Chemistry:Niobium triselenide
| |
| Names | |
|---|---|
| IUPAC name
Niobium triselenide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| NbSe3 | |
| Molar mass | 1292.425 g/mol |
| Appearance | black powder, silvery crystals |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Niobium triselenide is an inorganic compound belonging to the class of transition metal trichalcogenides. It has the formula NbSe3. It was the first reported example of one-dimensional compound to exhibit the phenomenon of sliding charge density waves.[1] Due to its many studies and exhibited phenomena in quantum mechanics, niobium triselenide has become the model system for quasi-1-D charge density waves.
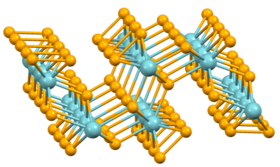
Structure
Niobium triselenide has a highly anisotropic structure. The Nb4+ centers are bound within trigonal prisms defined by six Se ligands. Two pairs of these six Se atoms are bonded to each other to make the polyselenide Se2−
2; the other two exist as the monatomic Se2−.[2] The NbSe6 prisms form infinite co-parallel chains. Although the prisms share the same coordination, the cell consists of three chain types repeated twice, where each chain is defined by its Se–Se bond length. The Se–Se bond lengths are 2.37, 2.48, and 2.91 angstroms.[3][4]
Synthesis
The compound is prepared by the solid state reaction by heating niobium and selenium at 600 to 700 °C:
- Nb + 3 Se → NbSe3
The resulting black crystals can contain NbSe2 impurities. Samples can be purified by chemical vapor transport (CVT) between 650 and 700 °C. The lower limit of CVT was determined by the temperature at which NbSe2 is no longer stable.[5]
Properties
Measurements on NbSe3 provided significant evidence for charge density wave (CDW) transport, CDW pinning, magnetism, Shubnikov-de Hass oscillations, and the Aharonov–Bohm effect.
The electrical resistivity of most metallic compounds decrease as temperature decreases. For the most part NbSe3 follows this trend except two anomalies exist where electrical resistivity reaches two local maxima at 145 K (−128 °C) and 59 K (−214 °C). The maxima result in a sharp decrease in electrical conductivity. This observation is explained by the charge density wave formations that open the gaps in the Fermi surface. The opening causes the 1-D linear system to behave more like a semiconductor and less like a metal, a transition commonly known as Peierls transition. NbSe3 continues to be metallic despite the Peierls transition because the charge density wave formation does not completely remove the Fermi surface, a phenomenon known as imperfect Fermi surface nesting.[6]
In the form of nanofibers, NbSe3 exhibits superconductivity below 2 K (−271 °C).
Niobium triselenide has been considered as a cathode material for rechargeable lithium batteries due to its fibrous structure, high electrical conductivity, and high gravimetric and volumetric energy densities at room temperature.[7]
References
- ↑ Monçeau, P.; Ong, N.; Portis, A.; Meerschaut, A.; Rouxel, J. (1976). "Electric Field Breakdown of Charge-Density-Wave—Induced Anomalies in NbSe3". Physical Review Letters 37 (10): 602. doi:10.1103/PhysRevLett.37.602. Bibcode: 1976PhRvL..37..602M.
- ↑ Hoffmann, Roald; Shaik, Sason; Scott, J.C.; Whangbo, Myung-Hwan; Foshee, Mary J. (September 1980). "The electronic structure of NbSe3". Journal of Solid State Chemistry 34 (2): 263–269. doi:10.1016/0022-4596(80)90230-3. Bibcode: 1980JSSCh..34..263H. http://www.roaldhoffmann.com/sites/all/files/157.pdf. Retrieved 2022-06-20.
- ↑ Gor'kov, L. P.; Grüner, G., eds (1985). "Transition Metal Trichalcogenides". Modern Problems in Condensed Matter Science. 25. North-Holland: Elsevier Science Publishers B.V.. ISBN 0-444-87370-8. https://books.google.com/books?id=3cu2QIeL0J4C&q=Niobium+Triselenide+oxidation+state&pg=PA29.
- ↑ Hodeau, J.L.; Marezio, M.; Roucau, C.; Ayroles, R.; Meerschaut, A.; Rouxel, J.; Monceau, P. (1978). "Charge Density Waves in NbSe3 at 145 K: Crystal structures x-ray and Electron Diffraction Studies". Journal of Physics C 11 (20): 4117–4134. doi:10.1088/0022-3719/11/20/009. Bibcode: 1978JPhC...11.4117H.
- ↑ Tang, H.; Li, C.; Yang, X.; Mo, C.; Cao, K.; Yan, F. (2011). "Synthesis and tribological properties of NbSe3 nanofibers and NbSe2 microsheets". Crystal Research and Technology 46 (4): 400. doi:10.1002/crat.201100030.
- ↑ Hodeau, J. L.; Marezio, M.; Roucau, C.; Ayroles, R.; Meerschaut, A.; Rouxel, J.; Monceau, P. (1978). "Charge-density waves in NbSe3 at 145K: Crystal structures, X-ray and electron diffraction studies". Journal of Physics C: Solid State Physics 11 (20): 4117. doi:10.1088/0022-3719/11/20/009. Bibcode: 1978JPhC...11.4117H.
- ↑ Ratnakumar, B. V.; Stefano, S.; Bankston, C. P. (1989). "A.c. Impedance of niobium triselenide cathode in secondary lithium cells". Journal of Applied Electrochemistry 19 (6): 813. doi:10.1007/BF01007927.
 |
