Chemistry:Uranyl fluoride
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
Uranium(VI) difluoride dioxide
| |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| UO 2F 2 | |
| Molar mass | 308.024 g·mol−1 |
| Appearance | yellow solid[1] |
| Density | 6.37 g/cm3[2] |
| very soluble | |
| Hazards | |
| GHS pictograms |   
|
| GHS Signal word | Danger |
| H300, H330, H373, H411 | |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Uranyl fluoride is the inorganic compound with the formula UO
2F
2. It is most notable as a contaminant in the production of uranium tetrafluoride.[3]
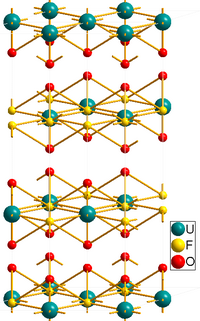
As shown by X-ray crystallography, the uranyl centers UO2+
2 are surrounded by six fluoride ligands F−
.[4]
Synthesis
It is formed in the hydrolysis of uranium hexafluoride (UF
6):
- UF
6 + 2 H
2O → UO
2F
2 + 4 HF
It can also be formed in the hydrofluorination of uranium trioxide (UO
3):
- UO
3 + 2 HF → UO
2F
2 + H
2O[5]
Physical properties
References
- ↑ Perry, Dale L. (2016-04-19) (in en). Handbook of Inorganic Compounds. CRC Press. p. 448. ISBN 978-1-4398-1462-8. https://books.google.com/books?id=SFD30BvPBhoC.
- ↑ https://ibilabs.com/uranium-uranyl-thorium-compounds/uranyl-compounds/uranyl-fluoride/
- ↑ Peehs, Martin; Walter, Thomas; Walter, Sabine; Zemek, Martin (2007). "Uranium, Uranium Alloys, and Uranium Compounds". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a27_281.pub2. ISBN 978-3-527-30385-4.
- ↑ Zachariasen, W. H. (1948). "Crystal chemical studies of the 5f-series of elements. III. A study of the disorder in the crystal structure of anhydrous uranyl fluoride". Acta Crystallographica 1 (6): 277–281. doi:10.1107/S0365110X48000764. Bibcode: 1948AcCry...1..277Z.
- ↑ Jang, Harry; Louis-Jean, James; Poineau, Frederic (2023-06-20). "Synthesis and Morphological Control of UO2F2 Particulates" (in en). ACS Omega 8 (24): 21996–22002. doi:10.1021/acsomega.3c01999. ISSN 2470-1343. PMID 37360455.
 |
