Medicine:Engineered CAR T cell delivery
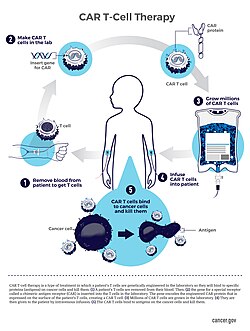
Engineered chimeric antigen receptor (CAR)-T cell delivery is the methodology by which clinicians introduce the cancer-targeting therapeutic system of the CAR-T cell to the human body. CAR-T cells, which utilizes genetic modification of human T-cells to contain antigen binding sequences in addition to the receptor systems CD4 or CD8, are useful in direct targeting and elimination of cancer cells through cytotoxicity.
CAR-T cell delivery involves many varying modalities for implementation, spurring innovative biomedical research to address these modalities. These delivery mechanisms serve to address the limitations of CAR-T cells in translational experimentation and clinical trials, including shelf-life, off-target effects, and tumor infiltration.[1] As of April 2023, six CAR-T cell therapies are clinically approved by the FDA, all of which target hematologic (blood-based) cancers, including multiple myeloma and B-cell leukemias.[2][3] Novel engineered compound-based delivery methods, some of which are in clinical trials, aim to address limitations related to CAR-T cell delivery with the focus to target non-blood based cancers.[4][5]
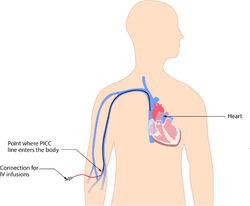
Systemic and intravenous delivery
The classic method of administration of CAR-T cells to cancers within the human body is through intravenous (IV) central line infusion.[6] This infusion allows the CAR-T cells to enter the body’s cardiovascular system, entering the circulation (systemically) amongst developing hematologic cancers. This facilitates the final step in generation and implementation of both autologous and allogeneic CAR-T cell therapy. While this delivery method is reliable for hematologic cancers, as demonstrated by successful clinical trials and FDA regulation, systemic delivery may result in an increase in autoimmune overload, leading to toxic disorders such as cytokine release syndrome (CRS).[1] Discrimination between healthy and malignant cancer cells may additionally result in aplasia, or extremely low or absent amounts of healthy blood cells.[7] Thus, clinically recommended dosage amounts are in place for current CAR-T cell therapies.[8][9][10][11][12][13][14] Current methods exploring ways to improve such complications have been introduced recently by researchers, including “off-switches” to turn off CAR-T cells after initial therapies and further genetic modification to avoid immune rejection.[1][7][15][16] While systemic delivery is important for targeting hematologic cancers, it remains inefficient at targeting solid, or non-circulatory, cancerous tumors. Therefore, regional, or localized targeting strategies utilizing CAR-T cells have arisen in pre-clinical research.[4][5][17][18][19]
Localized delivery mechanisms
Solid tumors, which typically take the form of neoplasms in epithelial cells or in bones, tissue, or adipose (fat), are different than hematologic cancers in that they form a mass of cells, thereby maintaining multiple layers of protection.[20] Because CAR-T cells attack cancerous cells at a surface level, this leaves the CAR-T cells vulnerable to cancerous cell resistance, which renders the CAR-T cell inefficient.[21][22] In recent years, cellular and genetic engineering methods have been explored by researchers to overcome layered protection of solid tumors, in addition to other challenges that have been presented in the advent of CAR-T cell delivery such as in-situ editing and manufacturing, negative immune responses, and biocompatibility of delivery structures.[4][5][17][23][24][25][26] Some of the methodologies used to suspend and deliver CAR-T cells include hydrogel and polymeric gel-based delivery systems, thin polymeric films, and microneedle patches. Most of these devices, currently still in the pre-clinical phase, are intended to be injected or surgically inserted directly into the solid tumor mass. While initial clinical trials have been unsuccessful due to relatively inefficient delivery as compared to direct injection and high immunosuppression, recent research has shown promise in overcoming these barriers.[4]
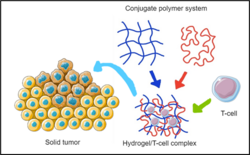
Gel-based delivery
Gel-based delivery of CAR-T cells involves implantation or injection of a hydrogel or polymer gel into the target solid tumor. These gels suspend CAR-T cells in various ways through manipulation of cell-specific chemistry or by fixing the cells in a polymeric matrix. One strength of gel-based delivery is that these systems are functionally biodegradable, so once the CAR-T cells have been administered, the depot does not stay in the body, reducing immunosuppressive conditions or tumor resistance.[24][27]
One of the earliest examples of this methodology developed by Luo et al. in 2020 consisted of a layered hydrogel microchip that facilitated CAR-T survival from immunosuppressive elements of the tumor system which could be injected into the tumor.[28] In addition, this system was able to take advantage of the hypoxic (low oxygen) conditions of the tumor by additionally containing oxygen-releasing agents that when released with interleukin-15 (IL-15) cytokines could cause tumor cell death.[28]
Two recent examples utilizing gel-based delivery towards specific cancers include targeted fibrin gel-based delivery to glioblastoma and modified CAR-T cell hydrogel complexes to retinoblastoma.[29][30] In the glioblastoma-targeting system, Ogunnaike et al. used CAR-T cells loaded in a fibrin matrix, which undergoes in-situ polymerization upon mixture of fibrinogen and thrombin (as part of the coagulation cascade). In addition, the CAR-T cells were modified to target the B7-H3 antigen, present on some forms of cancer including glioblastoma.[29][31] In the retinoblastoma-targeting system, Wang et al. fabricated CAR-T cells specific to the GD2 ganglioside, specific to retinoblastoma, and suspended them into an IL-15/chitosan-polyethylene glycol (PEG) hydrogel suspension, effectively targeting the tumor via injection.[30]
Additionally, researchers have developed hydrogel-based systems that limit further growth and proliferation of tumors, such as the hyaluronic acid hydrogel system developed by Hu et al.[32] This system, which targets melanoma through targeting the CSPG4 antigen, was shown to slowly but efficiently release IL-15 and CAR-T cells from a cross-linked hyaluronic acid matrix and poly(lactic-co-glycolic acid) (PLGA) nanoparticle suspension intratumorally.[32] In addition, the system utilized anti-PDL1 antibody delivery, increasing platelet release of programmed cell death molecules onto the tumors, killing them.[32]
Sustained delivery and long-term retention of CAR-T cells through hydrogel systems has also been developed, as shown by Grosskopf et al., to address controlled release of CAR-T cells onto tumors.[33] In this system, the researchers crosslinked a combination of dodecyl-modified hydroxypropyl methylcellulose (HPMC-C12) and PEG-poly(lactic acid) (PLA) nanoparticles to form the hydrogel, then mixed CAR-T cells and IL-15 into the matrix.[33] The sustained release profiles showed promise in tumor suppression treatments due to the slow release duration profiles.[33]
Codelivery of CAR-T cells with other agonists, such as stimulator of interferon (IFN) genes (STING), remain a popular new method of administration of CAR-T cells to solid tumors.[34] Smith et al. developed a silica nanoparticle based coating conjugated with lipid film to suspend CAR-T cells and select antibodies onto an alginate polymer scaffold. This system was tested in mouse pancreatic cancer and melanoma models, and showed expansion of CAR-T cells at the tumor site, increasing the therapeutic efficiency.
There are still challenges with these delivery systems, however, as they have limitations on CAR-T cell suspension amounts, host-specific immunogenic responses, and have been performed primarily in mouse models.[24][28][33] Regardless, hydrogel-based CAR-T delivery systems have shown promise in translational experimentations towards solid tumor targeting.
Thin Film Delivery
Thin-film targeting has emerged as an alternative method of CAR-T cell delivery to solid tumors, utilizing metal-based microfilms made of nitinol, an alloy of nickel and titanium, a composite commonly used in stents.[17][35] One such example using these nitinol scaffolds was developed by Coon et al., whereby the researchers suspended CAR-T cells into pores laden throughout the film, combined with fibrin.[35]
Upon implantation, the CAR-T cells would release from the film, but remain within the site of the tumor due to engineered antibody attraction of the film.[35] This caused increased localization and duration of CAR-T therapy to the tumor site as compared to intravenous or local injection of CAR-T cells.[35]
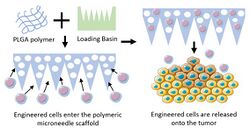
Microneedle Delivery
Microneedle patches have been used previously as a minimally-invasive, distributive system to release drugs across certain body systems, most notably on the skin.[36] Microneedles patches have been used recently as a method of CAR-T cell release to solid tumor cancers such as melanoma and pancreatic cancer.[23][37] Using a porous PLGA microneedle-shaped scaffold, Li et al. developed a system to suspend and release CAR-T cells.[37] An advantage of this method is that the patch could be inserted on the surface of the tumor, allowing for surface-targeting of CAR-T cells upon release. Additionally, the needles would penetrate into the solid tumor, allowing for a distribution of CAR-T cells release along the interior axis of the tumor.[37]
In-situ generation of CAR-T cells
Production of CAR-T cells involve removal of T cells via an extraction process known as leukapheresis, followed by cell culture with viral vectors containing ingredients needed to construct the chimeric system.[2][38][39] These methodologies, while important, have been shown to be expensive and time-consuming.[21][40] Recent advances in research have introduced “scaffold factories” to program, produce, and release autologous CAR-T cells into the body after implantation.[41][42][43][44] These “scaffold factories” have shown to function in targeting hematologic cancers and show promise in future research to target solid tumors.
One method of in-situ generation involves the injection of lentiviral vectors, containing genetic information relating to targeting of CD4 antigens directly to lymphocytes in conjugation with the CAR gene, allowing for the construction of this system within the body, as shown by Agarwal et al.[41] Another method of in-situ generation involves the manufacture polymeric nanocarriers to carry genes involved in the development of the CAR-T cell.[44] Smith et al. used poly(β-amino ester) (PBAE) particles laden with polyglutamic acid (PGA), nuclear localization signals, and T-cell targeting fragments to deliver CAR genes to T cells for translation into CAR-T cells.[44]
These advances have led to the investigation and development of a novel system for manufacturing CAR-T cells known as Multifunctional Alginate Scaffold for T Cell Engineering and Release (MASTER).[43] These alginate scaffolds, which are highly porous, are embedded with azide, cyclooctyne-conjugated targeting antibodies, CAR-based retroviral vectors, and mononuclear blood cells. The azide and cyclooctyne-conjugated particles undergo a rapid click chemistry reaction, whereby when implanted, alongside introduction of the blood cells, the scaffold will generate CAR-T cells. This system was shown to be long-lasting and biodegradable, factors that are important for long suppression of cancers.[44]
See also
- CAR T cell
- Cellular adoptive immunotherapy
- Cancer immunotherapy
- Bioinstructive material
- Cell encapsulation
References
- ↑ 1.0 1.1 1.2 Sterner, Sterner, R.C., R.M. (2021). "CAR-T cell therapy: current limitations and potential strategies". Blood Cancer J 11 (69): 69. doi:10.1038/s41408-021-00459-7. PMID 33824268.
- ↑ 2.0 2.1 "CAR T Cells: Engineering Immune Cells to Treat Cancer". NCI. 6 December 2013. https://www.cancer.gov/about-cancer/treatment/research/car-t-cells.
- ↑ "Approved Cellular and Gene Therapy Products". U.S. Food and Drug Administration (FDA). https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/approved-cellular-and-gene-therapy-products..
- ↑ 4.0 4.1 4.2 4.3 Sagnella, Sharon M.; White, Amy L.; Yeo, Dannel; Saxena, Payal; van Zandwijk, Nico; Rasko, John E.J. (Aug 2022). "Locoregional delivery of CAR-T cells in the clinic" (in en). Pharmacological Research 182: 106329. doi:10.1016/j.phrs.2022.106329. PMID 35772645.
- ↑ 5.0 5.1 5.2 Ding, Yingyue; Wang, Yixin; Hu, Quanyin (Jun 2022). "Recent advances in overcoming barriers to cell‐based delivery systems for cancer immunotherapy" (in en). Exploration 2 (3): 20210106. doi:10.1002/EXP.20210106. ISSN 2766-2098. PMID 37323702.
- ↑ Siddiqi, Haziq F.; Staser, Karl W.; Nambudiri, Vinod E. (Dec 2018). "Research Techniques Made Simple: CAR T-Cell Therapy" (in en). Journal of Investigative Dermatology 138 (12): 2501–2504.e1. doi:10.1016/j.jid.2018.09.002. PMID 30243656.
- ↑ 7.0 7.1 Safarzadeh Kozani, Pooria; Safarzadeh Kozani, Pouya; Rahbarizadeh, Fatemeh; Khoshtinat Nikkhoi, Shahryar (Apr 2021). "Strategies for Dodging the Obstacles in CAR T Cell Therapy". Frontiers in Oncology 11: 627549. doi:10.3389/fonc.2021.627549. ISSN 2234-943X. PMID 33869011.
- ↑ Dasyam, Nathaniel; George, Philip; Weinkove, Robert (Sep 2020). "Chimeric antigen receptor T-cell therapies: Optimising the dose". British Journal of Clinical Pharmacology 86 (9): 1678–1689. doi:10.1111/bcp.14281. ISSN 1365-2125. PMID 32175617.
- ↑ Research, Center for Biologics Evaluation and (2022-07-07). "KYMRIAH (tisagenlecleucel)" (in en). FDA. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/kymriah-tisagenlecleucel.
- ↑ Research, Center for Biologics Evaluation and (2022-11-04). "YESCARTA (axicabtagene ciloleucel)" (in en). FDA. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/yescarta-axicabtagene-ciloleucel.
- ↑ Research, Center for Biologics Evaluation and (2022-04-13). "TECARTUS (brexucabtagene autoleucel)" (in en). FDA. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/tecartus-brexucabtagene-autoleucel.
- ↑ Research, Center for Biologics Evaluation and (2022-07-01). "BREYANZI (lisocabtagene maraleucel)" (in en). FDA. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/breyanzi-lisocabtagene-maraleucel.
- ↑ Research, Center for Biologics Evaluation and (2021-04-21). "ABECMA (idecabtagene vicleucel)" (in en). FDA. https://www.fda.gov/vaccines-blood-biologics/abecma-idecabtagene-vicleucel.
- ↑ Research, Center for Biologics Evaluation and (2023-02-24). "CARVYKTI" (in en). FDA. https://www.fda.gov/vaccines-blood-biologics/carvykti.
- ↑ Ruella, Marco; Xu, Jun; Barrett, David M.; Fraietta, Joseph A.; Reich, Tyler J.; Ambrose, David E.; Klichinsky, Michael; Shestova, Olga et al. (Oct 2018). "Induction of resistance to chimeric antigen receptor T cell therapy by transduction of a single leukemic B cell" (in en). Nature Medicine 24 (10): 1499–1503. doi:10.1038/s41591-018-0201-9. ISSN 1546-170X. PMID 30275568.
- ↑ Mestermann, Katrin; Giavridis, Theodoros; Weber, Justus; Rydzek, Julian; Frenz, Silke; Nerreter, Thomas; Mades, Andreas; Sadelain, Michel et al. (Jul 2019). "The tyrosine kinase inhibitor dasatinib acts as a pharmacologic on/off switch for CAR T cells". Science Translational Medicine 11 (499): eaau5907. doi:10.1126/scitranslmed.aau5907. ISSN 1946-6242. PMID 31270272.
- ↑ 17.0 17.1 17.2 Fu, Ruxing; Li, Hongjun; Li, Ruoxin; McGrath, Kyle; Dotti, Gianpietro; Gu, Zhen (Feb 2021). "Delivery Techniques for Enhancing CAR T Cell Therapy against Solid Tumors". Advanced Functional Materials 31 (44). doi:10.1002/adfm.202009489. https://onlinelibrary.wiley.com/doi/full/10.1002/adfm.202009489.
- ↑ Sridhar, Praveen; Petrocca, Fabio (Jul 2017). "Regional Delivery of Chimeric Antigen Receptor (CAR) T-Cells for Cancer Therapy" (in en). Cancers 9 (7): 92. doi:10.3390/cancers9070092. ISSN 2072-6694. PMID 28718815.
- ↑ Safarzadeh Kozani, Pouya; Safarzadeh Kozani, Pooria; Ahmadi Najafabadi, Milad; Yousefi, Fatemeh; Mirarefin, Seyed Mohamad Javad; Rahbarizadeh, Fatemeh (May 2022). "Recent Advances in Solid Tumor CAR-T Cell Therapy: Driving Tumor Cells From Hero to Zero?". Frontiers in Immunology 13: 795164. doi:10.3389/fimmu.2022.795164. ISSN 1664-3224. PMID 35634281.
- ↑ "Cancer Classification | SEER Training". https://training.seer.cancer.gov/disease/categories/classification.html.
- ↑ 21.0 21.1 Shah, Nirali N.; Fry, Terry J. (Mar 2019). "Mechanisms of resistance to CAR T cell therapy" (in en). Nature Reviews Clinical Oncology 16 (6): 372–385. doi:10.1038/s41571-019-0184-6. ISSN 1759-4774. PMID 30837712.
- ↑ Edeline, Julien; Houot, Roch; Marabelle, Aurélien; Alcantara, Marion (Apr 2021). "CAR-T cells and BiTEs in solid tumors: challenges and perspectives". Journal of Hematology & Oncology 14 (1): 65. doi:10.1186/s13045-021-01067-5. ISSN 1756-8722. PMID 33874996.
- ↑ 23.0 23.1 Wen, Ping; Wu, Wei; Wang, Feifan; Zheng, Hanqi; Liao, Ziyan; Shi, Jiaqi; Zhu, Chaojie; Zhao, Peng et al. (Jan 2023). "Cell delivery devices for cancer immunotherapy" (in en). Journal of Controlled Release 353: 875–888. doi:10.1016/j.jconrel.2022.11.041. PMID 36442617. https://linkinghub.elsevier.com/retrieve/pii/S0168365922007854.
- ↑ 24.0 24.1 24.2 Correa, Santiago; Grosskopf, Abigail K.; Lopez Hernandez, Hector; Chan, Doreen; Yu, Anthony C.; Stapleton, Lyndsay M.; Appel, Eric A. (Sep 2021). "Translational Applications of Hydrogels" (in en). Chemical Reviews 121 (18): 11385–11457. doi:10.1021/acs.chemrev.0c01177. ISSN 0009-2665. PMID 33938724.
- ↑ Yousefpour, Parisa; Ni, Kaiyuan; Irvine, Darrell J. (Jan 2023). "Targeted modulation of immune cells and tissues using engineered biomaterials" (in en). Nature Reviews Bioengineering 1 (2): 107–124. doi:10.1038/s44222-022-00016-2. ISSN 2731-6092. PMID 37772035.
- ↑ Li, Zhaoting; Wang, Yixin; Ding, Yingyue; Repp, Lauren; Kwon, Glen S.; Hu, Quanyin (Jun 2021). "Cell‐Based Delivery Systems: Emerging Carriers for Immunotherapy" (in en). Advanced Functional Materials 31 (23): 2100088. doi:10.1002/adfm.202100088. ISSN 1616-301X. https://onlinelibrary.wiley.com/doi/10.1002/adfm.202100088.
- ↑ Jacob, Shery; Nair, Anroop B.; Shah, Jigar; Sreeharsha, Nagaraja; Gupta, Sumeet; Shinu, Pottathil (Mar 2021). "Emerging Role of Hydrogels in Drug Delivery Systems, Tissue Engineering and Wound Management" (in en). Pharmaceutics 13 (3): 357. doi:10.3390/pharmaceutics13030357. ISSN 1999-4923. PMID 33800402.
- ↑ 28.0 28.1 28.2 Luo, Zuyuan; Liu, Zhen; Liang, Zhen; Pan, Jijia; Xu, Jun; Dong, Jiebin; Bai, Yun; Deng, Hongkui et al. (Dec 2020). "Injectable Porous Microchips with Oxygen Reservoirs and an Immune-Niche Enhance the Efficacy of CAR T Cell Therapy in Solid Tumors" (in en). ACS Applied Materials & Interfaces 12 (51): 56712–56722. doi:10.1021/acsami.0c15239. ISSN 1944-8244. PMID 33306365. https://pubs.acs.org/doi/10.1021/acsami.0c15239.
- ↑ 29.0 29.1 Luo, Zuyuan; Liu, Zhen; Liang, Zhen; Pan, Jijia; Xu, Jun; Dong, Jiebin; Bai, Yun; Deng, Hongkui et al. (Dec 2020). "Injectable Porous Microchips with Oxygen Reservoirs and an Immune-Niche Enhance the Efficacy of CAR T Cell Therapy in Solid Tumors" (in en). ACS Applied Materials & Interfaces 12 (51): 56712–56722. doi:10.1021/acsami.0c15239. ISSN 1944-8244. PMID 33306365. https://pubs.acs.org/doi/10.1021/acsami.0c15239.
- ↑ 30.0 30.1 Wang, Kai; Chen, Yuhui; Ahn, Sarah; Zheng, Min; Landoni, Elisa; Dotti, Gianpietro; Savoldo, Barbara; Han, Zongchao (Oct 2020). "GD2-specific CAR T cells encapsulated in an injectable hydrogel control retinoblastoma and preserve vision" (in en). Nature Cancer 1 (10): 990–997. doi:10.1038/s43018-020-00119-y. ISSN 2662-1347. PMID 33898999.
- ↑ Du, Hongwei; Hirabayashi, Koichi; Ahn, Sarah; Kren, Nancy Porterfield; Montgomery, Stephanie Ann; Wang, Xinhui; Tiruthani, Karthik; Mirlekar, Bhalchandra et al. (Feb 2019). "Antitumor Responses in the Absence of Toxicity in Solid Tumors by Targeting B7-H3 via Chimeric Antigen Receptor T Cells" (in en). Cancer Cell 35 (2): 221–237.e8. doi:10.1016/j.ccell.2019.01.002. PMID 30753824.
- ↑ 32.0 32.1 32.2 Hu, Quanyin; Li, Hongjun; Archibong, Edikan; Chen, Qian; Ruan, Huitong; Ahn, Sarah; Dukhovlinova, Elena; Kang, Yang et al. (Apr 2021). "Inhibition of post-surgery tumour recurrence via a hydrogel releasing CAR-T cells and anti-PDL1-conjugated platelets" (in en). Nature Biomedical Engineering 5 (9): 1038–1047. doi:10.1038/s41551-021-00712-1. ISSN 2157-846X. PMID 33903744.
- ↑ 33.0 33.1 33.2 33.3 Grosskopf, Abigail K.; Labanieh, Louai; Klysz, Dorota D.; Roth, Gillie A.; Xu, Peng; Adebowale, Omokolade; Gale, Emily C.; Jons, Carolyn K. et al. (Apr 2022). "Delivery of CAR-T cells in a transient injectable stimulatory hydrogel niche improves treatment of solid tumors" (in en). Science Advances 8 (14): eabn8264. doi:10.1126/sciadv.abn8264. ISSN 2375-2548. PMID 35394838. Bibcode: 2022SciA....8N8264G.
- ↑ Smith, Tyrel T.; Moffett, Howell F.; Stephan, Sirkka B.; Opel, Cary F.; Dumigan, Amy G.; Jiang, Xiuyun; Pillarisetty, Venu G.; Pillai, Smitha P. S. et al. (Apr 2017). "Biopolymers codelivering engineered T cells and STING agonists can eliminate heterogeneous tumors" (in en). Journal of Clinical Investigation 127 (6): 2176–2191. doi:10.1172/JCI87624. ISSN 0021-9738. PMID 28436934. PMC 5451231. https://www.jci.org/articles/view/87624.
- ↑ 35.0 35.1 35.2 35.3 Coon, Michael E.; Stephan, Sirkka B.; Gupta, Vikas; Kealey, Colin P.; Stephan, Matthias T. (Dec 2019). "Nitinol thin films functionalized with CAR-T cells for the treatment of solid tumours" (in en). Nature Biomedical Engineering 4 (2): 195–206. doi:10.1038/s41551-019-0486-0. ISSN 2157-846X. PMID 31819155. https://www.nature.com/articles/s41551-019-0486-0.
- ↑ Aldawood, Faisal Khaled; Andar, Abhay; Desai, Salil (Aug 2021). "A Comprehensive Review of Microneedles: Types, Materials, Processes, Characterizations and Applications" (in en). Polymers 13 (16): 2815. doi:10.3390/polym13162815. ISSN 2073-4360. PMID 34451353.
- ↑ 37.0 37.1 37.2 Li, Hongjun; Wang, Zejun; Ogunnaike, Edikan A; Wu, Qing; Chen, Guojun; Hu, Quanyin; Ci, Tianyuan; Chen, Zhaowei et al. (Mar 2022). "Scattered seeding of CAR T cells in solid tumors augments anticancer efficacy" (in en). National Science Review 9 (3): nwab172. doi:10.1093/nsr/nwab172. ISSN 2095-5138. PMID 35265340.
- ↑ Zhang, Cheng; Liu, Jun; Zhong, Jiang F.; Zhang, Xi (Jun 2017). "Engineering CAR-T cells". Biomarker Research 5: 22. doi:10.1186/s40364-017-0102-y. ISSN 2050-7771. PMID 28652918.
- ↑ Jackson, Hollie J.; Rafiq, Sarwish; Brentjens, Renier J. (Jun 2016). "Driving CAR T-cells forward" (in en). Nature Reviews Clinical Oncology 13 (6): 370–383. doi:10.1038/nrclinonc.2016.36. ISSN 1759-4774. PMID 27000958.
- ↑ Bach, Peter B. (Oct 2018). "National Coverage Analysis of CAR-T Therapies — Policy, Evidence, and Payment" (in en). New England Journal of Medicine 379 (15): 1396–1398. doi:10.1056/NEJMp1807382. ISSN 0028-4793. PMID 30110578. http://www.nejm.org/doi/10.1056/NEJMp1807382.
- ↑ 41.0 41.1 Agarwal, Shiwani; Hanauer, Julia D.S.; Frank, Annika M.; Riechert, Vanessa; Thalheimer, Frederic B.; Buchholz, Christian J. (Aug 2020). "In Vivo Generation of CAR T Cells Selectively in Human CD4+ Lymphocytes" (in en). Molecular Therapy 28 (8): 1783–1794. doi:10.1016/j.ymthe.2020.05.005. PMID 32485137.
- ↑ Xin, Tianqing; Cheng, Li; Zhou, Chuchao; Zhao, Yimeng; Hu, Zhenhua; Wu, Xiaoyan (Feb 2022). "In-Vivo Induced CAR-T Cell for the Potential Breakthrough to Overcome the Barriers of Current CAR-T Cell Therapy". Frontiers in Oncology 12: 809754. doi:10.3389/fonc.2022.809754. ISSN 2234-943X. PMID 35223491.
- ↑ 43.0 43.1 Agarwalla, Pritha; Ogunnaike, Edikan A.; Ahn, Sarah; Froehlich, Kristen A.; Jansson, Anton; Ligler, Frances S.; Dotti, Gianpietro; Brudno, Yevgeny (Aug 2022). "Bioinstructive implantable scaffolds for rapid in vivo manufacture and release of CAR-T cells" (in en). Nature Biotechnology 40 (8): 1250–1258. doi:10.1038/s41587-022-01245-x. ISSN 1087-0156. PMID 35332339.
- ↑ 44.0 44.1 44.2 44.3 Smith, Tyrel T.; Stephan, Sirkka B.; Moffett, Howell F.; McKnight, Laura E.; Ji, Weihang; Reiman, Diana; Bonagofski, Emmy; Wohlfahrt, Martin E. et al. (Aug 2017). "In situ programming of leukaemia-specific T cells using synthetic DNA nanocarriers" (in en). Nature Nanotechnology 12 (8): 813–820. doi:10.1038/nnano.2017.57. ISSN 1748-3387. PMID 28416815.
 |