Chemistry:Paraffin wax

| |
| Identifiers | |
|---|---|
| UNII | |
| Properties | |
| CnH2n+2 | |
| Appearance | White solid[1] |
| Odor | Odorless |
| Boiling point | > 370 °C (698 °F) |
| ~1 mg/L[1] | |
| Hazards | |
| Flash point | 200–240 °C (392–464 °F; 473–513 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |

Paraffin wax (or petroleum wax) is a soft colorless solid derived from petroleum, coal, or oil shale that consists of a mixture of hydrocarbon molecules containing between 20 and 40 carbon atoms. It is solid at room temperature and begins to melt above approximately 37 °C (99 °F),[2] and its boiling point is above 370 °C (698 °F).[2] Common applications for paraffin wax include lubrication, electrical insulation, and candles;[3] dyed paraffin wax can be made into crayons. It is distinct from kerosene and other petroleum products that are sometimes called paraffin.[4]
Un-dyed, unscented paraffin candles are odorless and bluish-white. Paraffin wax was first created by Carl Reichenbach in Germany in 1830 and marked a major advancement in candlemaking technology, as it burned more cleanly and reliably than tallow candles and was cheaper to produce.[5]
In chemistry, paraffin is used synonymously with alkane, indicating hydrocarbons with the general formula CnH2n+2. The name is derived from Latin parum ("very little") + affinis, meaning "lacking affinity" or "lacking reactivity", referring to paraffin's unreactive nature.[6]
Properties
Paraffin wax is mostly found as a white, odorless, tasteless, waxy solid, with a typical melting point between about 46 and 68 °C (115 and 154 °F),[7] and a density of around 900 kg/m3.[8] It is insoluble in water, but soluble in ether, benzene, and certain esters. Paraffin is unaffected by most common chemical reagents but burns readily.[9] Its heat of combustion is 42 MJ/kg.[10]
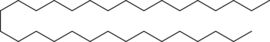
The hydrocarbon C31H64 is a typical component of paraffin wax.
Paraffin wax is an excellent electrical insulator, with a resistivity of between 1013 and 1017 ohm-metre.[11] This is better than nearly all other materials except some plastics (notably PTFE). It is an effective neutron moderator and was used in James Chadwick's 1932 experiments to identify the neutron.[12][13]
Paraffin wax is an excellent material for storing heat, with a specific heat capacity of 2.14–2.9 J⋅g−1⋅K−1 (joules per gram per kelvin) and a heat of fusion of 200–220 J⋅g−1.[14] Paraffin wax phase-change cooling coupled with retractable radiators was used to cool the electronics of the Lunar Roving Vehicle during the crewed missions to the Moon in the early 1970s.[15] Wax expands considerably when it melts and so is used in wax element thermostats for industrial, domestic and, particularly, automobile purposes.[16][17]
If pure paraffin wax melted to the approximate flash point in a half open glass vessel which is then suddenly cooled down, then its vapors may autoignite as result of reaching boiling liquid pressure.[18]
History
Paraffin wax was first created in 1830 by German chemist Karl von Reichenbach when he attempted to develop a method to efficiently separate and refine waxy substances naturally occurring in petroleum. Paraffin represented a major advance in the candle-making industry because it burned cleanly and was cheaper to manufacture than other candle fuels. Paraffin wax initially suffered from a low melting point. This was remedied by adding stearic acid. The production of paraffin wax enjoyed a boom in the early 20th century due to the growth of the oil and meatpacking industries, which created paraffin and stearic acid as byproducts.[5]
Manufacturing
The feedstock for paraffin is slack wax, which is a mixture of oil and wax, a byproduct from the refining of lubricating oil.
The first step in making paraffin wax is to remove the oil (de-oiling or de-waxing) from the slack wax. The oil is separated by crystallization. Most commonly, the slack wax is heated, mixed with one or more solvents such as a ketone and then cooled. As it cools, wax crystallizes out of the solution, leaving only oil. This mixture is filtered into two streams: solid (wax plus some solvent) and liquid (oil and solvent). After the solvent is recovered by distillation, the resulting products are called "product wax" (or "press wax") and "foots oil". The lower the percentage of oil in the wax, the more refined it is considered (semi-refined versus fully refined).[19] The product wax may be further processed to remove colors and odors. The wax may finally be blended together to give certain desired properties such as melt point and penetration. Paraffin wax is sold in either liquid or solid form.[20][21][22]
Applications
In industrial applications, it is often useful to modify the crystal properties of the paraffin wax, typically by adding branching to the existing carbon backbone chain. The modification is usually done with additives, such as EVA copolymers, microcrystalline wax, or forms of polyethylene. The branched properties result in a modified paraffin with a higher viscosity, smaller crystalline structure, and modified functional properties. Pure paraffin wax is rarely used for carving original models for casting metal and other materials in the lost wax process, as it is relatively brittle at room temperature and presents the risks of chipping and breakage when worked. Soft and pliable waxes, like beeswax, may be preferred for such sculpture, but "investment casting waxes," often paraffin-based, are expressly formulated for the purpose.
In a histology or pathology laboratory, paraffin wax is used to impregnate tissue prior to sectioning thin samples. Water is removed from the tissue through ascending strengths of alcohol (75% to absolute), and then that is cleared in an organic solvent such as xylene. The tissue is then placed in paraffin wax for several hours, then set in a mold with wax to cool and solidify. Sections are then cut on a microtome.
Other uses
- Candle-making
- Wax carving
- Bicycle chain lubrication
- Coatings for waxed paper or waxed cotton
- Food-grade paraffin wax:
- Shiny coating used in candy-making; although edible, it is nondigestible, passing through the body without being broken down
- Coating for many kinds of hard cheese, like Edam cheese
- Sealant for jars, cans, and bottles
- Chewing gum additive
- Investment casting
- Anti-caking agent, moisture repellent, and dustbinding coatings for fertilizers
- Agent for preparation of specimens for histology
- Bullet lubricant – with other ingredients, such as olive oil and beeswax
- Phlegmatizing agent, commonly used to stabilise/desensitize high explosives such as RDX
- Crayons
- Solid propellant for hybrid rocket motors[23][24]
- Component of surfboard wax, ski wax, and skateboard wax
- Ink. Used as the basis for solid ink different color blocks of wax for thermal printers. The wax is melted and then sprayed on the paper producing images with a shiny surface
- Microwax:[25] food additive, a glazing agent with E number E905
- Forensic investigations: the nitrate test uses paraffin wax to detect nitrates and nitrites on the hand of a shooting suspect
- Antiozonant agents: blends of paraffin and micro waxes are used in rubber compounds to prevent cracking of the rubber; the admixture of wax migrates to the surface of the product and forms a protective layer. The layer can also act as a release agent, helping the product separate from its mould.[26]
- Mechanical thermostats and actuators, as an expansion medium for activating such devices[17][27]
- As a potting material to encapsulate electronic components such as guitar pickups, transformers, and inductors, to prevent moisture ingress and to reduce electromagnetically-induced acoustic noise and microphonic effects
- Textile manufacturing processes, such as that used for Eisengarn thread.
- Thickening agent in many paintballs
- Moisturiser in toiletries and cosmetics such as Vaseline.
- Prevents oxidation on the surface of polished steel and iron[28]
- Phase change material for thermal energy storage
- Manufacture of boiled leather armor and books
- Neutron radiation shielding
- Wax baths for occupational and physical therapies and cosmetic treatments
- Paraffin is effective in the treatment of Osteoarthritis of the hand joints. Treatment consists of dip-wrapped paraffin bath therapy for 15 minutes until paraffin cooled for five days a weeks. The use of paraffin wax bath has been shown to decrease pain at rest and during ADLs (activities of daily living) compared to groups who did not receive wax therapy.[30]
- Improvements in grip strength and pinch strength have been found in patients with Carpel Tunnel Syndrome, Osteoarthritis, spasticity, and post-traumatic stiffness for those who have used paraffin bath therapy along with traditional physical therapy in their recovery. It has been found that patients who have used paraffin bath therapy have yielded lower VAS and AUSCAN scores (pain scores) compared to those who did not.[31]
- Used for wood finishing
- Used as a fuel for fire breathing
- Used in Lava Lamps
Occupational safety
People can be exposed to paraffin in the workplace by breathing it in, skin contact, and eye contact. The National Institute for Occupational Safety and Health (NIOSH) has set a recommended exposure limit (REL) for paraffin wax fume exposure of 2 mg/m3 over an 8-hour workday.[32]
See also
References
- ↑ 1.0 1.1 1.2 Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ↑ 2.0 2.1 Freund, Mihály; Mózes, Gyula (1982). Paraffin products: properties, technologies, applications. Amsterdam, the Netherlands: Elsevier. p. 121. ISBN 978-0-444-99712-8.
- ↑ Raw materials and candles production processes , AECM
- ↑ "Research Paper RP1670 | Vapor Pressures and Boiling Points of Some Paraffin, Alkylcycopentane, Alkylcyclohexane and Alkylbenzene Hydrocarbons". US Department of Commerce National Bureau of Standards. https://nvlpubs.nist.gov/nistpubs/jres/35/jresv35n3p219_A1b.pdf.
- ↑ 5.0 5.1 "History of Candles". http://candles.org/history/.
- ↑ "Paraffin, n". Oxford English Dictionary. Oxford, England: Oxford University Press. March 2009.
- ↑ Nasser, William E (1999). "Waxes, Natural and Synthetic". in McKetta, John J. Encyclopedia of Chemical Processing and Design. 67. New York: Marcel Dekker. p. 17. ISBN 978-0-8247-2618-8. This can vary widely, even outside the quoted range, according to such factors as oil content and crystalline structure.
- ↑ Kaye, George William Clarkson; Laby,Thomas Howell. "Mechanical properties of materials". Kaye and Laby Tables of Physical and Chemical Constants. National Physical Laboratory. http://www.kayelaby.npl.co.uk/general_physics/2_2/2_2_1.html.
- ↑ Seager, Spencer L.; Slabaugh, Michael (19 January 2010). "Alkane reactions". Chemistry for Today: General, Organic, and Biochemistry. Belmont, California: Cengage. p. 364. ISBN 978-0-538-73332-8.
- ↑ Wiener, Harry (January 1947). "Structural Determination of Paraffin Boiling Points". Journal of the American Chemical Society 69 (1): 17–20. doi:10.1021/ja01193a005. ISSN 0002-7863. PMID 20291038. https://pubs.acs.org/doi/pdf/10.1021/ja01193a005.
- ↑ "Electrical insulating materials". Kaye and Laby Tables of Physical and Chemical Constants. National Physical Laboratory. 1995. http://www.kayelaby.npl.co.uk/general_physics/2_6/2_6_3.html.
- ↑ "Attenuation of fast neutrons: neutron moderation and diffusion". Kaye and Laby Tables of Physical and Chemical Constants. National Physical Laboratory. http://www.kayelaby.npl.co.uk/atomic_and_nuclear_physics/4_7/4_7_3.html.
- ↑ Rhodes, Richard (1981). The Making of the Atomic Bomb. New York: Simon and Schuster. p. 163. ISBN 978-0-671-44133-3. https://archive.org/details/makingofatomicbo00rhod.
- ↑ "Specific Heat Capacity". Diracdelta.co.uk Science and Engineering Encyclopedia. Dirac Delta Consultants Ltd, Warwick, England. http://www.diracdelta.co.uk/science/source/s/p/specific%20heat%20capacity/source.html. Retrieved 25 October 2013.
- ↑ Dean, W. G.; Karu, Z. S. (February 1993). "Space Station thermal storage/refrigeration system research and development". Final Report Lockheed Missiles and Space Co.. Bibcode: 1993lock.rept.....D.
- ↑ Wax-pellet thermostat United States Patent 4948043
- ↑ 17.0 17.1 Bodén, Roger. "Paraffin Microactuator". Materials Science Sensors and Actuators. University of Uppsala. http://hermes.material.uu.se/~klas/Paraffin_lab_eng.pdf.
- ↑ Husting, Chad (2019-08-18). "Paraffin Wax – an Exothermic Reaction: Caution" (in en). https://www.chemedx.org/blog/paraffin-wax-exothermic-reaction-caution.
- ↑ "Paraffin Wax (Fully Refined)". Barasat Wax Refiner. http://www.barasatwax.com/fullyrefined.php.
- ↑ "Wax Refining". The International Group, Inc.. http://igiwax.com/reference/wax-refining.html.
- ↑ "Paraffin wax". Bitumen Engineering. http://bitumenengineering.com/materials/paraffin-wax.
- ↑ "Manufacturing Process". Barasat Wax Refiner. http://www.barasatwax.com/mp.php.
- ↑ Staff (Fall 2004). "Rocket motor uses common household product for fuel". OASIS Ocean Air Space Industry Site 1 (3): 6. http://www.nasa.gov/centers/stennis/pdf/69281main_fall.pdf. Retrieved 28 November 2008.
- ↑ Tabor, Abigail (18 April 2017). "From Pedicures to the Peregrine Rocket, Paraffin Wax Proves Its Worth". https://www.nasa.gov/feature/ames/from-pedicures-to-the-peregrine-rocket-paraffin-wax-proves-its-worth.
- ↑ "Paraffin, microcrystalline, petrolatum, wax blends - Microcrystalline Wax". igiwax.com. http://igiwax.com/igi-products/by-type/microcrystallinewax.html.
- ↑ (Freund Mózes)
- ↑ Ogden, Sam; Klintberg, Lena; Thornell, Greger; Hjort, Klas; Bodén, Roger (30 November 2013). "Review on miniaturized paraffin phase change actuators, valves, and pumps". Microfluidics and Nanofluidics 17: 53–71. doi:10.1007/s10404-013-1289-3. http://urn.kb.se/resolve?urn=urn:nbn:se:uu:diva-208904.
- ↑ Dick, William B (1872). "Encyclopedia Of Practical Receipts And Processes". New York: Dick and Fitzgerald. http://chestofbooks.com/reference/Encyclopedia-Of-Practical-Receipts-And-Processes/Steel-Part-6.html.
- ↑ "Instrument Information". NASA. 2007. https://pds.nasa.gov/ds-view/pds/viewInstrumentProfile.jsp?INSTRUMENT_ID=MDIS-NAC&INSTRUMENT_HOST_ID=MESS.
- ↑ Beasley, Jeanine; Ward, LeeAnn; Knipper-Fisher, Katie; Hughes, Katia; Lunsford, Dianna; Leiras, Claudia (April 2019). "Conservative therapeutic interventions for osteoarthritic finger joints: A systematic review" (in en). Journal of Hand Therapy 32 (2): 153–164.e2. doi:10.1016/j.jht.2018.01.001. PMID 30017415. https://linkinghub.elsevier.com/retrieve/pii/S0894113017302740.
- ↑ Kim, Sang-Gyun; Kang, Jong Woo; Boo, Joon Hyeok; Jin, Dong Uk; Choi, Sung Jae; Song, Gwan Gyu; Jung, Jae Hyun (March 2023). "Effectiveness of paraffin bath therapy for the symptoms and function of hand diseases: A systematic review and meta-analysis of randomized controlled trials" (in en). Journal of Hand Therapy: S0894113022001016. doi:10.1016/j.jht.2022.10.005. PMID 36914488. https://linkinghub.elsevier.com/retrieve/pii/S0894113022001016.
- ↑ "CDC – NIOSH Pocket Guide to Chemical Hazards – Paraffin wax fume". https://www.cdc.gov/niosh/npg/npgd0477.html.
External links
- "Odd Uses of Paraffin", Scientific American, 13 July 1878, p. 19
 |
