Chemistry:Orthocarbonic acid
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Methanetetrol[1] | |||
| Systematic IUPAC name
Orthocarbonic acid | |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| C(OH) 4 | |||
| Molar mass | 80.039 g·mol−1 | ||
| Related compounds | |||
Other cations
|
|||
Related compounds
|
| ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
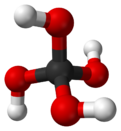
Orthocarbonic acid (also known as methanetetrol) is a chemical compound with the chemical formula H
4CO
4 or C(OH)
4. Its molecular structure consists of a single carbon atom bonded to four hydroxyl groups. It would be therefore a fourfold alcohol. In theory, it could lose four protons to give the hypothetical oxocarbon anion orthocarbonate CO4−
4, and is therefore considered an oxoacid of carbon.
Orthocarbonic acid is highly unstable and long held to be a hypothetical chemical compound. Calculations show that it decomposes into carbonic acid and water:[2][3]
- H
4CO
4 → H
2CO
3 + H
2O
However, orthocarbonic acid was first synthesized in 2025 from the electron-irradiation of a frozen mixture of water and carbon dioxide and identified by mass spectrometry.[4]
Researchers predict that orthocarbonic acid is stable at high pressure; thus, it may form in the interior of the ice giant planets Uranus and Neptune, where water and methane are common.[5]
Orthocarbonate anions
By loss of one through four protons, orthocarbonic acid could yield four anions: H
3CO−
4 (trihydrogen orthocarbonate), H
2CO2−
4 (dihydrogen orthocarbonate), HCO3−
4 (hydrogen orthocarbonate), and CO4−
4 (orthocarbonate).
Numerous salts of fully deprotonated CO4−
4, such as Ca
2CO
4 (calcium orthocarbonate) or Sr
2CO
4 (strontium orthocarbonate), have been synthesized under high pressure conditions and structurally characterized by X-ray diffraction.[6][7][8][9] Strontium orthocarbonate, Sr
2CO
4, is stable at atmospheric pressure. Orthocarbonate is tetrahedral in shape, and is isoelectronic to orthonitrate. The C-O distance is 1.41 Å.[10] Sr
3(CO
4)O is an oxide orthocarbonate (tristrontium orthocarbonate oxide), also stable at atmospheric pressure.[11]
Orthocarbonate esters
The tetravalent moiety CO
4 is found in stable organic compounds; they are formally esters of orthocarbonic acid, and therefore are called orthocarbonates. For example, tetraethoxymethane can be prepared by the reaction between chloropicrin and sodium ethoxide in ethanol.[12] Polyorthocarbonates are stable polymers that might have applications in absorbing organic solvents in waste treatment processes,[13] or in dental restorative materials.[14] The explosive trinitroethylorthocarbonate possesses an orthocarbonate core.
A linear polymer which can be described as a (spiro) orthocarbonate ester of pentaerythritol, whose formula could be written as [(–CH
2)
2C(CH
2–)
2 (–O)
2C(O–)
2]
n, was synthesized in 2002.[15]
The carbon atom in the spiro ester bis-catechol orthocarbonate was found to have tetrahedral bond geometry, contrasting with the square planar geometry of the silicon atom in the analogous orthosilicate ester.[16]
Orthocarbonates may exist in several conformers, that differ by the relative rotation of the C–O–C bridges. The conformation structures of some esters, such as tetraphenoxymethane, tetrakis(3,5-dimethyl-phenoxy)methane, and tetrakis(4-bromophenoxy)methane have been determined by X-ray diffraction.[17]
See also
- Pentaerythritol, C(CH
2OH)
4 - Silicic acid, Si(OH)
4 - Carbonic acid, H
2CO
3
References
- ↑ "Methanetetrol - PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information. https://pubchem.ncbi.nlm.nih.gov/compound/9547954.
- ↑ Bohm S.; Antipova D.; Kuthan J. (1997). "A Study of Methanetetraol Dehydration to Carbonic Acid". International Journal of Quantum Chemistry 62 (3): 315–322. doi:10.1002/(SICI)1097-461X(1997)62:3<315::AID-QUA10>3.0.CO;2-8.
- ↑ Carboxylic Acids and Derivatives "Carboxylic Acids and Derivatives". Glossary of Class Names of Organic Compounds and Reactive Intermediaries Based on Structure (IUPAC Recommendations 1994). 1994. http://www.chem.qmul.ac.uk/iupac/class/carba.html#15.
- ↑ Marks, Joshua H.; Bai, Xilin; Nikolayev, Anatoliy A.; Gong, Qi'ang; McAnally, Mason; Wang, Jia; Pan, Yang; Fortenberry, Ryan C. et al. (14 July 2025). "Methanetetrol and the final frontier in ortho acids". Nature Communications. doi:10.1038/s41467-025-61561-z.
- ↑ G. Saleh; A. R. Oganov (2016). "Novel Stable Compounds in the C-H-O Ternary System at High Pressure". Scientific Reports 6. doi:10.1038/srep32486. PMID 27580525. Bibcode: 2016NatSR...632486S.
- ↑ Sagatova, Dinara; Shatskiy, Anton; Sagatov, Nursultan; Gavryushkin, Pavel N.; Litasov, Konstantin D. (2020). "Calcium orthocarbonate, Ca2CO4-Pnma: A potential host for subducting carbon in the transition zone and lower mantle". Lithos 370-371. doi:10.1016/j.lithos.2020.105637. ISSN 0024-4937. Bibcode: 2020Litho.37005637S.
- ↑ Binck, Jannes; Laniel, Dominique; Bayarjargal, Lkhamsuren; Khandarkhaeva, Saiana; Fedotenko, Timofey; Aslandukov, Andrey; Milman, Victor; Glazyrin, Konstantin et al. (2022). "Synthesis of calcium orthocarbonate, Ca2CO4-Pnma at P-T conditions of Earth's transition zone and lower mantle". American Mineralogist 107 (3): 336–342. doi:10.2138/am-2021-7872. Bibcode: 2022AmMin.107..336B. https://bib-pubdb1.desy.de/search?p=id:%22PUBDB-2022-02872%22.
- ↑ Laniel, Dominique; Binck, Jannes; Winkler, Björn; Vogel, Sebastian; Fedotenko, Timofey; Chariton, Stella; Prakapenka, Vitali; Milman, Victor et al. (2021). "Synthesis, crystal structure and structure–property relations of strontium orthocarbonate, Sr2CO4". Acta Crystallographica Section B 77 (1): 131–137. doi:10.1107/S2052520620016650. ISSN 2052-5206. Bibcode: 2021AcCrB..77..131L.
- ↑ Gavryushkin, Pavel N.; Sagatova, Dinara N.; Sagatov, Nursultan; Litasov, Konstantin D. (2021). "Formation of Mg-Orthocarbonate through the Reaction MgCO3 + MgO = Mg2CO4 at Earth's Lower Mantle P–T Conditions". Crystal Growth & Design 21 (5): 2986–2992. doi:10.1021/acs.cgd.1c00140.
- ↑ Spahr, Dominik; Binck, Jannes; Bayarjargal, Lkhamsuren; Luchitskaia, Rita; Morgenroth, Wolfgang; Comboni, Davide; Milman, Victor; Winkler, Björn (4 April 2021). "Tetrahedrally Coordinated sp3-Hybridized Carbon in Sr2CO4 Orthocarbonate at Ambient Conditions". Inorganic Chemistry 60 (8): 5419–5422. doi:10.1021/acs.inorgchem.1c00159. PMID 33813824.
- ↑ Spahr, Dominik; König, Jannes; Bayarjargal, Lkhamsuren; Gavryushkin, Pavel N.; Milman, Victor; Liermann, Hanns-Peter; Winkler, Björn (4 October 2021). "Sr 3 [CO 4 ]O Antiperovskite with Tetrahedrally Coordinated sp 3 -Hybridized Carbon and OSr 6 Octahedra". Inorganic Chemistry 60 (19): 14504–14508. doi:10.1021/acs.inorgchem.1c01900. PMID 34520201.
- ↑ Orthocarbonic acid, tetraethyl ester Organic Syntheses, Coll. Vol. 4, p. 457 (1963); Vol. 32, p. 68 (1952).
- ↑ Sonmez, H.B.; Wudl, F. (2005). "Cross-linked poly(orthocarbonate)s as organic solvent sorbents". Macromolecules 38 (5): 1623–1626. doi:10.1021/ma048731x. Bibcode: 2005MaMol..38.1623S.
- ↑ Stansbury, J.W. (1992). "Synthesis and evaluation of new oxaspiro monomers for double ring-opening polymerization". Journal of Dental Research 71 (7): 1408–1412. doi:10.1177/00220345920710070901. PMID 1629456. http://jdr.iadrjournals.org/cgi/content/abstract/71/7/1408. Retrieved 2008-06-19.
- ↑ David T. Vodak, Matthew Braun, Lykourgos Iordanidis, Jacques Plévert, Michael Stevens, Larry Beck, John C. H. Spence, Michael O'Keeffe, Omar M. Yaghi (2002): "One-Step Synthesis and Structure of an Oligo(spiro-orthocarbonate)". Journal of the American Chemical Society, volume 124, issue 18, pages 4942–4943. doi:10.1021/ja017683i
- ↑ H. Meyer, G. Nagorsen (1979): "Structure and reactivity of the orthocarbonic and orthosilicic acid esters of pyrocatechol". Angewandte Chemie International Edition in English, volume 18, issue 7, pages 551-553. doi:10.1002/anie.197905511
- ↑ N. Narasimhamurthy, H. Manohar, Ashoka G. Samuelson, Jayaraman Chandrasekhar (1990): "Cumulative anomeric effect: A theoretical and x-ray diffraction study of orthocarbonates". Journal of the American Chemical Society, volume 112, issue 8, pages 2937–2941. doi:10.1021/ja00164a015
 |