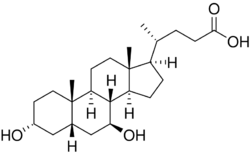
Chemistry:Ursodeoxycholic acid
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Actigall, Urso, others |
| Other names | Ursodiol |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699047 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| Chemical and physical data | |
| Formula | C24H40O4 |
| Molar mass | 392.580 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 203 °C (397 °F) |
| |
| |
| (verify) | |
Ursodeoxycholic acid (UDCA), also known as ursodiol, is a secondary bile acid, produced in humans and most other species from metabolism by intestinal bacteria. It is synthesized in the liver in some species, and was first identified in bile of bears of genus Ursus, from which its name derived.[8] In purified form, it has been used to treat or prevent several diseases of the liver or bile ducts.
It is available as a generic medication.[9][10]
Medical uses
UDCA has been used as medical therapy in gallstone disease (cholelithiasis) and for biliary sludge.[11][12] UDCA helps reduce the cholesterol saturation of bile and leads to gradual dissolution of cholesterol-rich gallstones.[11]
UDCA may be given after bariatric surgery to prevent cholelithiasis, which commonly occurs due to the rapid weight loss producing biliary cholesterol oversaturation and also biliary dyskinesia secondary to hormonal changes.[13]
Primary biliary cholangitis
UDCA is used as therapy in primary biliary cholangitis (PBC; previously known as primary biliary cirrhosis) where it can produce an improvement in biomarkers.[14] Meta-analyses have borne out conflicting results on the mortality benefit.[15] However analyses that exclude trials of short duration (i.e. < 2 years) have demonstrated a survival benefit and are generally considered more clinically relevant.[16] A Cochrane systematic review in 2012 found no significant benefit in reducing mortality, the rate of liver transplantation, pruritus or fatigue.[17] Ursodiol and obeticholic acid are FDA-approved for the treatment of primary biliary cholangitis.[18]
Primary sclerosing cholangitis
UDCA use in primary sclerosing cholangitis is associated with improved serum liver tests that do not always correlate with improved liver disease status.[19] WHO Drug Information advises against its use in primary sclerosing cholangitis in unapproved doses beyond 13–15 mg/kg/day.[20]
UDCA in a dose of 28–30 mg/kg/day increases risk of death and need for liver transplant by 2.3-fold among those with primary sclerosing cholangitis, despite decrease in liver enzymes.[21]
Intrahepatic cholestasis of pregnancy
UDCA has been used for intrahepatic cholestasis of pregnancy. UDCA lessens itching in the mother and may reduce the number of preterm births. Effects on fetal distress and other adverse outcomes are unlikely to be great.[22][23]
Cholestasis
UDCA use is not licensed in children, as its safety and effectiveness have not been established. Evidence is accumulating that ursodeoxycholic acid is ineffective, unsafe and its use is associated with significant risk of morbidity and mortality in neonatal hepatitis and neonatal cholestasis.[24][25][26][27]
Other conditions
UDCA has been suggested to be an adequate treatment of bile reflux gastritis.[28]
Ursodeoxycholic acid has been used to treat cystic fibrosis related cholestasis and liver disease; however, a 2021 study aimed at evaluating whether the incidence of severe liver disease differed between CF centers routinely prescribing or not prescribing UDCA found no reduction in portal hypertension.[29]
UDCA has been in effective in non-alcoholic fatty liver disease, in liver bile duct-paucity syndromes. It has been used as adjunct therapy in conditions with biliary tract obstruction such as biliary atresia; however liver transplant is often still required as definitive treatment for atresia. It is not effective in liver allograft rejection, and in graft-versus-host disease involving the liver.
Adverse effects
Diarrhea was the most frequent adverse event seen in trial of UDCA in gallstone dissolution, occurring in 2 to 9%, which is less frequent than with chenodeoxycholic acid therapy. Bacterial conversion of UDCA to chenodeoxycholic acid may be the mechanism for this side effect. Right upper quadrant abdominal pain and exacerbation of pruritus was occasionally reported in trials in patients with PBC.[30] Additional symptoms may include bloating, weight gain, and occasionally, thinning of hair.[31]
Mechanisms of action
Choleretic effects
Primary bile acids are produced by the liver and stored in the gall bladder. When secreted into the intestine, primary bile acids can be metabolized into secondary bile acids by intestinal bacteria. Primary and secondary bile acids help the body digest fats. Ursodeoxycholic acid helps regulate cholesterol by reducing the rate at which the intestine absorbs cholesterol molecules while breaking up micelles containing cholesterol. The drug reduces cholesterol absorption and is used to dissolve (cholesterol) gallstones in patients who want an alternative to surgery.[32] There are multiple mechanisms involved in cholestatic liver diseases.[33]
Immunomodulating effects
Ursodeoxycholic acid has also been shown experimentally to suppress immune response such as immune cell phagocytosis. Prolonged exposure and/or increased quantities of systemic (throughout the body, not just in the digestive system) ursodeoxycholic acid can be toxic.[34]
Anti-inflammatory effects
Ursodeoxycholic acid has been shown to exert anti-inflammatory and protective effects in human epithelial cells of the gastrointestinal tract. It has been linked to regulation of immunoregulatory responses by regulation of cytokines,[35] antimicrobial peptides defensins,[36] and take an active part in increased restitution of wound in the colon.[37] Moreover, UDCA's effects has been shown to have exert actions outside the epithelial cells.[38]
While some bile acids are known to be colon tumor promoters (e.g. deoxycholic acid),[39][40][41] others such as ursodeoxycholic acid are thought to be chemopreventive, perhaps by inducing cellular differentiation and/or cellular senescence in colon epithelial cells.[42][43][44]
Chemistry
Ursodeoxycholic acid is an epimer of chenodeoxycholic acid, which has similar choleretic effects and a wider species distribution. However, CDCA is not as well-tolerated in humans and it does not show immunomodulating or chemoprotective effects. Both are 7-hydroxyl derivatives of deoxycholic acid, but UDCA has the group in the beta instead of the alpha orientation.[45]
Biosynthesis
Among mammals, only bears (Ursidae; excluding giant pandas) produce UDCA at useful amounts[45] (>30%). It is produced in the bear liver, but the pathway remains unknown.[8]
Other vertebrates produce UDCA in much smaller amounts by gut bacteria. CDCA is oxidized into 7-oxo-CDCA then reduced into UDCA.[46]
Industrial production
UDCA is most commonly produced from cholic acid (CA) derived from bovine bile, a by-product of the beef industry. The current yield of this semisynthesis is about 30%.[47]
Society and culture
Names
The term is from the Latin noun ursus meaning bear, as bear bile contains the substance.[48]
Ursodeoxycholic acid can be chemically synthesized and is marketed under multiple trade names, including Ursetor, Udikast, Actibile, Actigall,[6] Biliver, Deursil, Egyurso, Heptiza 300/150, Stener, Udcasid, Udiliv, Udinorm, Udoxyl, Urso, Urso Forte, Ursocol, Ursoliv, Ursofalk,[49] Ursosan, Ursoserinox, Udimarin, and Ursonova.
History
Bear bile, a natural source of UDCA, has been used in traditional Chinese medicine since the seventh century. Japanese scientists successfully synthesized UDCA chemically in 1955.[45] The earliest reference to UDCA in PubMed dates to 1957 under an alternative spelling "ursodesoxycholic acid", in a small-scale clinical trial.[50]
Ursodeoxycholic acid (application filed by Allergan) was approved for use in the United States in December 1987,[51] and was designated an orphan drug.[52]
References
- ↑ "Ursodiol, Heptiza 300/150 Use During Pregnancy". 4 November 2019. https://www.drugs.com/pregnancy/ursodiol.html.
- ↑ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". 21 June 2022. https://www.tga.gov.au/resources/publication/publications/prescription-medicines-registration-new-generic-medicines-and-biosimilar-medicines-2017.
- ↑ "Product information". 3 August 2000. https://health-products.canada.ca/dpd-bdpp/info?lang=eng&code=11967.
- ↑ "Details for: Ursodiol C". 25 October 2021. https://dhpp.hpfb-dgpsa.ca/dhpp/resource/100493.
- ↑ "Ursodeoxycholic acid 300mg Tablets - Summary of Product Characteristics (SmPC)". 10 July 2019. https://www.medicines.org.uk/emc/product/10064/smpc.
- ↑ 6.0 6.1 "Actigall (ursodiol, USP) capsules". https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=ad414461-262f-43fe-9a3d-bab300b719ed.
- ↑ "Urso 250- ursodiol tablet, film coated Urso Forte- ursodiol tablet, film coated". https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e8fc4fc2-fe5c-4cba-b6e0-5ceaf2157a61.
- ↑ 8.0 8.1 "Ursodeoxycholic acid in the Ursidae: biliary bile acids of bears, pandas, and related carnivores". Journal of Lipid Research 34 (11): 1911–1917. November 1993. doi:10.1016/S0022-2275(20)35109-9. PMID 8263415.
- ↑ "2020 First Generic Drug Approvals". 23 February 2021. https://www.fda.gov/drugs/first-generic-drug-approvals/2020-first-generic-drug-approvals.
- ↑ "Ursodiol: FDA-Approved Drugs". https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=205789.
- ↑ 11.0 11.1 "Medical dissolution of gallstones by oral bile acid therapy". American Journal of Surgery 158 (3): 198–204. September 1989. doi:10.1016/0002-9610(89)90252-3. PMID 2672842.
- ↑ "Gallstone disease: Microlithiasis and sludge". Best Practice & Research. Clinical Gastroenterology 20 (6): 1053–1062. 2006. doi:10.1016/j.bpg.2006.03.007. PMID 17127187.
- ↑ "Ursodeoxycholic Acid in the Prevention of Gallstone Formation After Bariatric Surgery: an Updated Systematic Review and Meta-analysis". Obesity Surgery 27 (11): 3021–3030. November 2017. doi:10.1007/s11695-017-2924-y. PMID 28889240.
- ↑ "A multicenter, controlled trial of ursodiol for the treatment of primary biliary cirrhosis. UDCA-PBC Study Group". The New England Journal of Medicine 324 (22): 1548–1554. May 1991. doi:10.1056/NEJM199105303242204. PMID 1674105.
- ↑ "Randomised controlled trials of ursodeoxycholic-acid therapy for primary biliary cirrhosis: a meta-analysis". Lancet 354 (9184): 1053–1060. September 1999. doi:10.1016/S0140-6736(98)11293-X. PMID 10509495.
- ↑ "Long-term effects of mid-dose ursodeoxycholic acid in primary biliary cirrhosis: a meta-analysis of randomized controlled trials". The American Journal of Gastroenterology 101 (7): 1529–1538. July 2006. doi:10.1111/j.1572-0241.2006.00634.x. PMID 16863557.
- ↑ "Ursodeoxycholic acid for primary biliary cirrhosis". The Cochrane Database of Systematic Reviews 12 (12). December 2012. doi:10.1002/14651858.CD000551.pub3. PMID 23235576.
- ↑ "Primary Biliary Cholangitis: Medical and Specialty Pharmacy Management Update". Journal of Managed Care & Specialty Pharmacy 22 (10-a-s Suppl): S3–S15. October 2016. doi:10.18553/jmcp.2016.22.10-a-s.s3. PMID 27700211.
- ↑ "Bile acids for primary sclerosing cholangitis". The Cochrane Database of Systematic Reviews 2011 (1). January 2011. doi:10.1002/14651858.CD003626.pub2. PMID 21249655.
- ↑ "Ursodeoxycholic acid: serious hepatic events". WHO Drug Information 26 (1). 2012. https://www.who.int/medicines/publications/druginformation/issues/26-1.pdf.
- ↑ "High-dose ursodeoxycholic acid for the treatment of primary sclerosing cholangitis". Hepatology 50 (3): 808–814. September 2009. doi:10.1002/hep.23082. PMID 19585548.
- ↑ "Pharmacological interventions for treating intrahepatic cholestasis of pregnancy". The Cochrane Database of Systematic Reviews 2020 (7). July 2020. doi:10.1002/14651858.CD000493.pub3. PMID 32716060.
- ↑ "Ursodeoxycholic acid versus placebo in women with intrahepatic cholestasis of pregnancy (PITCHES): a randomised controlled trial". Lancet 394 (10201): 849–860. September 2019. doi:10.1016/S0140-6736(19)31270-X. PMID 31378395.
- ↑ "Review of historical cohort: ursodeoxycholic acid in extrahepatic biliary atresia". Journal of Pediatric Surgery 43 (7): 1321–1327. July 2008. doi:10.1016/j.jpedsurg.2007.11.043. PMID 18639689.
- ↑ Paediatric Formulary Committee (2008). British National Formulary for Children 2008. London: Pharmaceutical Press. p. 91. ISBN 978-0-85369-780-0.
- ↑ "Urso package insert.". Birmingham, AL: Axcan Pharma U.S.. January 2000. http://www.axcan.com/pdf/urso_patient_brochure.pdf.
- ↑ "Ursodeoxycholic acid use is associated with significant risk of morbidity and mortality in infants with cholestasis: A strobe compliant study". Medicine 99 (7). February 2020. doi:10.1097/MD.0000000000018730. PMID 32049781.
- ↑ "New Causes for the Old Problem of Bile Reflux Gastritis". Clinical Gastroenterology and Hepatology 16 (9): 1389–1392. September 2018. doi:10.1016/j.cgh.2018.02.034. PMID 29505908.
- ↑ "Ursodeoxycholic acid and liver disease associated with cystic fibrosis: A multicenter cohort study". Journal of Cystic Fibrosis 21 (2): 220–226. March 2022. doi:10.1016/j.jcf.2021.03.014. PMID 33814323.
- ↑ "Systematic review: ursodeoxycholic acid--adverse effects and drug interactions". Alimentary Pharmacology & Therapeutics 18 (10): 963–972. November 2003. doi:10.1046/j.1365-2036.2003.01792.x. PMID 14616161.
- ↑ "Primary biliary cholangitis". Lancet 396 (10266): 1915–1926. December 2020. doi:10.1016/S0140-6736(20)31607-X. PMID 33308474.
- ↑ "Effect of ursodeoxycholic acid and chenodeoxycholic acid on cholesterol and bile acid metabolism". Gastroenterology 91 (4): 1007–1018. October 1986. doi:10.1016/0016-5085(86)90708-0. PMID 3527851.
- ↑ "Ursodeoxycholic acid in cholestatic liver disease: mechanisms of action and therapeutic use revisited". Hepatology 36 (3): 525–531. September 2002. doi:10.1053/jhep.2002.36088. PMID 12198643.
- ↑ "Ursodiol". Material Safety Data Sheet (MSDS). Fisher Scientific. https://fscimage.fishersci.com/msds/70916.htm.
- ↑ "Ursodeoxycholic acid and lithocholic acid exert anti-inflammatory actions in the colon". American Journal of Physiology. Gastrointestinal and Liver Physiology 312 (6): G550–8. June 2017. doi:10.1152/ajpgi.00256.2016. PMID 28360029.
- ↑ "Bile acids deoxycholic acid and ursodeoxycholic acid differentially regulate human β-defensin-1 and -2 secretion by colonic epithelial cells". FASEB Journal 31 (9): 3848–57. September 2017. doi:10.1096/fj.201601365R. PMID 28487283.
- ↑ "The bile acids, deoxycholic acid and ursodeoxycholic acid, regulate colonic epithelial wound healing". American Journal of Physiology. Gastrointestinal and Liver Physiology 314 (3): G378–87. March 2018. doi:10.1152/ajpgi.00435.2016. PMID 29351391.
- ↑ "Ursodeoxycholic acid inhibits TNFα-induced IL-8 release from monocytes". American Journal of Physiology. Gastrointestinal and Liver Physiology 311 (2): G334–41. August 2016. doi:10.1152/ajpgi.00406.2015. PMID 27340129.
- ↑ "The Gut Microbial Bile Acid Modulation and Its Relevance to Digestive Health and Diseases". Gastroenterology 164 (7): 1069–85. June 2023. doi:10.1053/j.gastro.2023.02.022. PMID 36841488.
- ↑ "Bile acids as carcinogens in the colon and at other sites in the gastrointestinal system". Exp Biol Med (Maywood) 248 (1): 79–89. January 2023. doi:10.1177/15353702221131858. PMID 36408538.
- ↑ "The effect of fecal bile acids on the incidence and risk-stratification of colorectal cancer: an updated systematic review and meta-analysis". Sci Rep 15 (1): 740. January 2025. doi:10.1038/s41598-024-84801-6. PMID 39753873. Bibcode: 2025NatSR..15..740Y.
- ↑ "Ursodeoxycholic acid modulates histone acetylation and induces differentiation and senescence". International Journal of Cancer 119 (12): 2958–69. December 2006. doi:10.1002/ijc.22231. PMID 17019713.
- ↑ "Prediagnostic Plasma Bile Acid Levels and Colon Cancer Risk: A Prospective Study". J Natl Cancer Inst 112 (5): 516–524. May 2020. doi:10.1093/jnci/djz166. PMID 31435679.
- ↑ "Phase III trial of ursodeoxycholic acid to prevent colorectal adenoma recurrence". J Natl Cancer Inst 97 (11): 846–53. June 2005. doi:10.1093/jnci/dji144. PMID 15928305.
- ↑ 45.0 45.1 45.2 "Bear bile: dilemma of traditional medicinal use and animal protection". Journal of Ethnobiology and Ethnomedicine 5 (1): 2. January 2009. doi:10.1186/1746-4269-5-2. PMID 19138420.
- ↑ "A biosynthetic pathway for a prominent class of microbiota-derived bile acids". Nature Chemical Biology 11 (9): 685–690. September 2015. doi:10.1038/nchembio.1864. PMID 26192599.
- ↑ "Latest development in the synthesis of ursodeoxycholic acid (UDCA): a critical review". Beilstein Journal of Organic Chemistry 14: 470–483. 2018. doi:10.3762/bjoc.14.33. PMID 29520309.
- ↑ "Ursodiol". PubChem. U.S. National Library of Medicine. https://pubchem.ncbi.nlm.nih.gov/compound/Ursodiol.
- ↑ "Ursofalk" (in cs). Registered LP name. Prague, Czech Republic: State Institute for Drug Control. https://prehledy.sukl.cz/prehled_leciv.html#/detail-reg/0148926.
- ↑ "Ursodesoxycholic acid as in accelerator for riboflavin phosphorylation in children with nutritional dystrophy". The Journal of Vitaminology 3 (3): 165–167. September 1957. doi:10.5925/jnsv1954.3.165. PMID 13476545.
- ↑ "Actigall: FDA-Approved Drugs". https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=019594.
- ↑ "Actigall Orphan Drug Designation and Approval". https://www.accessdata.fda.gov/scripts/opdlisting/oopd/detailedIndex.cfm?cfgridkey=55390.
 |
