Chemistry:Uranium pentabromide
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| UBr 5 | |
| Molar mass | 637.549 g/mol |
| Appearance | dark brown, hygroscopic crystalline solid |
| decomposes | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Uranium pentabromide is an inorganic chemical compound with the formula U
2Br
10.
Synthesis
The compound is made by reacting the elements in an acetonitrile solvent, or by reacting bromine with uranium metal or uranium tetrabromide at 55 °C (131 °F; 328 K).[1]
Properties
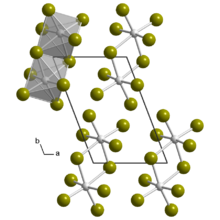
Uranium pentabromide is a hygroscopic dark brown solid that decomposes in water and most organic solvents, the exceptions being acetonitrile or dichloromethane.[1] The compound is rather unstable and difficult to purify,[2] decomposing at 80 °C (176 °F; 353 K) into uranium tetrabromide and bromine.[3] The crystal structure is the same as that of β-UCl
5, which is triclinic and consists of U
2Br
10 dimers.[4]
Complexes
Stable complexes of the form UBr
5L are known with such ligands as triphenylphosphine oxide and hexamethylphosphoramide, and are obtained by brominating UBr
4 in the presence of the desired ligand.[2] In addition, it is possible to obtain a hexabromouranate(V) salt by reacting UBr
5 with a monovalent bromide in thionyl bromide:[1]
- U
2Br
10 + 2MBr → 2M+
[UBr
6]−
References
- ↑ 1.0 1.1 1.2 Morss, L. R.; Edelstein, Norman M.; Fuger, Jean (2010-10-21) (in en). The Chemistry of the Actinide and Transactinide Elements (Set Vol.1-6): Volumes 1-6. Springer Science & Business Media. p. 526. ISBN 978-94-007-0211-0. https://books.google.com/books?id=9vPuV3A0UGUC&pg=PA526. Retrieved 29 May 2021.
- ↑ 2.0 2.1 Brown, D.; Holah, D. G.; Rickard, C. E. F. (1968-01-01). "The stabilisation of plutonium tetrabromide and uranium pentabromide" (in en). Chemical Communications (11): 651–652. doi:10.1039/C19680000651. ISSN 0009-241X. https://pubs.rsc.org/en/content/articlelanding/1968/c1/c19680000651.
- ↑ Blair, A.; Ihle, H. (1973-11-01). "The thermal decomposition and thermodynamic properties of uranium pentabromide" (in en). Journal of Inorganic and Nuclear Chemistry 35 (11): 3795–3803. doi:10.1016/0022-1902(73)80071-5. ISSN 0022-1902. https://www.sciencedirect.com/science/article/abs/pii/0022190273800715. Retrieved 29 May 2021.
- ↑ Levy, J.H.; Taylor, J.C.; Wilson, P.W. (1978-01-01). "The crystal structure of uranium pentabromide by powder neutron diffraction" (in en). Journal of Inorganic and Nuclear Chemistry 40 (6): 1055–1057. doi:10.1016/0022-1902(78)80507-7. ISSN 0022-1902. https://www.sciencedirect.com/science/article/abs/pii/0022190278805077. Retrieved 29 May 2021.
Template:Bromides Template:Actinide halides
 |
