3.
- ↑ 2.0 2.1 Stinn, C., Nose, K., Okabe, T. et al. Metall and Materi Trans B (2017) 48: 2922. https://doi.org/10.1007/s11663-017-1107-5
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- ↑ Vij, D. R.; Singh, N. (1992). "Optical and electrical properties of II-VI wide gap semiconducting barium sulfide". Conf. Phys. Technol. Semicond. Devices Integr. Circuits, 1992. 1523. 608–612. doi:10.1117/12.634082. Bibcode: 1992SPIE.1523..608V.
- ↑ F. Licetus, Litheosphorus, sive de lapide Bononiensi lucem in se conceptam ab ambiente claro mox in tenebris mire conservante, Utini, ex typ. N. Schiratti, 1640. See http://www.chem.leeds.ac.uk/delights/texts/Demonstration_21.htm
- ↑ Kresse, Robert; Baudis, Ulrich; Jäger, Paul; Riechers, H. Hermann; Wagner, Heinz; Winkler, Jochen; Wolf, Hans Uwe (2007). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_325.pub2.
- ↑ Harvey E. Newton (1957). A History of Luminescence: From the Earliest Times until 1900. Memoirs of the American Physical Society, Philadelphia, J. H. FURST Company, Baltimore, Maryland (USA), Vol. 44, Chapter 1, pp. 11-43.
- ↑ Smet, Philippe F.; Moreels, Iwan; Hens, Zeger; Poelman, Dirk (2010). "Luminescence in Sulfides: A Rich History and a Bright Future". Materials 3 (4): 2834–2883. doi:10.3390/ma3042834. ISSN 1996-1944. Bibcode: 2010Mate....3.2834S.
- ↑ Hardev Singh Virk (2014). "History of Luminescence from Ancient to Modern Times". ResearchGate. https://www.researchgate.net/publication/259713568.
- ↑ "Lapis Boloniensis". www.zeno.org. http://www.zeno.org/Lemery-1721/A/Lapis+Boloniensis.
- ↑ Lemery, Nicolas (1714). Trait℗e universel des drogues simples. https://books.google.com/books?id=K25AAAAAcAAJ&pg=PA458.
- ↑ Ozanam, Jacques; Montucla, Jean Etienne; Hutton, Charles (1814). Recreations in mathematics and natural philosophy ... https://books.google.com/books?id=jAYAAAAAQAAJ&pg=PA411.
- ↑ P. Ehrlich (1963). "Alkaline Earth Metals". in G. Brauer. Handbook of Preparative Inorganic Chemistry, 2nd Ed.. 2. NY, NY: Academic Press. pp. 937.
 |

